NEET PG Exam > NEET PG Notes > Gynaecology and Obstetrics > Chapter Notes: Heart disease in pregnancy
Chapter Notes: Heart disease in pregnancy
Introduction
- Heart disease is now the leading cause of maternal death in the UK, despite being rare in pregnancies in the UK, Europe, and the developed world.
- The maternal mortality rate from cardiac disease has been increasing since the early 1980s.
- Between 2000 and 2002, there were 44 indirect deaths attributed to cardiac disease, resulting in a rate of 2.2 per 100,000 maternities.
- The main causes of cardiac deaths in the last decade include:
- Cardiomyopathy, mostly occurring during the peri-partum period
- Myocardial infarction, primarily due to coronary artery dissection
- Dissection of the thoracic aorta
- Pulmonary hypertension
- Rheumatic heart disease is now very uncommon among women of childbearing age in the UK and is mostly found in immigrants.
- There have been no reported maternal deaths from rheumatic heart disease since 1994.
- Women with congenital heart disease who underwent corrective or palliative surgery in childhood are now reaching adulthood and experiencing pregnancies, which can be complex.
- Women with metal prosthetic valves face difficult decisions regarding anticoagulation during pregnancy.
- Pregnancy brings significant physiological changes, making symptoms like palpitations and signs such as an ejection systolic murmur common and usually harmless. However, not all women with serious heart conditions can handle these increased physiological demands.
- Therefore, the care of pregnant and postpartum women with heart disease requires a multidisciplinary approach involving obstetricians, cardiologists, and anaesthetists, ideally in a dedicated antenatal cardiac clinic.
- This setup allows for a documented management plan for both planned and emergency deliveries.
- The following sections discuss the most common and important cardiac conditions encountered during pregnancy.
Physiological Changes During Pregnancy, Labor, and Delivery
- Blood Volume: Begins to increase by the 5th week after conception due to oestrogen and prostaglandin, which relax smooth muscle and expand the capacity of the veins.
- Plasma Volume: Rises significantly, while red cell mass increases but not as much, leading to physiological anaemia during pregnancy.
- Systemic Vascular Resistance: Decreases due to the relaxation of smooth muscle in arteries, contributing to an early rise in cardiac output.
- Blood Pressure: May slightly decrease initially but usually returns to pre-pregnancy levels by term.
- Cardiac Output: Increases mainly due to a rise in stroke volume, with a smaller increase in resting heart rate (10-20 beats/min).
- Second Trimester: By the end, blood volume and stroke volume have increased by 30-50%, especially in multiple pregnancies.
Cardiovascular Changes
- Pulmonary Capillary Wedge Pressure (PCWP): Does not increase during pregnancy.
- Serum Colloid Osmotic Pressure: Decreases, making pregnant women more susceptible to pulmonary oedema.
- Gradient Drop: The gradient between colloid oncotic pressure and PCWP drops by 28%, increasing the risk of pulmonary oedema.
- Triggers of Pulmonary Oedema: Can be caused by increased cardiac preload (e.g., fluid infusion) or increased pulmonary capillary permeability (e.g., in pre-eclampsia).
Positioning and Cardiac Output
- Late Pregnancy: Lying on the back compresses the inferior vena cava (IVC), reducing venous return, stroke volume, and cardiac output.
- Position Changes: Moving from a side position to supine can cause a 25% drop in cardiac output.
- Recommended Positions: Pregnant women should ideally be positioned on their left or right side to optimise cardiac output and blood flow to the uterus and placenta.
- Supine Positioning: If lying on the back, the pelvis should be rotated to enhance cardiac output and blood flow to the uterus and placenta.
- Fetal Blood Flow:. decrease in cardiac output can compromise blood flow to the uterus and placenta, affecting the fetus.
Changes During Labour
- Cardiac Output: Increases by 15% in the first stage and 50% in the second stage of labour.
- Uterine Contractions: Cause an auto transfusion of 300-500 ml of blood back into circulation, contributing to increased cardiac output.
- Pain and Anxiety: Raise heart rate and blood pressure during labour.
- Cardiac Output During Contractions: Rises more during contractions but also increases between contractions.
- Pain Relief: Can reduce the increase in stroke volume with contractions, particularly with epidural analgesia.
- Epidural Anaesthesia: May cause arterial vasodilation and a drop in blood pressure during labour.
- General Anaesthesia: Typically raises blood pressure and heart rate during induction but stabilises afterwards.
- Prostaglandins: Used to induce labour with minimal effects on haemodynamics.
- Ergometrine: Can cause vasoconstriction during labour.
- Syntocinon: May lead to vasodilation and fluid retention during labour.
Postpartum Changes
- Third Stage of Labour: Up to a litre of blood may return to circulation as pressure on the IVC is relieved and the uterus contracts, leading to increased intrathoracic and cardiac blood volume.
- Cardiac Output: Rises by 60-80% immediately postpartum, then quickly drops back to pre-labour levels within about 1 hour after delivery.
- Fluid Transfer: From the extravascular space further increases venous return and stroke volume postpartum.
- Risk of Pulmonary Oedema: Women with cardiovascular issues are at higher risk of pulmonary oedema during the second stage of labour and immediately after delivery.
- Recovery of Changes: Physiological changes return rapidly in the first week postpartum and more slowly over the following 6 weeks, with significant changes persisting even a year later, especially after subsequent pregnancies.
Normal Cardiovascular Findings During Pregnancy
- First Heart Sound and Second Heart Sound: During pregnancy, it is common to hear a loud first heart sound (S1) along with a pronounced split in the second heart sound (S2). This means that the sounds of the heart are more noticeable and have distinct characteristics.
- Third Heart Sound:. physiological third heart sound (S3) may be detected at the apex of the heart. This sound is typically a normal finding during pregnancy and indicates increased blood flow.
- Systolic Ejection Murmur:. systolic ejection murmur is often heard at the left sternal edge during pregnancy. This murmur can be quite loud and may be audible across the entire precordium (the area of the chest over the heart). It is important to note that this murmur changes with posture and, in the absence of other abnormalities, indicates increased stroke volume (the amount of blood the heart pumps with each beat).
- Venous Hums and Mammary Soufflés: Venous hums and mammary soufflés may also be present during pregnancy. These sounds are related to increased blood flow and are generally harmless.
- Pulse: Due to peripheral vasodilation (widening of blood vessels), the pulse may feel bounding during pregnancy. This means that the pulse is stronger and more forceful.
- Ectopic Beats: Ectopic beats, which are irregular heartbeats, are fairly common during pregnancy. These beats are usually benign and not a cause for concern.
Cardiac Evaluations During Pregnancy
- In late pregnancy, the electrocardiographic (ECG) axis shifts upwards because the heart's position becomes more horizontal. It's common to observe small Q-waves and T-wave inversion in the right precordial leads during clinical assessments.
- Atrial and ventricular ectopic beats are also frequently seen.
- Safety of Radiation and Imaging During Pregnancy
- Chest X-rays (CXR) during pregnancy are generally considered safe for fetal exposure to radiation.
- It is crucial to refer to the latest medical guidelines or studies for the most current safety evaluations regarding imaging during pregnancy.
- CXRs should not be avoided if they are clinically necessary during pregnancy.
- Transthoracic echocardiograms are the preferred method for checking or monitoring structural heart disease in pregnant women.
- Transoesophageal echocardiograms (TOE) are also safe during pregnancy when precautions to prevent aspiration are taken.
- Magnetic resonance imaging (MRI) and chest computed tomography (CT) are considered safe during pregnancy.
- Routine tests such as electrophysiological studies and angiography are usually postponed until after pregnancy.
- However, angiography should not be delayed in urgent situations like acute coronary syndromes.
General considerations in pregnant women with heart disease
- Factors Influencing Pregnancy Outcome and Safety:
- Pulmonary Hypertension: The presence and severity of pulmonary hypertension significantly impact the outcome of pregnancy in women with heart disease.
- Cyanosis: The presence of cyanosis, which indicates low oxygen levels in the blood, is a critical factor.
- Hemodynamic Significance: The hemodynamic significance of the heart condition plays a crucial role in determining the safety of pregnancy.
- Functional Class: The functional class, as per the New York Heart Association (NYHA) classification, based on activity level causing dyspnoea, is an important consideration.
- Pregnancy Management: Most women with existing heart disease can manage pregnancy effectively if they are asymptomatic or only mildly symptomatic (NYHA class II or less) before pregnancy.
- High-Risk Conditions:
- Pulmonary Hypertension: Women with pulmonary hypertension face significant risks during pregnancy.
- Marfan's Syndrome: Women with Marfan's syndrome and a dilated aortic root are at high risk during pregnancy.
- Mitral or Aortic Stenosis: Some women with mitral or aortic stenosis may face increased risks during pregnancy.
- Predicting Cardiac Complications: Cardiac complications during pregnancy in women with structural heart disease can be predicted by several factors:
- Prior Cardiac Event or Arrhythmia:. history of a cardiac event or arrhythmia increases the risk of complications.
- NYHA Classification:. NYHA classification greater than II indicates a higher risk.
- Left Ventricular Ejection Fraction: The left ventricular ejection fraction is a critical indicator of heart function.
- Left Heart Obstruction:
- Mitral valve area < 2 cm²
- Aortic valve area < 1.5 cm²
- Aortic valve gradient > 30 mmHg
- Referral to Specialist Centres: Women with the aforementioned risk factors should be referred to specialist centres for counselling and management during pregnancy.
- Risks for Women with Cyanosis: Women with cyanosis (oxygen saturation < 80-85%) face higher risks of
- Intrauterine Growth Restriction: The fetus may experience growth restriction.
- Fetal Loss: There is an increased risk of fetal loss.
- Thromboembolism: Thromboembolism due to polycythaemia is a potential risk.
- Multidisciplinary Team Management: Women with risk factors for adverse cardiac or obstetric events should be managed by a multidisciplinary team, including:
- Cardiologists: Specialists in cardiology with expertise in pregnancy.
- Obstetricians: Obstetricians experienced in managing cardiac disease during pregnancy.
- Fetal Medicine Specialists: Experts in fetal medicine.
- Paediatricians: Paediatricians involved in the care of the newborn.
- Early Involvement of Obstetric Anaesthetists: Early involvement of obstetric anaesthetists is crucial, and a carefully documented delivery plan is essential.
Specific Cardiac Conditions
Congenital Heart Disease
- Women with mild congenital heart defects that do not cause any symptoms usually have a good experience during pregnancy.
- Many heart defects may have been surgically repaired or treated by a pediatric cardiologist earlier in life.
- Some defects might only be diagnosed during pregnancy.
- Women with congenital heart disease have a higher risk of having a child with the same condition.
- It is recommended to offer genetic counselling before conception, if possible.
- Detailed scans for fetal cardiac anomalies should be performed using fetal echocardiography by 18-20 weeks of gestation.
Acyanotic Congenital Heart Disease
Atrial Septal Defect
- Atrial septal defect (ASD), particularly the secundum type, is the most prevalent congenital heart defect found in adults.
- Paradoxical embolism is a rare occurrence, and arrhythmias associated with ASD typically manifest in middle age.
- Up to 15% of untreated ASDs may lead to mitral regurgitation caused by mitral leaflet prolapse.
- The development of pulmonary hypertension in these cases is uncommon.
- During pregnancy, complications are generally not expected. However, acute blood loss is poorly tolerated and can result in:
- A substantial increase in left-to-right shunting.
- A rapid decline in left ventricular output, blood pressure, coronary blood flow, and, in severe cases, cardiac arrest.
Ventricular Septal Defect and Patent Arterial Duct
- Ventricular septal defects (VSD) and patent arterial ducts (PAD) that elevate the workload on the left ventricle are typically manageable during pregnancy.
- However, larger defects complicated by pulmonary vascular disease can pose challenges.
Pulmonary Stenosis
- Symptoms of pulmonary stenosis are usually absent during pregnancy.
- In cases of severe pulmonary stenosis leading to right ventricular failure, balloon pulmonary valvotomy can be successfully performed during pregnancy, preferably in the second trimester to maximize uterine protection.
Aortic Stenosis
- Aortic stenosis can lead to complications during pregnancy due to left ventricular outflow obstruction.
- Pre-pregnancy assessment is recommended to evaluate the severity of the obstruction.
- Significant obstruction is indicated by an aortic valve area of less than 1 cm² or a mean gradient across the valve exceeding 50 mmHg.
- High-risk indicators for complications during pregnancy include:
- Inability to achieve normal blood pressure increases without ST- or T-wave changes during exercise.
- Impaired left ventricular function.
- Development of symptoms such as angina or dyspnea.
- Electrocardiograms (ECG) typically show left ventricular hypertrophy in these patients.
- Doppler assessment of transaortic valve velocity should increase during pregnancy if stroke volume rises appropriately.
- In cases of impaired left ventricular function, the left ventricle may not generate a high gradient across the aortic valve, potentially leading to false reassurance from a low gradient.
- If patients experience symptoms such as angina, dyspnea, or resting tachycardia, they should be admitted for rest and treatment.
- Administering a β-adrenergic blocker can improve diastolic coronary flow time, enhance left ventricular filling, alleviate angina, and improve overall cardiac function.
- If symptoms persist despite treatment, balloon aortic valvotomy may be considered. While these valves are not ideal, successful procedures can provide temporary relief until the completion of pregnancy.
- Balloon aortic valvotomy can also relieve discrete subaortic stenosis but carries the risk of inducing mitral regurgitation.
Coarctation of the Aorta
- Most cases of coarctation of the aorta will have been surgically corrected, but residual narrowing is a common occurrence.
- Aortic coarctation may be diagnosed during pregnancy, particularly in cases of elevated blood pressure at booking, and should be ruled out.
- While blood pressure can be lowered, maintaining control during physical exertion is challenging, increasing the risk of serious complications such as cerebral hemorrhage and aortic dissection.
- Patients with pre-existing aortic abnormalities, such as coarctation, Marfan syndrome, or other inherited connective tissue disorders, are at higher risk for aortic dissection.
- Hypertension in these patients should be treated aggressively, with beta-blockers being the preferred medication to reduce the risk of rupture and dissection.
- Left ventricular failure is unlikely to occur unless there is an associated stenotic bicuspid aortic valve or endocardial fibroelastosis with impaired left ventricular function.
- Normal delivery is generally achievable for patients with coarctation of the aorta, although severe cases may require a shortened second stage of labor.
Marfan Syndrome
- Approximately 80% of individuals with Marfan syndrome experience cardiac involvement, primarily involving mitral valve prolapse and regurgitation.
- Pregnancy increases the risk of aortic rupture or dissection, particularly during the third trimester or shortly after delivery.
- Aortic root dilation, especially with a root dimension exceeding 4 cm, is associated with a 10% increased risk of complications.
- Women with aortic roots measuring over 4.6 cm should consider delaying pregnancy until after surgical repair or replacement of the aortic root.
- In contrast, women with minimal cardiac involvement and an aortic root under 4 cm can generally expect favorable pregnancy outcomes.
- However, those with a family history of aortic dissection or sudden death may face increased risk, as dissection can occur without prior warning in some families.
- Management:
- Counseling about the dominant inheritance of Marfan syndrome.
- Regular monitoring of the aortic root through monthly echocardiograms for individuals with heart involvement.
- Prescribing beta-blockers for patients with high blood pressure or a dilated aortic root.
- Delivery Options:
- Vaginal delivery is feasible for patients with stable aortic root measurements.
- An elective Caesarean section with regional anaesthesia is advised for patients with an enlarged or dilating aortic root.
Cyanotic Congenital Heart Disease
- Adults with cyanotic congenital heart disease often face complications such as pulmonary hypertension or pulmonary stenosis.
- Despite the severity of their conditions, patients with single ventricle defects, transposition of the great arteries, and complex pulmonary atresias can survive into adulthood, even if they have undergone previous palliative surgeries.
Tetralogy of Fallot
- Condition Overview: Tetralogy of Fallot is characterized by a combination of severe right ventricular outflow tract obstruction, a large subaortic ventricular septal defect, and an overriding aorta. This leads to right ventricular hypertrophy and right-to-left shunting, causing cyanosis.
- Pregnancy Considerations: During pregnancy, women with this condition may experience poor fetal growth, resulting in a higher risk of miscarriage, premature birth, and small-for-dates infants. The hematocrit level often increases during pregnancy due to systemic vasodilation, which can exacerbate right-to-left shunting.
- Successful Outcomes: Women with a resting arterial saturation of 85% or higher, a hemoglobin level below 18 g, and a hematocrit level below 55% are more likely to have positive pregnancy outcomes. It is important for these women to rest, as exertion can significantly decrease arterial saturation and hinder fetal growth.
- Preventive Measures: Subcutaneous low molecular weight heparin (LMWH) should be administered during pregnancy to prevent blood clots and paradoxical embolism.
- Post-Surgical Outcomes: Women who have undergone surgical correction of tetralogy of Fallot generally experience good pregnancy outcomes.
Post-Operative Congenital Heart Disease
- Neonatal Palliative Surgery: Survivors of neonatal palliative surgery for complex congenital heart disease require individual evaluation to assess their condition and the feasibility of pregnancy.
- Echocardiography:. thorough assessment through echocardiography by a paediatric or adult congenital cardiologist is crucial for evaluating the heart's condition in post-operative patients.
- Fontan Operation: After the Fontan operation, the right ventricle is bypassed, and the left ventricle is responsible for pumping blood to both the systemic and pulmonary circulations. While pregnancy can still be successful after this procedure, increases in venous pressure may lead to complications such as hepatic congestion and severe swelling.
Eisenmenger Syndrome and Pulmonary Hypertension
- Pulmonary vascular disease, whether due to a reversed large left-to-right shunt like a ventriculo-septal defect (VSD) or caused by lung/connective tissue diseases (such as scleroderma) or primary pulmonary hypertension, presents significant risks during pregnancy.
- Counselling and Risks: Women with known serious pulmonary vascular disease should receive early counselling to avoid pregnancy and appropriate contraceptive advice, as maternal mortality rates can be as high as 40%. The risk arises from fixed pulmonary vascular resistance that does not decrease during pregnancy, leading to an inability to increase pulmonary blood flow and causing severe hypoxaemia.
- Definition of Pulmonary Hypertension: Pulmonary hypertension is defined as a resting mean pulmonary artery pressure of 25 mmHg or more, or 30 mmHg during exercise, without a left-to-right shunt.
- Screening and Diagnosis: Pulmonary artery systolic pressure is typically estimated using Doppler ultrasound to measure the velocity of the regurgitant jet across the tricuspid valve; this serves as a screening test. If Doppler results suggest pulmonary hypertension, a specialist cardiac opinion is recommended. Diagnosing pulmonary vascular disease can be challenging, especially in cases with a left-to-right shunt, and may require further tests, including cardiac catheterisation to determine pulmonary vascular resistance.
- Management During Pregnancy: For women with pulmonary hypertension who still have dominant left-to-right shunts, the risk is lower, and they may manage better during pregnancy. However, they must be closely monitored for potential development of fixed pulmonary vascular resistance.
- Unplanned Pregnancies: In cases of unplanned pregnancies, therapeutic termination should be offered. Elective termination carries a 7% risk of mortality, highlighting the importance of avoiding pregnancy if possible.
- Multidisciplinary Care: If pregnancy occurs against medical advice, multidisciplinary care is essential, including elective admission for bed rest, administration of 60% oxygen, and thromboprophylaxis with low molecular weight heparin (LMWH). Careful monitoring of fetal growth is also crucial.
Acquired Valve Disease
Mitral Valve Prolapse
- A floppy mitral valve can occur randomly or be passed down through families as a dominant trait, particularly in families with Marfan syndrome.
- Pregnancy is usually manageable for individuals with mitral valve prolapse, and antibiotics are only necessary if there is accompanying mitral regurgitation.
Rheumatic Heart Disease
Mitral Stenosis
- Mitral stenosis is the most prevalent serious heart condition observed during pregnancy on a global scale.
- Patients who are asymptomatic may experience a deterioration of their condition during pregnancy, and the severity of mitral stenosis can escalate compared to a previous uncomplicated pregnancy.
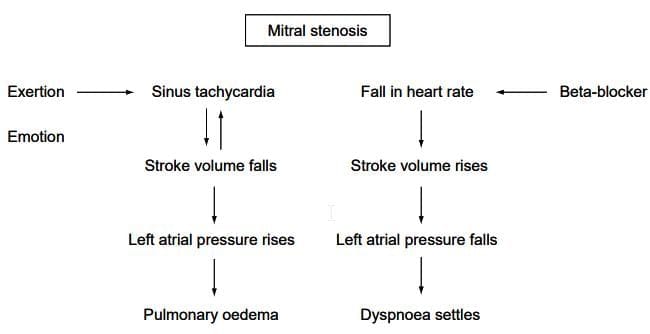
Mitral stenosis
- Stenosis has the potential to recur or worsen following procedures such as valvuloplasty or valvotomy.
- In some instances, mitral stenosis may be overlooked during routine examinations due to the absence of typical diastolic murmurs.
- Women may face deterioration of their condition due to factors such as:
- Increased heart rate resulting from pain, anxiety, physical activity, or infections.
- The occurrence of arrhythmias.
- The rise in cardiac output during pregnancy.
- The presence of sinus tachycardia at rest should raise concerns, as it indicates an inability to increase stroke volume, leading to elevated left atrial pressure.
- This situation creates a cycle of increased heart rate and left atrial pressure, which can potentially result in pulmonary edema.
- Anxiety caused by dyspnea can further exacerbate the condition by increasing heart rate.
- Pulmonary edema may also occur due to increased volume during labor or inappropriate administration of intravenous fluids.
- The risks associated with mitral stenosis are heightened in cases of severe mitral stenosis (valve area < 1 cm²),moderate to severe symptoms prior to pregnancy, and late presentations.
- An electrocardiogram (ECG) may reveal left atrial P waves and right axis deviation, while a chest X-ray (CXR) may show a small heart with an enlarged left atrial appendage and signs of pulmonary congestion.
- Transthoracic echocardiography is a diagnostic tool that can confirm the presence of mitral stenosis.
- Women diagnosed with severe mitral stenosis are advised to postpone pregnancy until after undergoing appropriate valve surgery.
- Beta-blockers may be beneficial in managing the condition by reducing heart rate and the risk of pulmonary edema, with a target heart rate of under 90 beats per minute.
- Diuretics should be initiated or continued if deemed necessary.
- In cases of pulmonary edema, patients should be positioned upright, receive oxygen therapy, and have anxiety alleviated with diamorphine; intravenous frusemide (20 mg) should be administered.
- The use of digoxin is only recommended in the presence of atrial fibrillation.
- For severe cases of mitral stenosis, balloon mitral valvotomy can be safely performed during pregnancy if the valve is suitable, although this procedure requires access to a cardiac facility.
- Balloon valvotomy carries an approximate 1% risk of major complications; closed valvotomy is associated with a fetal mortality rate of 5-15% and maternal mortality of 3%, while open valvotomy has higher mortality rates.
- If open surgery is deemed necessary, it is advisable to postpone the procedure until after delivery.
- During labor and delivery, women with mitral stenosis should avoid lying flat, and measures should be taken to prevent fluid overload, even in cases of low urine output.
- Intravenous colloids should be limited, and moderate epidural analgesia is considered appropriate.
- Vaginal delivery is preferred, although instrumental assistance may be required in some cases.
Regurgitant Valve Disease
- Patients with regurgitant valve disease, whether it is mitral or aortic, generally handle pregnancy better than those with valvular stenosis.
- During pregnancy, the body experiences systemic vasodilatation, which helps to decrease the regurgitant flow.
- In patients with aortic regurgitation, an increase in heart rate, known as tachycardia, also plays a role in reducing this regurgitant flow.
- If the valve disease is due to rheumatic fever, a sudden episode of atrial fibrillation can lead to serious complications, such as pulmonary edema.
Mechanical Heart Valves
- Most women with prosthetic heart valves can manage pregnancy safely due to their good cardiovascular health.
- However, there is debate about the best way to manage anticoagulation during pregnancy for these women.
- They need lifelong anticoagulation to prevent thrombosis, which must be maintained throughout pregnancy.
- Warfarin is generally safe for the mother if the international normalized ratio (INR) is monitored closely.
- However, it can cause warfarin embryopathy (chondrodysplasia punctata) if used during organ development (6 to 12 weeks of pregnancy) and may lead to fetal intracerebral hemorrhage at any time.
- Overdose risks exist because the fetus is more affected by the anticoagulant than the mother.
- The immature fetal liver produces low levels of vitamin K-dependent clotting factors, and maternal procoagulants do not cross the placenta.
- Warfarin also increases the risk of miscarriage and stillbirth.
- The risks to the fetus from warfarin depend on the dosage, with most women needing less than 5 mg per day.
- Higher doses pose greater risks of teratogenesis, miscarriage, and stillbirth.
- Heparin and low molecular weight heparin are preferable options as they do not cross the placenta.
- However, they carry a higher risk of valve thrombosis and embolic events, even at full anticoagulant doses.
- Unfractionated heparin has additional drawbacks, including the need for parenteral administration, short duration of action, narrow therapeutic index, increased dose requirement during pregnancy, and higher bleeding risk with increasing dosage.
- Long-term high doses may lead to osteoporosis. Low molecular weight heparins (LMWHs) have a better safety profile in pregnancy, but manufacturers advise against using them as anticoagulants for patients with prosthetic heart valves.
- Anticoagulation Management Options
- There are three main approaches to managing anticoagulation during pregnancy:
- Continue warfarin throughout pregnancy, stopping only for delivery.
- This is the safest option for the mother.
- Switch from warfarin to high-dose unfractionated or low molecular weight heparin from 6 to 12 weeks of pregnancy to prevent warfarin embryopathy.
- Use high-dose unfractionated or low molecular weight heparin for the entire pregnancy.
- The choice among these options depends on several factors:
- Type of mechanical valve: Newer bi-leaflet valves (e.g., Carbomedics) have a lower risk of thrombosis compared to older ball and cage (e.g., Starr-Edwards) or single tilting disc (e.g., Bjork Shiley) valves.
- Position of valve replacement: Aortic valves have a lower risk of thrombosis than mitral valves.
- Number of mechanical valves: Having two valves increases the risk of thrombosis.
- Amount of warfarin needed: Higher warfarin doses increase risks.
- History of embolic events: Past history influences risk assessment.
- If warfarin is prescribed during pregnancy, regular fetal scans are important to identify severe embryopathy, such as stippled epiphyses and intracerebral hemorrhage.
- Warfarin should be stopped and replaced with LMWH for 10 days before delivery to clear it from fetal circulation.
- LMWH therapy should be paused during delivery.
- Transitioning from LMWH back to warfarin should be delayed for at least 3 to 5 days after childbirth to reduce the risk of obstetric hemorrhage.
- In cases of bleeding or urgent delivery in fully anticoagulated patients, warfarin can be reversed using fresh frozen plasma (FFP) and vitamin K, while heparin can be reversed with protamine sulfate.
- Vitamin K should be avoided if possible, as it complicates post-delivery anticoagulation with warfarin.
- Thrombolytic treatment for prosthetic valve thrombosis during pregnancy may be considered, as its risks are lower than those associated with cardiothoracic surgery.
- Women with metal valve replacements require antibiotic endocarditis prophylaxis for delivery, regardless of the delivery method.
Coronary Artery Disease
- Myocardial infarctions during pregnancy often occur without the preceding symptom of angina, and are typically caused by spontaneous coronary artery dissection or coronary artery thrombosis.
- These events are characterized by sudden and severe chest pain, primarily affecting the heart's anterior wall, and are more common in late pregnancy or around the time of delivery. The maternal mortality rate in such cases is approximately 20%.
- Myocardial infarctions can also occur after the administration of oxytocic agents, and coronary thrombosis may be linked to drug abuse, particularly crack cocaine. It is important to consider embolic occlusion and identify potential sources such as mitral stenosis or infective endocarditis.
- Risk Factors
- The risk factors for ischaemic heart disease during pregnancy are similar to those in non-pregnant individuals.
- Multigravid women and those who smoke have an increased risk.
- Other contributing factors include diabetes, obesity, hypertension, and hypercholesterolaemia.
- Management
- The management of acute myocardial infarction in pregnant women adheres to similar protocols as those for non-pregnant women, with specific considerations for the unique circumstances of pregnancy.
- Coronary angiography should be conducted promptly to evaluate the condition and determine the appropriate treatment.
- Interventions such as intravenous and intracoronary thrombolysis, percutaneous transluminal coronary angioplasty, and stenting have been successfully performed during pregnancy.
- Aspirin and beta-blockers are generally considered safe for use during pregnancy.
- There is limited information regarding the use of clopidogrel and glycoprotein IIb/IIIa inhibitors during pregnancy, although some case reports indicate successful usage.
- Statins should be discontinued during pregnancy due to their association with an increased risk of fetal malformations.
Hypertrophic Cardiomyopathy
- Hypertrophic Cardiomyopathy (HCM) is a genetic condition passed down in an autosomal dominant manner. It is characterized by the thickening of the left ventricle of the heart without any abnormal increase in pressure.
- This condition involves alterations in the muscle cells of the heart and their arrangement.
- Studies within families, often supported by genetic testing, reveal a broad spectrum of symptoms that can differ not only among individuals but also within the same family.
- Previous research primarily focused on high-risk patients referred due to severe symptoms or a concerning family history.
- Before the advent of echocardiography, only severe cases of HCM could be diagnosed, which laid the groundwork for many early studies on the condition.
- HCM is often first identified during pregnancy, typically when a systolic murmur leads to further investigation through an ECG and echocardiogram.
- Most individuals with HCM are asymptomatic and manage their condition well.
- Recent studies suggest that HCM affects approximately 1 in 500 young adults and is generally a mild condition.
- Symptoms and Management
- During pregnancy, patients with HCM may experience positive changes such as an increase in the left ventricular cavity size and stroke volume.
- The primary concern is the potential for obstruction in the left ventricular outflow tract, which can be triggered by low blood pressure or low blood volume.
- Symptoms such as shortness of breath, chest pain, dizziness, or fainting may indicate the need for a β-blocking drug to manage the condition.
- Ventricular arrhythmias are uncommon in young patients with HCM but may be more prevalent in older individuals.
- Sudden cardiac death during pregnancy is extremely rare in patients with HCM.
- It is essential for all patients with HCM to avoid blood vessel dilation during labor and delivery, as well as during regional anesthesia or analgesia.
- Any instances of low blood volume must be promptly and effectively corrected to ensure the safety of both the mother and the baby.
- It is uncommon to observe hypertrophy (thickening of the heart muscle) in infants born to mothers with HCM.
Peripartum Cardiomyopathy
- Peripartum cardiomyopathy is a condition where heart failure occurs during the last month of pregnancy or within 5 months after giving birth. This happens in individuals who did not have any known heart disease or risk factors before the last month of pregnancy.
- The diagnosis is based on specific echocardiographic findings, including:
- Left ventricular ejection fraction <45%>
- Fractional shortening <30%
- Left ventricular end diastolic pressure (LVEDP) >2.7 cm/m²
- Echocardiographic Findings: Echocardiograms in peripartum cardiomyopathy typically reveal enlargement of all four heart chambers, with a particular emphasis on the left ventricle.
- The left ventricle may show reduced movement (hypokinesia), which can be generalized or localized to a specific area.
- Incidence and Risk Factors: Peripartum cardiomyopathy is a rare condition, and its true incidence is not well-defined because mild cases may go undetected.
- Recognized risk factors include:
- Multiple pregnancies
- Hypertension (whether pre-existing, related to pregnancy, or associated with conditions like pre-eclampsia)
- Multiparity (having many children)
- Older maternal age
- Afro-Caribbean ethnicity
- Clinical Presentation: Clinically, peripartum cardiomyopathy resembles dilated cardiomyopathy, with the key difference being its timing in relation to pregnancy. The severity of the condition can vary from mild to severe, with mild cases potentially being detected only through echocardiography.
- Symptoms to monitor for include:
- Breathlessness
- Rapid heartbeat (tachycardia)
- Signs of heart failure
- Fluid Overload and Triggers: Fluid overload is often a significant factor in peripartum cardiomyopathy. This overload can be triggered by medications such as syntocinon or by fluids administered during spinal anaesthesia for delivery.
Chest X-Ray Findings: Chest X-ray findings in peripartum cardiomyopathy can vary widely among patients and may include:
- An enlarged heart
- Pulmonary congestion or fluid accumulation in the lungs
- Bilateral pleural effusions
- Complications: Systemic embolism due to blood clots can lead to serious heart rhythm disturbances or exacerbate heart failure. Pulmonary embolism may also complicate the clinical picture.
Differential Diagnosis: To differentiate peripartum cardiomyopathy from other conditions, it is important to consider:
- Pre-existing dilated cardiomyopathy
- Pulmonary thromboembolism
- Amniotic fluid embolism
- Myocardial infarction
- β2 agonist-associated pulmonary oedema in patients given this medication to delay premature labour
- Management:
- Management of peripartum cardiomyopathy is similar to the treatment of other causes of heart failure and may include:
- Oxygen therapy
- Diuretics
- Vasodilators
- Angiotensin-converting enzyme (ACE) inhibitors (if postpartum)
- Thromboprophylaxis: Thromboprophylaxis is an important aspect of management. If tachycardia persists, a careful addition of a β-adrenergic blocker may be beneficial, especially if cardiac output is stable.
- Severe Cases:The most severely affected patients may require:
- Intubation and ventilation
- Monitoring with inotropic support
- Occasional temporary support from devices such as an intra-aortic balloon pump or ventricular assist device
- In extreme cases, heart transplantation may be the only option for survival
- Prognosis: Approximately 50% of women with peripartum cardiomyopathy recover spontaneously and fully. Most maternal deaths occur shortly after diagnosis, with cardiomyopathy being responsible for nearly a quarter of these deaths.
- Recent data indicate a 5-year survival rate of 94%. Patients are advised to continue taking an ACE inhibitor as long as left ventricular function remains impaired.
- The prognosis and risk of recurrence depend on whether left ventricular size normalizes within 6 months of delivery.
- Women with severe heart dysfunction, characterized by an LV end diastolic dimension of ≥6 cm and fractional shortening ≤21%, are unlikely to regain normal heart function.
- Those whose left ventricular function and size do not return to normal before a subsequent pregnancy are at significant risk of worsening heart failure (50% risk), death (25% risk), or recurrent peripartum cardiomyopathy in future pregnancies.
- Such women should be counseled against future pregnancies.
Arrhythmias
- Ventricular Premature Complexes (VPC) are frequently observed during pregnancy.
- Many pregnant women notice strong heartbeats following a VPC, which can cause symptoms like dizziness, faintness, and palpitations; however, these incidents typically do not signify serious arrhythmias.
- Sinus Tachycardia
- Sinus tachycardia requires further investigation to rule out potential underlying conditions such as:
- Blood loss
- Infection
- Heart failure
- Thyrotoxicosis
- Pulmonary embolism
- The most prevalent arrhythmia during pregnancy is supraventricular tachycardia (SVT).
- While uncommon, SVT can manifest for the first time during pregnancy; research indicates that 22% of 63 women with SVT experienced worsened symptoms during pregnancy.
- Vagal manoeuvres are ineffective in half of SVT cases.
- Treatment Options
- Propranolol, verapamil, and adenosine are approved by the FDA for the rapid termination of SVT.
- Adenosine may be preferred over verapamil as it likely does not cross the placenta and can be safely used during pregnancy for SVTs unresponsive to vagal stimulation.
- Flecainide is considered safe for treating fetal tachycardias.
- Propafenone and amiodarone should be avoided, particularly amiodarone due to its impact on fetal thyroid function.
- Temporary and permanent pacing, cardioversion, and implantable defibrillators are deemed safe during pregnancy.
Cardiac Arrest
- Manage cardiac arrest in pregnant women using the same protocols as for non-pregnant patients.
- Position pregnant women, especially those in late pregnancy, to relieve pressure on the inferior vena cava (IVC) and improve blood flow.
- If cardiopulmonary resuscitation (CPR) is necessary, tilt the pelvis while keeping the torso flat to facilitate effective external chest compressions.
- Consider an emergency Caesarean section to assist maternal resuscitation only if other resuscitative efforts fail.
Endocarditis Prophylaxis
- Infective endocarditis is uncommon during pregnancy, but it poses serious risks for both the mother and the baby.
- There have been fatal cases of endocarditis occurring during pregnancy before delivery, rather than due to infections caught at the time of giving birth.
Stratification of cardiac conditions according to risk of bacterial endocarditis

- Treatment for endocarditis in pregnant women is similar to treatment for non-pregnant individuals, which may include urgent valve replacement if necessary.
- If the baby is able to survive, it should be delivered before any operation on the mother.
- Antibiotic prophylaxisis required for:
- Individuals with prosthetic valves
- Those who have had a previous case of endocarditis
- Many cardiologists suggest that women with structural heart defects (like Ventricular Septal Defect (VSD)) also receive prophylaxis.
- The American Heart Associationcategorizes heart conditions into three risk levels:
- High risk
- Moderate risk
- Negligible risk (no need for antibiotic prophylaxis)
- Current recommendations in the UK state that:
- Administer amoxicillin 2 g intravenously (i.v.) along with gentamicin 120 mg i.v. at the start of labor or when membranes rupture, or before a Caesarean section.
- Follow this with amoxicillin 500 mg orally (or i.m./i.v. depending on the patient's condition) 6 hours later.
- For women allergic to penicillin, vancomycin 1 g i.v. or teicoplanin 400 mg i.v. can be used instead of amoxicillin.
The document Chapter Notes: Heart disease in pregnancy is a part of the NEET PG Course Gynaecology and Obstetrics.
All you need of NEET PG at this link: NEET PG
FAQs on Chapter Notes: Heart disease in pregnancy
| 1. What are the physiological adaptations that occur in the cardiovascular system during pregnancy? |  |
Ans. During pregnancy, the cardiovascular system undergoes several adaptations to accommodate the increased metabolic demands of the mother and fetus. These include an increase in blood volume (by approximately 30-50%), an increase in cardiac output (primarily due to increased stroke volume), and changes in heart rate. Systemic vascular resistance decreases due to the effects of progesterone and other factors, leading to lower blood pressure in the first and second trimesters. Additionally, the heart may undergo slight hypertrophy and changes in position due to the growing uterus.
| 2. How are cardiac conditions managed during pregnancy? |  |
Ans. Management of cardiac conditions during pregnancy is tailored to the specific condition and its severity. It often involves a multidisciplinary approach, including obstetricians, cardiologists, and anesthesiologists. Close monitoring is essential, and treatment may include medication adjustments, lifestyle modifications, and in some cases, surgical interventions. In high-risk situations, such as severe congenital heart disease or significant arrhythmias, careful planning for labor and delivery is crucial to ensure maternal and fetal safety.
| 3. What are the risks associated with mechanical heart valves in pregnant women? |  |
Ans. Pregnant women with mechanical heart valves face several risks, including thrombosis, which can lead to embolic events, and bleeding complications from anticoagulation therapy. The use of anticoagulants, particularly warfarin, poses risks during pregnancy due to potential teratogenic effects and bleeding. Therefore, careful management of anticoagulation is critical, often involving switching from warfarin to heparin during the first trimester and the immediate postpartum period to reduce these risks.
| 4. What are the common arrhythmias that can occur during pregnancy, and how are they treated? |  |
Ans. Common arrhythmias during pregnancy include atrial fibrillation, supraventricular tachycardia, and ectopic beats. These conditions can be exacerbated by the increased blood volume and hormonal changes. Treatment typically focuses on symptom management and may include beta-blockers or antiarrhythmic medications deemed safe during pregnancy. In severe cases, electrical cardioversion may be considered. Continuous monitoring is essential, especially in women with pre-existing cardiac conditions.
| 5. Why is endocarditis prophylaxis important during pregnancy, and who should receive it? |  |
Ans. Endocarditis prophylaxis is crucial for pregnant women with certain heart conditions, such as prosthetic heart valves, previous endocarditis, or specific congenital heart defects. The risk of infection during dental or surgical procedures is heightened due to increased cardiac workload and potential bacteremia. Prophylactic antibiotics are recommended before procedures that are likely to cause bacteremia to prevent endocarditis. Consultation with a healthcare provider is essential to determine the need for prophylaxis based on individual risk factors.
Related Searches
Free, MCQs, past year papers, Important questions, Viva Questions, study material, video lectures, Chapter Notes: Heart disease in pregnancy, shortcuts and tricks, Extra Questions, practice quizzes, Sample Paper, ppt, Previous Year Questions with Solutions, pdf , Exam, Summary, mock tests for examination, Chapter Notes: Heart disease in pregnancy, Semester Notes, Objective type Questions, Chapter Notes: Heart disease in pregnancy;






