Chapter Notes: The menstrual cycle
Introduction
- Human females produce one oocyte per cycle (monotocous).
- Menstruation occurs monthly to ensure a single oocyte is ovulated and implantation can occur.
- Menstruation shows that the hypothalamus, pituitary, ovary, and uterus are functioning properly.
- Disruption in this system causes irregular periods, requiring investigation and treatment by gynaecologists.
- The menstrual cycle is mainly driven by ovarian events, though hypothalamus, pituitary, and uterus play roles.
- Ovarian follicle growth increases oestrogen, promoting endometrial growth.
- Luteinizing hormone (LH) surge triggers ovulation, followed by progesterone production from the corpus luteum, preparing the endometrium for implantation.
- Without pregnancy, declining progesterone leads to menstruation.
- The cycle is controlled by endocrine (hormonal) and paracrine (local tissue) mechanisms.
- Endocrine feedback loops involve pituitary gonadotropins and ovarian steroids.
- Paracrine processes regulate ovary and uterus locally.
Step One: Ensuring Monovulation
Folliculogenesis and the 'Follicular Phase'
- At birth, ovaries contain about 1,000,000 primordial follicles, reduced from 7,000,000 in fetal life.
- Follicle numbers decrease throughout life due to atresia (follicle death).
- Follicles escape the resting phase and resume meiosis, independent of external hormones.
- This process continues before puberty, during pregnancy, or with contraceptive use.
- Only about 400 follicles develop gonadotropin receptors and can ovulate.
- Most follicles undergo atresia before reaching the pre-antral stage.
- Early follicle growth is controlled by intra-ovarian paracrine factors like growth differentiation factor-9 (GDF-9), anti-Müllerian hormone (AMH), and Bax family proteins.
- Understanding these mechanisms may explain premature ovarian failure or early menopause.
- Pre-antral follicle growth depends on gonadotropins (FSH and LH).
- Early follicular phase FSH rise supports a group of pre-antral follicles to develop into antral follicles.
The development of a follicle, from
(a) The primordial follicle is surrounded by a single layer of undifferentiated epithelial cells and is insensitive to gonadotropins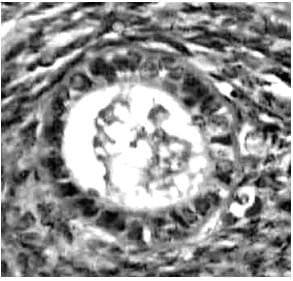 (b) The early antral follicle has well-differentiated theca (immunostained brown) and granulosa cell layers surrounding the developing antral cavity with the oocyte ( Small antral)
(b) The early antral follicle has well-differentiated theca (immunostained brown) and granulosa cell layers surrounding the developing antral cavity with the oocyte ( Small antral)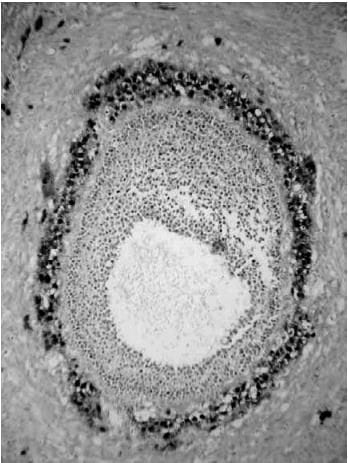 (c) The pre-ovular follicle with the oocyte surrounded by the cumulus oophorus with well-differentiated granulosa and theca cell layers (Preovulatory stages)
(c) The pre-ovular follicle with the oocyte surrounded by the cumulus oophorus with well-differentiated granulosa and theca cell layers (Preovulatory stages)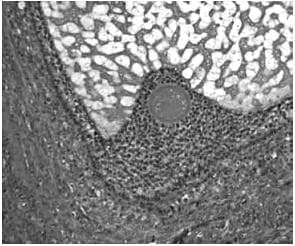
Specific Gene 'Knockouts' and Their Effects on Ovarian Function in the Mouse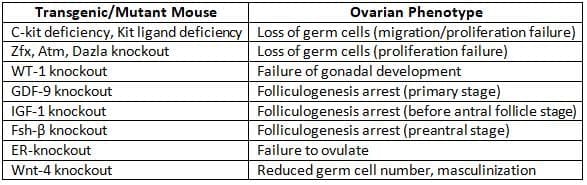
- One dominant follicle gains aromatase enzyme activity, converting androgens to oestradiol.
- The 'two-cell, two gonadotropin' model: LH stimulates theca cells to produce androgens; FSH drives granulosa cells to convert androgens to oestradiol.
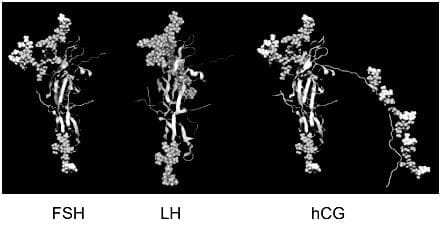
- FSH, LH, and human chorionic gonadotropin (hCG) share an identical alpha subunit but differ in beta subunits, which determines their specificity.
- Antibodies in assays target beta-subunit differences for FSH, LH, and hCG.
Molecular structure of FSH, LH and hCG.
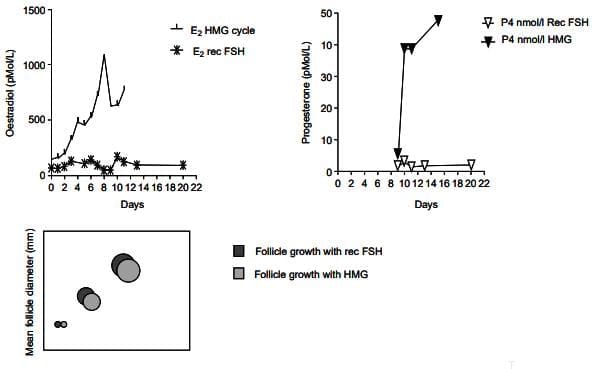
- In Kallmann's syndrome (no gonadotropin secretion), treatment with both FSH and LH (human menopausal gonadotropins, HMG) leads to normal follicle growth, oestradiol rise, and progesterone production post-hCG.
- FSH alone causes follicle growth but minimal oestradiol and no progesterone rise.
Effects of FSH alone and FSH and LH in combination on follicle development in a hypogonadotrophic patient with Kallmann's Syndrome.
Histological appearance of
(a) late secretory 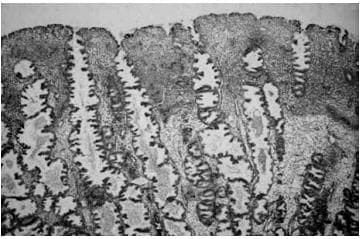
(b) menstrual endometrium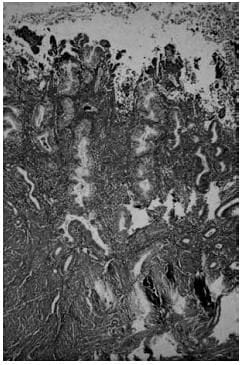
- Pituitary secretes LH and FSH in response to hypothalamic gonadotropin-releasing hormone (GnRH) pulses via the hypothalamo hypophyseal portal tract.
- LH secretion depends on GnRH pulse frequency (every 90 minutes in follicular phase, every 4 hours in luteal phase).
- FSH secretion is regulated by GnRH and other factors like inhibins and activins.
- Conditions like anorexia nervosa slow GnRH pulses, causing low LH/FSH and amenorrhoea.
- Mid-follicular phase oestradiol rise suppresses FSH via negative feedback, possibly with inhibin B from the dominant follicle.
The 'threshold concept' illustrating dependance of advanced follicle growth and maturation on a rise in circulating FSH concentration above an arbitrary threshold, with subsequent suppression of FSH preventing multiple follicle development.
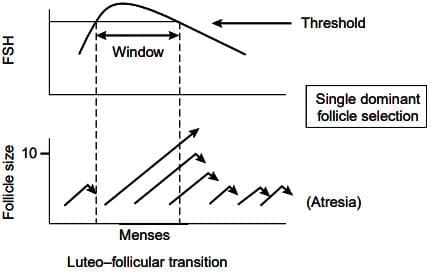
- FSH decline causes atresia of non-dominant follicles, ensuring single follicle ovulation (mono-ovulation).
- The 'threshold concept' explains mono-ovulation: FSH rise above a threshold allows one follicle to grow; FSH suppression prevents others from maturing.
- In IVF, high FSH doses aim for multiple follicles but risk poor yield in low follicle pools or overstimulation in conditions like polycystic ovary syndrome.
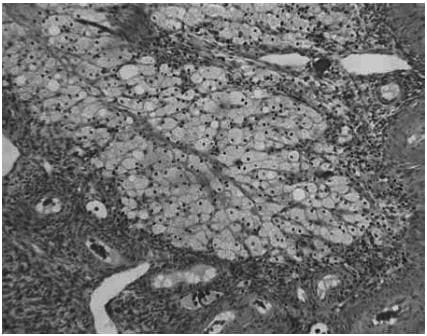
The histological appearances of the corpus luteum, showing
(a) an active corpus luteum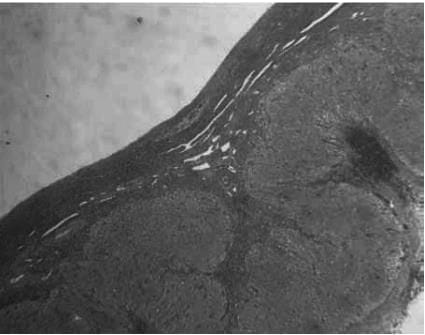 (b) regression of the corpus luteum with histiocyte infiltration
(b) regression of the corpus luteum with histiocyte infiltration
Step Two: Ensuring Maintenance of Very Early Pregnancy
The LH Surge and Ovulation
- The LH surge triggers final oocyte maturation, completing meiosis and extruding the first polar body.
- LH surge is caused by a rapid oestradiol rise in the late follicular phase, increasing GnRH pulse frequency and magnitude.
- A slight progesterone rise before ovulation occurs, but its role is unclear.
- Blocking progesterone with mifepristone prevents efficient ovulation, suggesting contraceptive potential.
- LH surge causes inflammation at the follicle apex, with new blood vessel formation, prostaglandins, and cytokines, leading to follicle rupture 38 hours later.
- Ovarian cytokines attract the Fallopian tube fimbria, forming a mucus strand to guide the oocyte into the tube.
- The ruptured follicle fills with blood, and theca/granulosa cells luteinize to form the corpus luteum.
- The corpus luteum produces high levels of progesterone (>25 nmol/L) and oestradiol.
- Progesterone levels increase further if pregnancy occurs.
Endometrial Development During the Menstrual Cycle and Early Pregnancy
- Follicular phase oestradiol rise acts on the endometrial basalis layer, which persists across cycles.
- Oestradiol stimulates rapid proliferative endometrium growth, synchronized with oocyte/follicle development.
- Endometrium develops organized glands and stromal compartments.
- After the LH surge, corpus luteum progesterone transforms the endometrium into a secretory phase for implantation.
- Endometrial angiogenesis (new blood vessel formation) occurs in three stages: vascular bed reformation during menstruation, vascular supply development in the proliferative phase, and spiral arteriole growth for implantation.
- Endometrial vessels grow and regress each cycle, unlike other stable vascular networks.
- Angiogenic factors like vascular endothelial growth factor (VEGF) and interleukins respond to ovarian steroids.
- Healthy secretory endometrium is critical for implantation.
- Fertilization occurs in the Fallopian tube's ampulla; the morula enters the uterus on day 3, becoming a blastocyst on day 4.
- The blastocyst sheds its zona pellucida and adheres to the endometrium, starting implantation.
- Endometrial proteins like integrins, glycosylated cell adhesion molecule 1 (GlyCAM-1), and osteopontin regulate implantation.
- Progesterone exposure in early pregnancy reduces epithelial progesterone receptors, decreases MUC1, and induces adhesion proteins.
- hCG from the trophoblast sustains the corpus luteum, maintaining progesterone for pregnancy.
- Mifepristone disrupts progesterone, used to terminate early pregnancy.
- IVF pregnancies require luteal phase support (hCG or progesterone) due to disrupted luteinization from GnRH agonists.
Menstruation
- Menstruation involves shedding superficial endometrial layers, followed by repair from the basalis layer.
- It is triggered by progesterone decline after corpus luteum regression without pregnancy.
- Corpus luteum progesterone depends on pituitary LH; during luteolysis, it becomes less responsive, reducing progesterone.
- In pregnancy, hCG from the trophoblast maintains corpus luteum function.
- Progesterone withdrawal activates intrauterine signals: chemotactic factors, matrix metalloproteinases (MMPs), prostaglandins (PGs), and other compounds.
- Leucocyte infiltration and inflammatory mediators make menstruation resemble an inflammatory event.
- High endometrial PGs (E and F series) are regulated by ovarian steroids.
- PGF2α causes myometrial contractions and vasoconstriction; PGE2 increases pain, oedema, and vasodilation, and induces IL-8 (a chemotactic mediator).
- Vasoconstriction leads to tissue hypoxia, amplifying inflammatory mediators.
- Spiral arteriole constriction and uterine muscle contraction expel shed tissue.
- PG synthesis inhibitors treat menorrhagia but may cause anovulation and infertility by affecting ovulation.
FAQs on Chapter Notes: The menstrual cycle
| 1. What are the main phases of the menstrual cycle and their durations? |  |
| 2. What hormonal changes occur during the menstrual cycle? |  |
| 3. How does stress affect the menstrual cycle? |  |
| 4. What are common menstrual cycle disorders? |  |
| 5. What factors can influence the regularity of the menstrual cycle? |  |





