NEET PG Exam > NEET PG Notes > Gynaecology and Obstetrics > Chapter Notes: Trophoblast disease
Chapter Notes: Trophoblast disease
Introduction
- Trophoblast disease happens when the tissue from pregnancy that helps form the placenta grows in an unusual way.
- It forms a range of conditions from the mostly harmless partial hydatidiform mole up to the very dangerous choriocarcinoma and placental site trophoblast tumours.
- The study of how these diseases work, how to find them, and how to treat them, combined with their effects on mental health, makes trophoblast disease a very key and fascinating area in women's health care and cancer care.
- Even though these conditions do not happen often, the total success rate in curing them is more than 95 percent.
- Using treatments that have been set up for more than 20 years, most patients with trophoblast disease, including those with spread-out disease, can be treated with a strong hope of full cure and with very little long-lasting harm.
- Because the current medical treatments work so well, the main changes in handling trophoblast disease in the UK now aim at better helpful care.
- These areas cover plans for making sure human chorionic gonadotropin monitoring happens after molar pregnancies, better ways in pathology reports, and more knowledge in clinical settings for finding choriocarcinoma and placental site tumours early.
- Starting from 1973, the UK has had a central setup for watching over, following up, and treating these cases, and a lot of this chapter comes from the knowledge gained at the National Trophoblast Tumour Centre at Charing Cross Hospital in London.
Classifcation
- The World Health Organization organizes trophoblast disease into pre-malignant categories like partial and complete hydatidiform moles, and malignant types such as invasive mole, choriocarcinoma, and placental site tumors.
- There are some differences across regions and ethnic groups, with possibly higher rates in Africa and Asia, due to varying standards in how often and how accurately pathology reporting makes comparisons difficult.
- However, the reported frequency of molar pregnancies in Europe and North America is typically between 0.2 to 1.5 per 1000 live births, though this data has some limitations in precision.
- The likelihood of hydatidiform mole increases at the extremes of childbearing age, with a slight increase in teenagers (about 1.3 times higher) and a much greater increase (up to 10 times) in women over 40 years old.
- Historically, the incidence of choriocarcinoma and complete molar pregnancy was estimated at 3 in 1000 and 1 in 1000 respectively, but this might overestimate due to potential misdiagnosis of partial moles. Around 40 percent of partial moles sent for expert review are reclassified as either complete moles or non-molar conditions.
Premalignant pathology and presentation
Partial mole
- The way partial moles start from genes is shown in Image.
- Partial moles have three sets of chromosomes, with two from the father and one from the mother.
- On a large scale, partial moles often look like normal pregnancy results, with a baby starting that usually stops growing by week 8 or 9.
- The tissue study shows less swelling in the chorionic villi compared to complete mole, and there are usually only changes in some areas.
- Because of this, finding partial mole can often be overlooked after a loss of pregnancy or ending it.
- The usual way partial mole shows up is through uneven bleeding or found during normal ultrasound checks.
- The way to handle it in pregnancy care is by using suction to remove it, and then all these patients need to be watched with regular human chorionic gonadotropin measures.
- About 4 percent move to bad disease rarely, with usually only one or two cases of bad disease seen each year at Charing Cross Hospital, with a total chance of 0.5 percent needing drug treatment after a partial mole.
Complete mole
- In most complete moles, all the gene material comes from the male and happens from the joining with an empty egg cell without mother DNA.
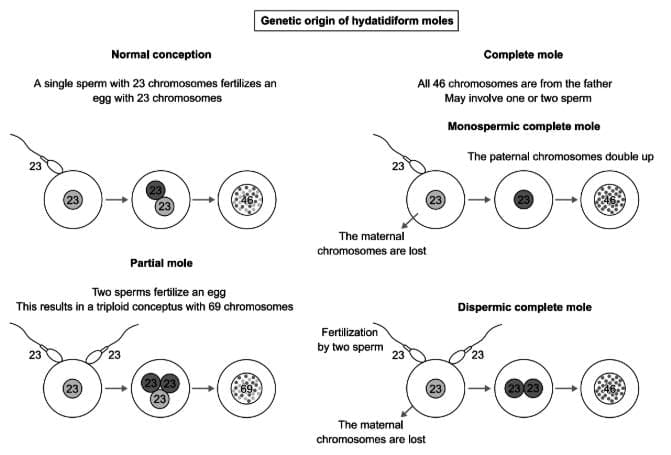
Genetic formation of hydatidiform moles.
- Oocyte lacking maternal DNA. The chromosome count is most commonly 46XX, which results from one sperm that duplicates its DNA, or less frequently 46XX or 46XY from the presence of two different sperms.
- The clinical diagnosis of CM is most often from a large for date uterus or an abnormal ultrasound.
- Macroscopically there is no visible foetal material although microscopically some embryonic cells can be present.
- The histology shows the characteristic oedematous villous stroma, however, the textbook bunch of grapes appearance is only seen in the second trimester and as most cases are diagnosed earlier, this is now rarely seen.
- Normal conception: A single sperm with 23 chromosomes fertilizes an egg with 23 chromosomes.
- Complete mole: All 46 chromosomes are from the father. May involve one sperm where paternal chromosomes double up and maternal chromosomes are lost.
- Partial mole: Two sperms fertilize an egg leading to a triploid with 69 chromosomes.
- Dispermic complete mole: Fertilization by two sperms where maternal chromosomes are lost.
- Different from partial mole, complete mole more often goes to invasive disease with 8 to 20 percent of patients needing drug treatment
Registration and surveillance
- Most patients with molar pregnancies will turn out to need no more treatment after the removal.
- But the remaining cells of the trophoblast tissue will not die and keep growing and their numbers will reduce the human chorionic gonadotropin levels back to normal.
- However, at present the system that works best prognostic allows distinction between the patients who after removal will develop invasive disease and the majority who will not.
- As a result all molar pregnancy patients should be registered for a human chorionic gonadotropin follow-up system.
- The use of this system allows the early finding of patients whose disease is continuing to grow, while also allowing the care-full watch of patients with more slowly falling human chorionic gonadotropin, so producing minimum situation of unnecessary chemotherapy.
- Analysis of the natural history of the illness in this surveillance phase has allowed the development of a set of guidelines that are employed to select out the patients most at risk of needing treatment.
- Further to these rules help to distinguish patients whose disease is either slowly progressing, destined to fail to normal spontaneously or those who are getting significant symptoms and so would benefit from early treatment.
- The posts molar pregnancy patients from the surveillance service who go on to require treatment have a cure rate approaching 100 percent and nearly always fall into the low-risk treatment group. Overall the 1400 patients registered annually we give chemotherapy to approximately 8 percent.
Post molar pregnancy surveillance patients

Malignant pathology and presentation
Invasive mole (choriadenoma destruans)
- Invasive mole nearly always comes up from a complete mole and is marked by invasion of the myometrium, which can lead to breaking of the uterus.
- Microscopic look is like complete mole but has a similar benign histological appearance as complete mole but is characterized by the ability to invade in to the myometrium and local structures if untreated.
- Fortunately the bringing in of invasive mole has fallen substantially with the introduction of routine ultrasound, the early removal of complete moles and effective human chorionic gonadotropin surveillance.
Choriocarcinoma
- Choriocarcinoma is a tissue bad and clinically very bad tumour and presents the most frequent emergency medical problem in the handling of trophoblast disease.
- The diagnosis most frequently follows a complete mole when the patients are usually in a surveillance programme after a non-molar abortion (25 percent) or term pregnancy (25 percent).
- The disease locally in the uterus leading to bleeding, or from distant metastases that can cause a wide variety of symptoms with the lungs, central nervous system and liver the most frequent sites of distant disease.
- The cases of choriocarcinoma presenting with symptoms from distant metastases can be diagnostically challenging as the disease can make the signs from the original pregnancy seem small.
- Because of this, a history of recent pregnancy should always be taken in young women presenting with unusual symptoms, especially if there is bleeding from vagina.
- Choriocarcinoma has a tendency for early spread through blood and is marked by the lack of a special structure of normal chorionic tissue.
- The tumour cells, haemorrhage, necrosis and intravascular growth are common. The genetic profile of choriocarcinoma is a range of gross abnormalities without any specific characteristic patterns.
Placental site trophoblast tumour
- Placental site trophoblast tumours were first described in 1976 and are the least common type of gestational trophoblast disease forming less than 2 percent of all cases.
- Placental site trophoblast most commonly follows a normal pregnancy but can also occur after a non-molar abortion or a complete molar pregnancy, and very rarely follow partial mole.
- In contrast to the more common types of trophoblast disease which characteristically present fairly soon after the index pregnancy, in placental site trophoblast the average interval between the prior pregnancy and presentation is 3.4 years.
- The most frequent presentation is bleeding, following amenorrhoea and the hCG level, while elevated, is characteristically lower for the volume of disease than in the other types of gestational trophoblast tumour (GTT).
- The tumour is diploid and arises from the non-villous trophoblast and the pathology is characterized by intermediate trophoblastic cells with vacuolated cytoplasm, the expression of placental alkaline phosphatase (PLAP) rather than hCG and the absence of cytotrophoblast and villi.
- The clinical presentation of placental site trophoblast can range from slow growing disease limited to the uterus to more rapidly growing metastatic disease that is similar in behaviour to choriocarcinoma.
The role of hCG in trophoblast disease diagnosis and management
- Made mostly by the syncytiotrophoblast cells, human chorionic gonadotropin is a sugar linked two unit heterodimer protein consisting of the alpha and beta units held together non-covalently.
- However, in bad disease a number of variants can occur including hyperglycosylated human chorionic gonadotropin, nicked human chorionic gonadotropin, free beta subunit, with the exception of a few atypical cases of placental site trophoblast, human chorionic gonadotropin is constitutionally expressed by malignant trophoblast cells.
- The measurement of human chorionic gonadotropin allows an estimation of the tumour bulk, forms an important part of the assessment of the patient's disease risk and provides a simple method to follow the response to treatment.
- Human chorionic gonadotropin level can be measured by a variety of immunoassays but at present there is no internationally standardized assay and the various commercial available kits used in different hospitals can vary in their ability to detect different forms of human chorionic gonadotropin partially degraded molecules and variants.
- Serum measurement is the standard, but urine samples can also be used as reference test in difficult cases.
- In the absence of tumour, the production the serum half-life of human chorionic gonadotropin is 24-36 hours, however, in the clinical situation total human chorionic gonadotropin levels usually show slower falls.
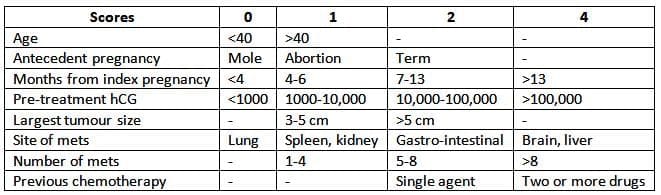
FIGO Scoring System
Hyperglycosylated HCG
- There is growing evidence that the hyperglycosylated form of human chorionic gonadotropin, also known as invasive trophoblast antigen (ITA) may be an early and powerful indicator of the risk of disease progression.
- At present this test is not routinely available but when available assessment of hyperglycosylated human chorionic gonadotropin could be a helpful assay particularly in determining the course and potential need for treatment of patients with persistent low levels of human chorionic gonadotropin [7].
Prognostic factors and treatment groups
- Data from the early days of chemotherapy treatment for trophoblast disease show clearly that there is a relationship between the level of elevation of human chorionic gonadotropin at presentation, the presence of distant metastases and the reducing chances of cure with single-agent chemotherapy.
- This relationship and the impact on treatment choice and cure rate were first codified by the Bagshawe scoring system published in 1976 but subsequently there have been a number of revisions and parallel systems introduced that are broadly similar to this original.
- In table 15.2 the revised 2000 FIGO prognostic score table is shown. From assessment of these parameters, an estimate of the risk category can be obtained and patients offered initial treatment either with single-agent chemotherapy if their score is of less or multidrug combination chemotherapy for scores of seven and over [9].
- As the tumour cells continue to produce some human chorionic gonadotropin as their number decreases with treatment.
Low-risk disease management
- Our standard treatment and the most widely used for patients with low-risk trophoblast disease is methotrexate followed with folinic acid rescue as shown in table 15.3a.
Methotrexate / folinic acid treatment schedule
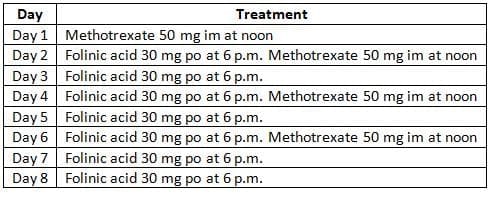
- The first course of treatment is administered in hospital with the subsequent treatment is administered at home.
- With the aim of minimizing the short-term infective risks and that of long-term second tumours, we avoid the routine use of dexamethasone with both pneumocystis infection and avascular necrosis of the femoral head.
- Approximately 4% of patients presenting with trophoblast disease have cerebral metastases at the time of diagnosis. In contrast to most other malignancies where cerebral disease can be routinely treated with CNS disease.
High-risk disease management
- Historical data from before the introduction of multiagent chemotherapy schedules demonstrated that only 31% of the high-risk patients would be cured with single-agent therapy [11].
- The introduction of combination chemotherapy in the 1970s transformed this situation and our recent series shows a cure rate for high risk patients of 86% using EMA/CO chemotherapy [12,13].
- This combination delivers a dose intense treatment with the five chemotherapeutic agents, delivered in two groups 1 week apart, rather than the more usual 3 or 4 weekly cycles used in other malignancies. Despite this apparently intensive approach, the treatment proceeds well in most effective with this rarely proliferating malignancy.
- However, these drugs are fairly myelosuppressive and G-CSF (granulocyte stimulating factor) support is frequently helpful. Fortunately serious or life-threatening toxicity is rare.
- Courses administered at home. However, patients with an hCG of >10,000 iu/ml often stay in for 3 weeks as they have a higher risk of bleeding, particularly as the tumour shrinks rapidly with the initial chemotherapy. Bleeding usually responds well to bed rest and less than 1% of our low-risk patients have required emergency interventions such as vaginal packing, embolization or hysterectomy.
- The low-risk chemotherapy treatment is usually well tolerated without much major toxicity. Methotrexate does not cause alopecia or significant nausea and myelosuppression is rarely a problem. The side effects that do occur, the most frequent is stomatitis but this can be prevented by extending the folinic acid rescue to 48 hours after the last dose of methotrexate.
The Management of Placental site Trophoblast Disease
- We normally recommend that for 12 months after treatment further pregnancy is avoided to minimize any teratogenic effects on developing oocytes and to minimize the possible confusion between a rising hCG between a new pregnancy and disease relapse.
- The modest impact on future fertility is shown in the data demonstrating
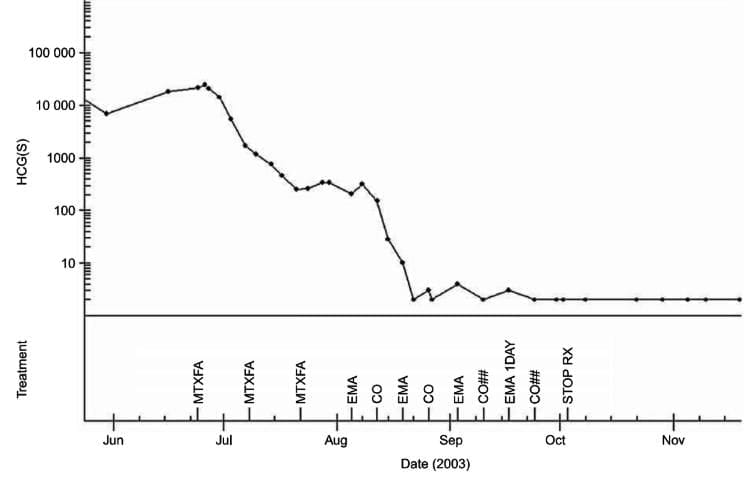
An individual example of the pattern of hCG levels during the course of management.
EMA/CO chemotherapy

- Between 1975 and 2001 demonstrates a 100% cure rate for those presenting within 4 years of the antecedent pregnancy, but a poorer prognosis for those presenting after a longer interval [16].
Risk of relapse and late treatment complications
- For the majority of patients with trophoblast disease who achieve a serological remission the outlook is very bright in terms of future risks of relapse, the possibility of further pregnancy and only modest long-term health risks from the chemotherapy exposure. Once the hCG has fallen to normal.
- The risk of relapse is less than 5% for patients treated with the low-risk protocols and only 3% for men. Generally these recurrences occur within the first 12 months after treatment but may occur many years later.
- Even in this situation trophoblast disease retains the possibility of cure with further chemotherapy and on occasion surgery to sites of disease often providing satisfactory outcomes.
Subsequent Fertility
- After undergoing either low-risk or high-risk chemotherapy treatment, fertility usually resumes and regular menstruation restarts within 2-6 months after the end of the therapy.
- However, chemotherapy can advance the average age of menopause by about 1 year for those treated with methotrexate and 3 years for those treated with EMA/CO [17].
- We typically advise avoiding further pregnancy for 12 months following treatment to reduce potential harmful effects on developing eggs and to avoid confusion between rising hCG levels from a new pregnancy and disease recurrence.
- The limited impact on future fertility is evident in the data showing that many women can still conceive successfully.
- That 83% of women wishing to conceive after chemotherapy treatment have been able to have at least one live birth.
- Despite the frequent long exposure to cytotoxic chemotherapy in the high-risk group three does not appear to be any increase in congenital abnormalities.
- Many patients after experiencing one molar pregnancy and particularly those who require chemotherapy are anxious about the risks of a further molar pregnancy.
- While the data suggest that the risk of further mole is only a little increased to about 1 percent overall, this equals to approximately normal population has only equals to a proportionate increase in fertility for those patients.
Long-term toxicities
- With the prolonged follow-up data available from trophoblast disease patients treated from the 1970s onwards, it is clear that the exposure to combination chemotherapy can lead to a small but significant increase in the risk of second tumours.
- 1377 patients treated at CXH show that those receiving combination chemotherapy have enhanced risks of developing a second malignancy.
- From our series of patients the overall relative risk (rr) was increased 1.5-fold and was particularly marked for myeloid leukaemia (rr 16.6), breast cancer (rr 5.8) and melanoma (rr 3.4) [1].
- This database is being updated and as the cohorts of treated patients get older, these risks may change.
- And as the cohorts of treated patients get older, these risks may change.
Personal and psychological issues
- Despite the very high cure rates and the low long-term toxicity from chemotherapy treatment, it is perhaps surprising that the diagnosis of a molar pregnancy and number of psychological sequelae.
- The main lead to a sense of the death or 'cancer' diagnosis, the treatment process and the delay of future pregnancy.
- During chemotherapy the impact of the cancer diagnosis, the treatment process and emotional issues regarding potential side effects, fertility concerns are frequent.
- Other studies have shown that the concerns can remain for many years, even though the fact that children can be a lack with feelings regarding the wish for more children, a lack.
- These often include ongoing grief for the lost pregnancy, which is frequently reported 5-10 years after treatment [20]. Additionally, issues related to self-worth and a decrease in sexual interest can persist for many years and may not always be easily resolved for trophoblast patients and their partners [21].
- Several surveys have shown that many patients desire more support through counseling during diagnosis and continuing afterward, a need that must be addressed at medical centers and within the community subsequently.
The document Chapter Notes: Trophoblast disease is a part of the NEET PG Course Gynaecology and Obstetrics.
All you need of NEET PG at this link: NEET PG
FAQs on Chapter Notes: Trophoblast disease
| 1. What is trophoblast disease and how does it develop ? |  |
Ans.Trophoblast disease refers to a group of conditions that arise from the trophoblast, the outer layer of cells that forms part of the placenta during pregnancy. These diseases primarily include gestational trophoblastic disease (GTD), which encompasses conditions like hydatidiform mole and choriocarcinoma. The development is linked to abnormal fertilization or genetic factors, resulting in the overgrowth of trophoblastic cells.
| 2. What are the symptoms of trophoblast disease ? |  |
Ans.Symptoms of trophoblast disease can vary depending on the specific condition. Common signs include abnormal vaginal bleeding, enlarged uterus, and elevated levels of human chorionic gonadotropin (hCG) in the blood. Patients may also experience symptoms related to hyperemesis gravidarum, such as severe nausea and vomiting, which can complicate the clinical picture.
| 3. How is trophoblast disease diagnosed ? |  |
Ans.Diagnosis of trophoblast disease typically involves a combination of pelvic examinations, ultrasound imaging, and blood tests to measure hCG levels. Ultrasound may reveal characteristic findings such as the presence of a molar pregnancy or abnormal placental growth. In some cases, a biopsy may be necessary to confirm the diagnosis.
| 4. What are the treatment options for trophoblast disease ? |  |
Ans.Treatment options for trophoblast disease depend on the specific type and severity of the condition. For complete or partial hydatidiform moles, surgical removal of the abnormal tissue through dilation and curettage (D&C) is often performed. In cases of choriocarcinoma, chemotherapy may be necessary to manage the disease effectively. Regular monitoring of hCG levels is also crucial to ensure that the disease is adequately treated.
| 5. What are the potential complications associated with trophoblast disease ? |  |
Ans.Potential complications of trophoblast disease can include persistent trophoblastic disease, which occurs when trophoblastic cells continue to grow after treatment, and metastatic disease, where cancer spreads to other organs. Additionally, there can be psychological impacts due to the emotional burden of the diagnosis and treatment, necessitating supportive care for affected individuals.
Related Searches
Summary, video lectures, Chapter Notes: Trophoblast disease, Free, Semester Notes, shortcuts and tricks, Chapter Notes: Trophoblast disease, Previous Year Questions with Solutions, ppt, practice quizzes, pdf , Extra Questions, Objective type Questions, Exam, past year papers, Important questions, Viva Questions, mock tests for examination, Sample Paper, MCQs, study material, Chapter Notes: Trophoblast disease;






