MCQ (Solution) - The Amazing World of Solutes, Solvents, and Solutions
Question 1:
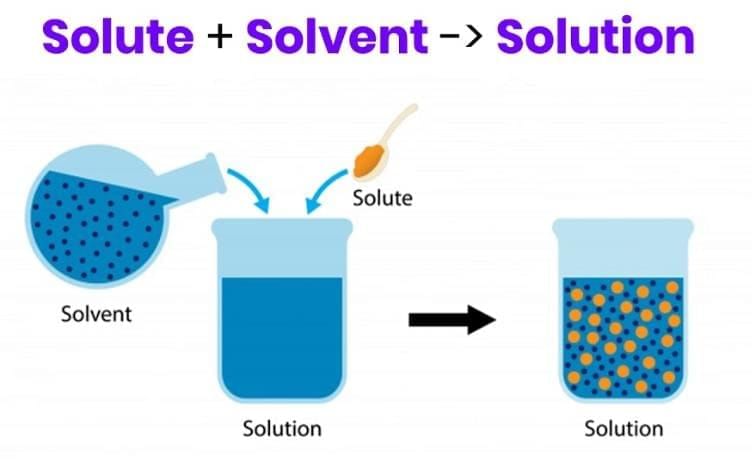
In a solution of sugar in water, which terms correctly identify the roles of sugar and water?
Option A: Sugar is the solvent; water is the solute
Option B: Both are solutes
Option C: Sugar is the solute; water is the solvent
Option D: Both are solvents
Question 2:
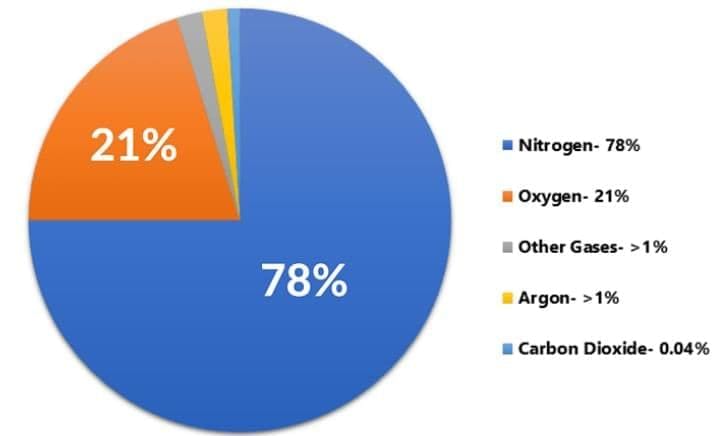
Air is considered a gaseous solution because:
Option A: Nitrogen chemically binds to oxygen
Option B: Gases are evenly mixed; nitrogen is the solvent and other gases are solutes
Option C: Air contains dust
Option D: Air is a pure compound
Question 3:
A solution in which no more solute dissolves at a given temperature is called:
Option A: Dilute solution
Option B: Unsaturated solution
Option C: Saturated solution
Option D: Colloid
Question 4:
What is the most direct way to make a saturated sugar solution start dissolving more sugar again?
Option A: Cool the solution
Option B: Heat the solution
Option C: Stop stirring
Option D: Add sand first
Question 5:
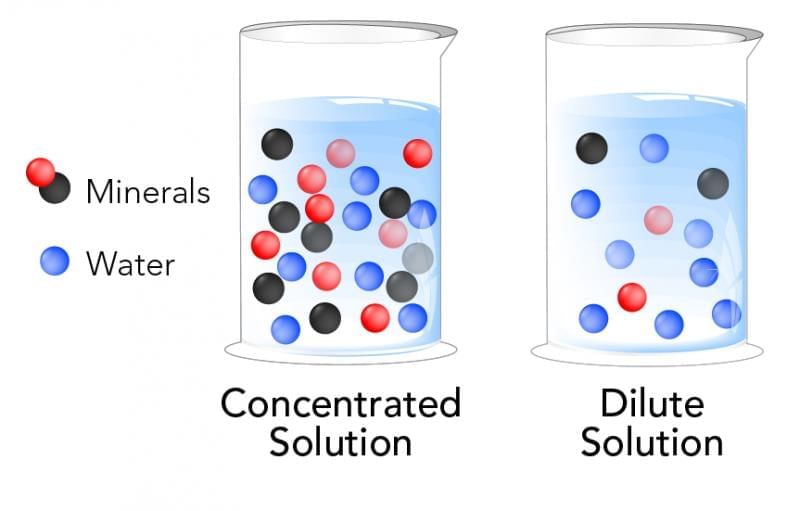
Which statement correctly compares concentrated and dilute solutions?
Option A: Concentrated has more solute per fixed amount of solution than dilute
Option B: Dilute has more solute per fixed amount of solution than concentrated
Option C: Both have the same amount of solute
Option D: Dilute and concentrated are absolute, not relative
Question 6:
Which change in temperature generally increases the solubility of a gas in water?
Option A: Increasing temperature
Option B: Decreasing temperature
Option C: Holding temperature constant
Option D: Heating and cooling alternately
Question 7:
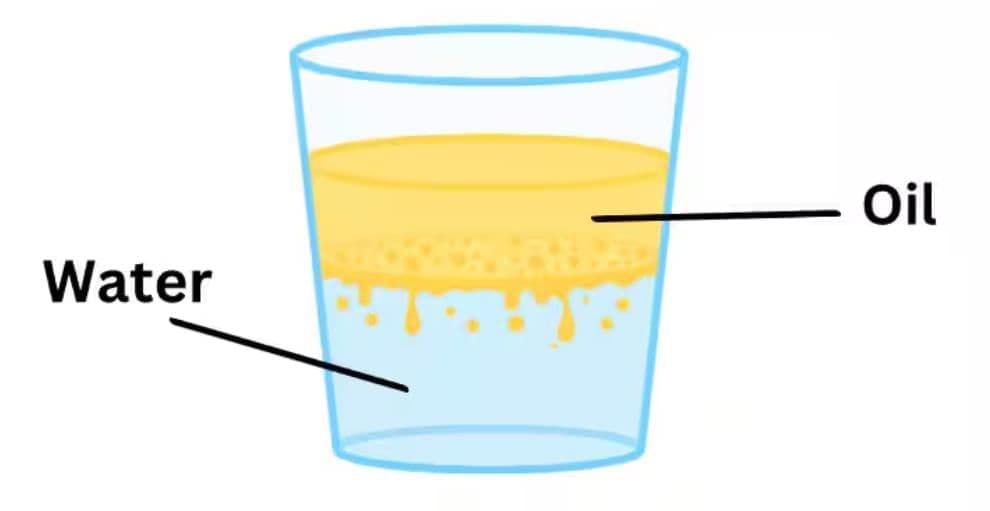
Which best explains why oil floats on water in a glass?
Option A: Oil has higher density than water
Option B: Oil and water chemically react
Option C: Oil has lower density and is immiscible with water
Option D: Oil is the solvent for water
Question 8:
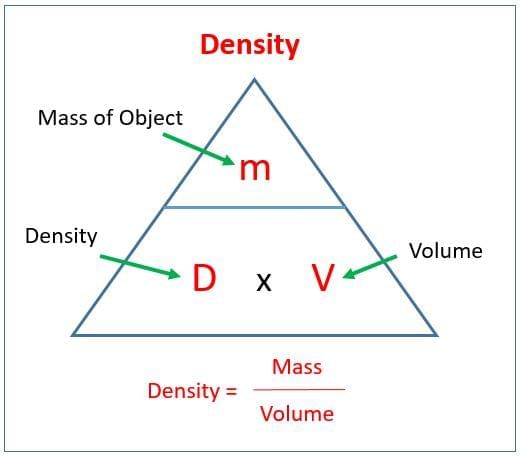
Which formula and SI unit pair for density is correct?
Option A: Density = Volume/Mass; unit m³/kg
Option B: Density = Mass/Volume; unit kg/m³
Option C: Density = Mass × Volume; unit kg·m³
Option D: Density = Mass/Area; unit kg/m²
Question 9:
Why are measuring cylinders tall and narrow rather than short and wide for the same capacity?
Option A: To get a higher, clearer meniscus and finer scale divisions for better accuracy
Option B: To reduce glass usage only
Option C: To prevent liquids from evaporating
Option D: To make pouring impossible
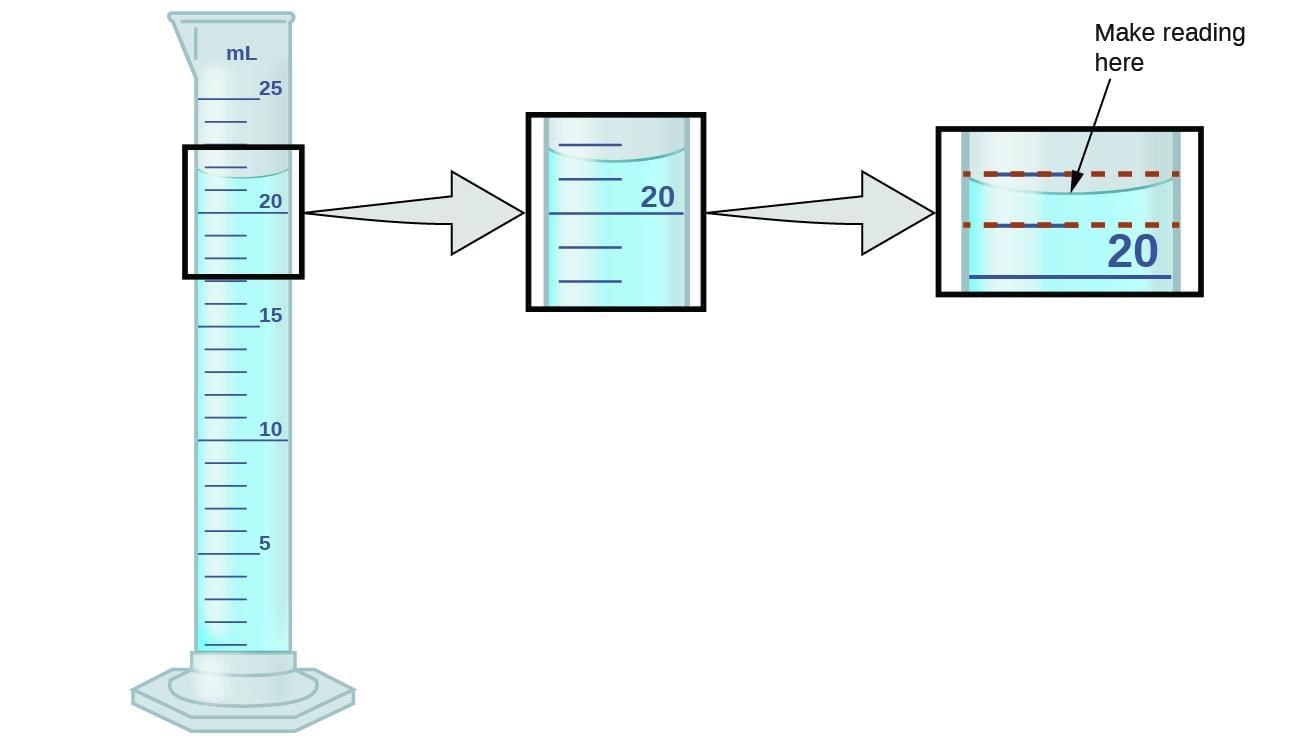
Question 10:
Which reading rule for a colourless liquid in a measuring cylinder is correct?
Option A: Read at the top of the meniscus, from above eye level
Option B: Read at the bottom of the meniscus, with eyes level to it
Option C: Read any visible line, eye position doesn't matter
Option D: Always add 1 mL for meniscus
Question 11:
An object has mass 16.400 g and displaces water from 50 mL to 55 mL in a cylinder. Its density is:
Option A: 0.30 g/cm³
Option B: 0.91 g/cm³
Option C: 16.40 g/cm³
Option D: 3.28 g/cm³
Question 12:
Which statement about temperature's effect on density is generally true?
Option A: Heating increases density of all substances
Option B: Heating decreases density because volume expands while mass stays same
Option C: Temperature has no effect on density
Option D: Cooling always decreases density
Question 13:
Why does ice float on liquid water?
Option A: Ice is denser because it is colder
Option B: Ice has a structure that occupies more volume for the same mass, making it less dense than water
Option C: Water loses mass on cooling
Option D: Floating objects must be heavier
Question 14:
Which change increases a gas's density most directly?
Option A: Increase pressure at constant temperature
Option B: Decrease pressure at constant temperature
Option C: Heat the gas at constant pressure
Option D: Remove mass but keep volume same
Question 15:
Two objects have these mass-volume pairs: A(200 g, 40 cm³), B(240 g, 60 cm³). Which is denser and what does that imply about floating in water?
Option A: A is denser (5.0 g/cm³) and sinks; B is 4.0 g/cm³ and sinks
Option B: A is less dense than B and floats
Option C: Both are 1.0 g/cm³ and float
Option D: B is denser (6.0 g/cm³) and floats
FAQs on MCQ (Solution) - The Amazing World of Solutes, Solvents, and Solutions
| 1. What is the difference between a solute and a solvent? |  |
| 2. How does temperature affect the solubility of a substance? |  |
| 3. What are some common examples of solutions in everyday life? |  |
| 4. What is the process of dissolution, and what factors influence it? |  |
| 5. Can solutions be separated back into their components? If so, how? |  |






