Unit Test (Solutions): The Amazing World of Solutes, Solvents, and Solutions
Time: 1 hour
M.M. 30
Attempt all questions.
- Question numbers 1 to 5 carry 1 mark each.
- Question numbers 6 to 8 carry 2 marks each.
- Question numbers 9 to 11 carry 3 marks each.
- Question numbers 12 & 13 carry 5 marks each.
- 1-mark questions include MCQs.
Q1: A solution is defined as (1 Mark)
(i) any mixture of two solids
(ii) a uniform mixture in which components are evenly distributed
(iii) a non-uniform mixture where components settle
(iv) only liquids mixed with liquids
Ans: (ii)
Explanation: A solution is a uniform (homogeneous) mixture. For example, when salt or sugar dissolves in water the particles spread evenly throughout the liquid, so every sip tastes the same because the components are evenly distributed.
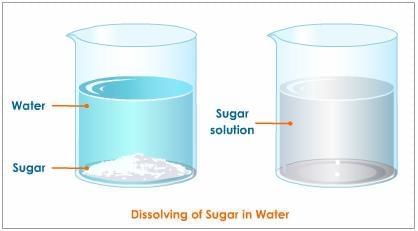
Q2: In the solution of salt in water, salt is the ______ and water is the ______. (1 Mark)
(i) solvent, solute
(ii) solute, solvent
(iii) suspension, colloid
(iv) liquid, solid
Ans: (ii)
Explanation: The substance that is dissolved (present in smaller amount) is called the solute (salt). The substance that dissolves the solute (present in larger amount) is called the solvent (water).
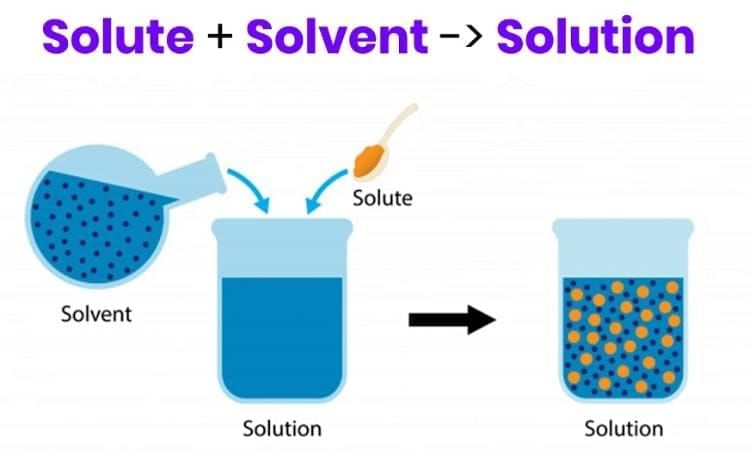
Q3: When no more solute can dissolve in a solution at a given temperature, the solution is (1 Mark)
(i) dilute
(ii) concentrated
(iii) saturated
(iv) supersaturated at all temperatures
Ans: (iii)
Explanation: A saturated solution contains the maximum amount of solute that can dissolve at that temperature. If more solute is added, it will remain undissolved because the solution cannot dissolve any more at that temperature.
Q4: Generally, the solubility of most solids in liquids ______ with increase in temperature, while the solubility of gases in liquids ______. (1 Mark)
(i) decreases, increases
(ii) increases, decreases
(iii) unchanged, unchanged
(iv) decreases, decreases
Ans: (ii)
Explanation: For most solids (for example, sugar), solubility increases when temperature rises, so warmer water can dissolve more solid. For gases (for example, oxygen), solubility decreases with higher temperature, so cold water holds more dissolved gas than warm water.
Q5: Density is defined as (1 Mark)
(i) mass × volume
(ii) volume/mass
(iii) mass/volume
(iv) weight/area
Ans: (iii)
Explanation: Density is mass divided by volume. Common units used in this chapter are g/cm3 or g/mL for solids and liquids.
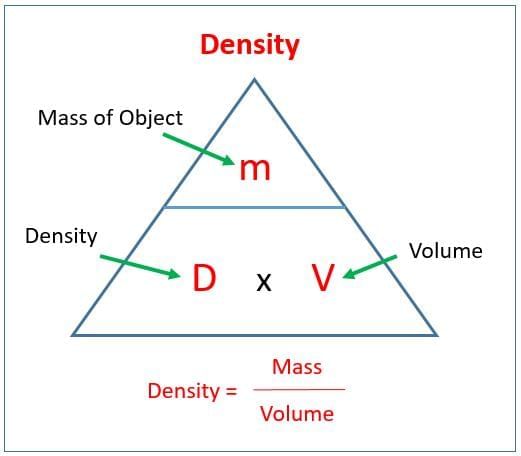
Q6: State the difference between a saturated and an unsaturated solution with one example from class activities. (2 Marks)
Ans: A saturated solution has dissolved the maximum amount of solute possible at a given temperature. Example: salt in water where some crystals remain undissolved at the bottom shows saturation. An unsaturated solution can still dissolve more solute at the same temperature. Example: when a few spoonfuls of salt completely dissolve in water with nothing left over, the solution is unsaturated.
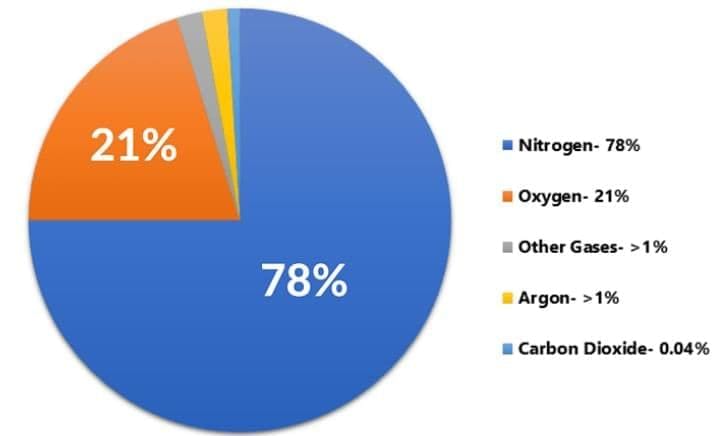
Q7: Give one reason why air is considered a solution. Identify the solvent in air. (2 Marks)
Ans: Air is a uniform mixture of different gases, so it is a gaseous solution. Nitrogen is present in the largest proportion and therefore acts as the solvent; oxygen, argon, carbon dioxide and other gases are the solutes.
Q8: Explain why oxygen dissolves more in cold water than in hot water. How is this important for aquatic life? (2 Marks)
Ans: Gas solubility decreases with increasing temperature, so cold water can hold more dissolved oxygen than warm water. This is important because aquatic organisms such as fish and water plants need dissolved oxygen to breathe; therefore colder waters generally provide more oxygen and support greater aquatic life.
Q9: In an experiment, baking soda is added to water at 20 °C until some remains undissolved. On heating to 50 °C, the undissolved part disappears. Explain what this shows about solubility and the status of the solution at the higher temperature. (3 Marks)
Ans: The observation shows that the solubility of the solid (baking soda) increases with temperature. At 20 °C the solution was saturated because extra baking soda remained undissolved. When the temperature rose to 50 °C, the solution became unsaturated at that higher temperature, so more solute dissolved until the new saturation point for 50 °C was reached.
Q10: Define concentration in simple terms. Which is more concentrated and why: (a) 2 spoons of salt in 100 mL water or (b) 4 spoons of salt in 50 mL water? (3 Marks)
Ans: Concentration tells how much solute is present in a given amount of solution or solvent. Comparing the two cases: (a) has 2 spoons in 100 mL, while (b) has 4 spoons in 50 mL. Using the same spoon size, (b) has much more solute in a much smaller volume, so (b) is more concentrated. (For example, (a) gives 0.02 spoon per mL, whereas (b) gives 0.08 spoon per mL.)
Q11: A student mixes sawdust in water and notices it floats, while sand sinks. Use density ideas to explain both observations. (3 Marks)
Ans: Whether an object floats or sinks depends on its average density compared with water. Sawdust has a lower average density than water, so it floats. Sand has a higher density than water, so it sinks. Trapped air or the shape of particles can also help floating, but the main reason here is the difference in density.
Q12: Measurement and density applications. (5 Marks)
(a) Describe how to measure 50 mL of water accurately in a measuring cylinder (mention meniscus and eye position).
(b) A stone has mass 16.400 g. Using water displacement, its volume increases from 50 mL to 55 mL after immersion. Calculate its density.
(c) Predict qualitatively how heating a liquid generally affects its density and why.
Ans:
(a) Place the measuring cylinder on a flat, level surface. Pour the water slowly near the 50 mL mark. Bend down so your eye is at the same level as the mark and read the volume from the bottom of the meniscus (the curved surface of the liquid). This avoids parallax error.
(b) Mass of stone = 16.400 g.
Initial volume of water = 50 mL; final volume = 55 mL.
Displaced volume = 55 mL - 50 mL = 5 mL = 5 cm3.
Density = mass ÷ volume = 16.400 g ÷ 5 cm3 = 3.28 g/cm3.
(c) Heating a liquid generally causes its particles to move apart, increasing its volume while its mass stays the same. Since density = mass/volume, the density usually decreases on heating; therefore warm liquids are typically less dense than cold ones.
Q13: (a) Give one everyday example showing that solutions can be formed from gases too; identify solute and solvent.
(b) An unpeeled orange floats while a peeled one sinks. Explain.
(c) A 2 L bottle currently holds 500 mL of water. How much more water can it hold? If filled completely, what mass of water would it contain approximately at room temperature? (5 Marks)
Ans:
(a) Air itself is a gaseous solution: nitrogen is the solvent (the major component) and oxygen, argon, carbon dioxide etc. are the solutes, all mixed uniformly.
(b) The unpeeled orange floats because its peel contains many tiny air pockets, which lower the orange's overall (average) density so it is less than that of water. When the peel is removed, the trapped air is lost, the average density increases and the peeled orange becomes denser than water and sinks.
(c) Total capacity = 2 L = 2000 mL.
Current amount = 500 mL.
Additional water it can hold = 2000 mL - 500 mL = 1500 mL = 1.5 L.
If completely filled, the bottle would contain 2000 mL of water. At room temperature 1 mL of water ≈ 1 g, so mass ≈ 2000 g = 2.0 kg of water.
FAQs on Unit Test (Solutions): The Amazing World of Solutes, Solvents, and Solutions
| 1. What is the difference between a solute and a solvent in a solution? |  |
| 2. How do temperature and pressure affect the solubility of substances? |  |
| 3. What are some common examples of solutions in everyday life? |  |
| 4. What is saturation in the context of solutions? |  |
| 5. How can we separate solutes from solvents in a solution? |  |






