CBSE Class 8 > Class 8 Notes > Science > NCERT Based Activity: Nature of Matter: Elements, Compounds, and Mixtures
NCERT Based Activity: Nature of Matter: Elements, Compounds, and Mixtures
Activity 8.1: Let us experiment
- Take a glass tumbler and fill it half with water.
- Add a small amount of calcium oxide (quick lime) slowly to it.
- What do you observe?
- Stir continuously to make a solution of calcium hydroxide. This solution is called lime water.
- Filter it and observe its colour.
- Leave this colourless solution in a petri dish for a few hours (Fig. 8.5a).
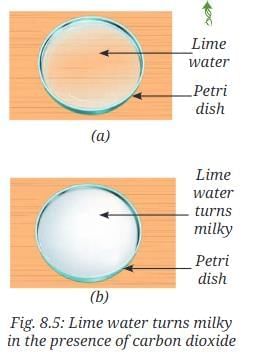
- Keep stirring the solution at regular intervals.
- What do you observe (Fig. 8.5b)?
- Does it turn milky?
- Can you explain why the solution has turned milky?
Answer:
- Observation: When calcium oxide is added to water, it reacts vigorously and the mixture becomes warm. The filtered solution is initially colourless. After a few hours the solution turns milky.
- Explanation and chemical equations: Calcium oxide reacts with water to form calcium hydroxide (lime water):
CaO + H2O → Ca(OH)2. Carbon dioxide from the air dissolves in lime water and reacts with calcium hydroxide to form insoluble calcium carbonate, which makes the solution milky:
Ca(OH)2 + CO2 → CaCO3 + H2O. The formation of the white precipitate CaCO3 causes the milky appearance and demonstrates the presence of CO2 in air.
Activity 8.2: Let us explore
- Take a black sheet of paper. Ensure that it is free from any visible dust particles.
- Place the black sheet of paper undisturbed near an open window (Fig. 8.6a), or in the garden, for a few hours.
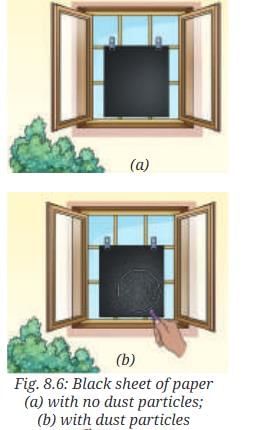
- What do you observe?
Answer:
- Observation: Tiny particles settle on the black sheet and become visible. Under a magnifying glass, these particles can be seen more clearly.
- Explanation: The particles are dust suspended in air. They settle out when the air is still. These dust particles are not a part of the air itself but are solid impurities or pollutants suspended in it. The experiment shows that air contains suspended particles which can be collected on surfaces.
Activity 8.3: Let us experiment (Demonstration activity)
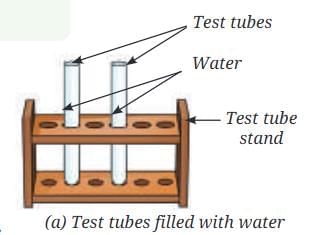
- Collect two small test tubes, a beaker or a glass tumbler, and a 9 V battery.
- Fill 2/3rd of the beaker with water and add a few drops of dilute sulfuric acid to it.
- Fill both the small test tubes completely with water taken from the beaker (Fig. 8.8a).
- Place a 9 V battery inside the beaker (Fig. 8.8b).
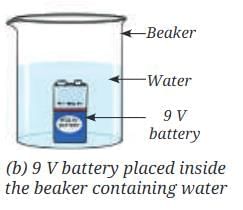
- Without spilling the water, carefully place the water-filled test tubes on each of the terminals of the battery (Fig. 8.8c).
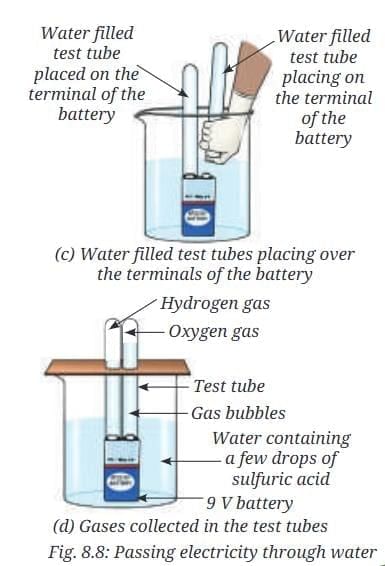
- Wait for a few minutes.
- Do you observe the formation of any gas bubbles at both the terminals inside the test tubes?
- Let it continue for 10-15 minutes.
- Observe the volume of gas collected in each test tube (Fig. 8.8d).
- Is the volume of the gas collected the same in both the test tubes?
- Remove these test tubes one-by-one carefully.
- Test these gases one-by-one by bringing a burning candle close to the mouth of the test tubes.
- What happens in each case?
- Which gas is present in each test tube?
- Can these collected gases be water vapour?
Answer:
- Observation: Bubbles appear at both terminals; the volumes of gas collected in the two test tubes are not the same. On testing, one tube gives a characteristic 'pop' sound with a burning splint (hydrogen), while the other supports combustion or rekindles a glowing splint (oxygen).
- Explanation and chemistry: Electric current splits water into two gases: hydrogen collects at the negative terminal (cathode) and oxygen at the positive terminal (anode). The overall reaction for the electrolysis of water is:
2 H2O (l) → 2 H2 (g) + O2 (g). This gives a volume ratio of hydrogen to oxygen of 2:1, so the test tube at the negative terminal usually contains about twice the volume of gas as the positive terminal. The dilute H2SO4 acts only as an electrolyte to increase conductivity; it is not electrolysed in significant amounts in this demonstration. The gases are not water vapour because they do not condense back into water on cooling and they show the characteristic tests: hydrogen burns with a pop and oxygen supports glowing splints.
Activity 8.4: Let us experiment
- Put a teaspoon of sugar in a boiling tube.
- Heat it gently as shown in Fig. 8.12a.
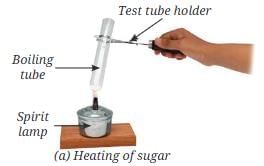
- What do you observe?
Answer:
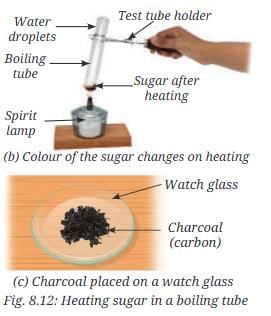
- Observation: On gentle heating, sugar first melts and turns brown, then chars and becomes black. Small droplets of water may form on the cooler parts of the tube near the open end.
- Explanation and chemical change: On heating, sucrose decomposes to form carbon (char) and water vapour. A simplified balanced decomposition is often written as:
C12H22O11 → 12 C + 11 H2O. The brown and then black material is carbon (char), and the droplets indicate release and subsequent condensation of water formed during decomposition. This behaviour shows that sugar is a compound composed of carbon, hydrogen and oxygen; heating breaks the chemical bonds and produces new substances with different properties.
Activity 8.5: Let us experiment (Demonstration activity)
- Take 5.6 grams of iron filings (Fig. 8.13a) and 3.2 grams of sulfur powder (Fig. 8.13b) on a watch glass. Observe them carefully.

- Mix them thoroughly in a watch glass. Label this mixture as Sample A (Fig. 8.14).
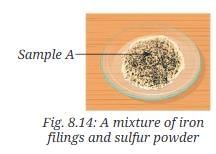
- Observe it carefully.
- Is this a uniform or a non-uniform mixture? Can you still observe both iron and sulfur as separate substances?
- Take half of Sample A in a china dish and gently heat it (Fig. 8.15a) with continuous stirring until a black mass is formed.
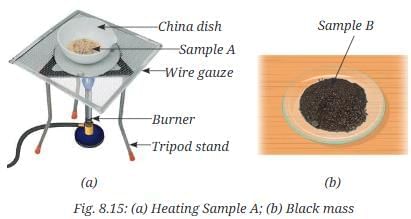
- Let the content of the china dish cool.
- Place this black mass in a mortar and grind it with the help of a pestle.
- Now, put it on another watch glass and label it as Sample B (Fig. 8.15b).
- Now, you have two samples-Sample A and Sample B (Fig. 8.16a and 8.16b). Compare both the Samples A and B step by step and record your observations in Table 8.2.

- Step 1-Appearance: Compare the appearance of Sample A and Sample B like colour and texture.
- Step 2-Magnet test: Take a magnet and move it over the Samples A (Fig. 8.17a) and B (Fig. 8.17b), one by one.
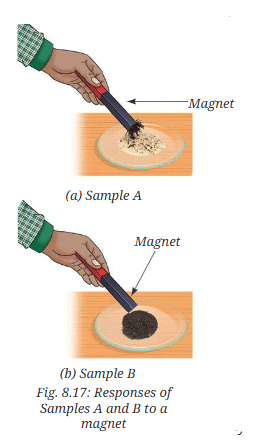
- What do you observe?
- Step 3-Gas test: Take a small amount of Sample A in a test tube and add a few drops of dilute hydrochloric acid (Fig. 8.18a).
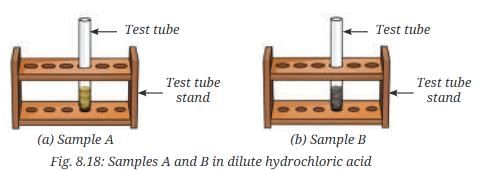
- What do you observe?
- Gently smell the evolved gas by wafting it towards your nose (Fig. 8.19).

- Test the evolved gas by bringing a burning splinter or a lighted candle near the mouth of the test tube (Fig. 8.20a).
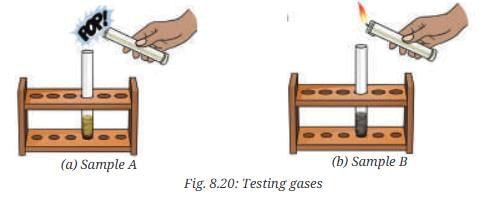
- What do you observe?
- Repeat the above steps with Sample B as well (Fig. 8.18b and 8.20b).
Answer:
- Observation for Sample A (mixture): The mixture of iron filings and sulfur is a heterogeneous (non-uniform) mixture. Iron particles and sulfur particles can be seen separately; the magnet attracts iron filings from the mixture. When dilute hydrochloric acid is added to the mixture, iron reacts with acid to produce hydrogen gas (which gives a pop with a burning splint) while the sulfur remains unreacted.
- When heated (formation of Sample B): Heating the mixture causes a chemical reaction between iron and sulfur to form a new substance, iron(II) sulfide (FeS), which appears as a black, uniform mass. Grinding it gives a fine, uniformly black powder. The magnet does not attract Sample B.
- Chemical equations and tests:
Formation of iron sulfide on heating:
Fe + S → FeS (one atom of Fe combines with one atom of S to form iron sulphide).
Reaction of iron in the mixture with hydrochloric acid:
Fe + 2 HCl → FeCl2 + H2↑ (hydrogen gas is produced and gives a pop on ignition).
Reaction of iron sulphide with dilute hydrochloric acid:
FeS + 2 HCl → FeCl2 + H2S↑ (hydrogen sulphide gas is produced; it is colourless but has a rotten-egg smell and can be detected by wafting).
The change in properties-loss of magnetic attraction and production of H2S on reaction with acid-confirms that Sample B is a compound with properties different from the original iron and sulfur. - Conclusion: Sample A is a physical mixture of iron and sulfur (components retain their identities). Sample B is a chemical compound (iron sulphide) formed by a chemical change during heating; its properties are different from those of iron and sulfur.
The document NCERT Based Activity: Nature of Matter: Elements, Compounds, and Mixtures is a part of the Class 8 Course Science Class 8.
All you need of Class 8 at this link: Class 8
FAQs on NCERT Based Activity: Nature of Matter: Elements, Compounds, and Mixtures
| 1. What are elements, compounds, and mixtures? |  |
Ans.Elements are pure substances that consist of only one type of atom, such as hydrogen (H) or oxygen (O). Compounds are substances formed when two or more elements chemically combine in fixed ratios, like water (H₂O) or carbon dioxide (CO₂). Mixtures are combinations of two or more substances that retain their individual properties, such as air or salad.
| 2. How can we differentiate between a compound and a mixture? |  |
Ans.The main difference between a compound and a mixture is that compounds are formed through chemical reactions and have a fixed composition, whereas mixtures can be separated by physical methods and do not have a fixed composition. For example, saltwater is a mixture of salt and water that can be separated by evaporation, while sodium chloride (table salt) is a compound that cannot be separated by simple physical means.
| 3. What are some common methods to separate mixtures? |  |
Ans.Common methods to separate mixtures include filtration, which separates solids from liquids; distillation, which separates liquids based on differences in boiling points; and chromatography, which separates substances based on their movement through a medium. Each method utilizes the physical properties of the components in the mixture.
| 4. Why is understanding the nature of matter important? |  |
Ans.Understanding the nature of matter is crucial because it forms the basis of chemistry and helps us comprehend the material world. It allows us to categorize substances, understand their properties and reactions, and apply this knowledge in various fields such as medicine, engineering, and environmental science.
| 5. What experiments can illustrate the concepts of elements, compounds, and mixtures? |  |
Ans.Experiments such as creating a simple solution (like saltwater) to demonstrate a mixture, reacting vinegar with baking soda to form a compound (carbon dioxide gas), or separating a mixture of sand and salt using water and filtration can effectively illustrate these concepts. These hands-on activities help reinforce theoretical knowledge through practical application.
Related Searches
Compounds, Compounds, mock tests for examination, pdf , Sample Paper, past year papers, MCQs, and Mixtures, shortcuts and tricks, Important questions, Exam, NCERT Based Activity: Nature of Matter: Elements, Summary, and Mixtures, and Mixtures, Objective type Questions, video lectures, Previous Year Questions with Solutions, practice quizzes, Viva Questions, Semester Notes, Compounds, NCERT Based Activity: Nature of Matter: Elements, Extra Questions, ppt, study material, Free, NCERT Based Activity: Nature of Matter: Elements;






