Assignment: Is Matter Around Us Pure?
Multiple Choice Questions (MCQs)
Q1. Which of the following is a homogeneous mixture?
(a) Oil and water
(b) Air
(c) Sand and iron filings
(d) Smoke
Ans: (b)
Air has uniform composition throughout, so it is a homogeneous mixture.
Q2. Which of the following will show the Tyndall effect?
(a) Salt solution
(b) Sugar solution
(c) Milk
(d) Copper sulphate solution
Ans: (c)
Milk is a colloid; its particles scatter light and show the Tyndall effect.
Q3. Which method is best to separate camphor from a mixture of salt and camphor?
(a) Evaporation
(b) Sublimation
(c) Filtration
(d) Sedimentation
Ans: (b)
Camphor sublimes on heating and can be separated by sublimation.
Q4. Which technique is most suitable to separate the pigments of ink?
(a) Filtration
(b) Chromatography
(c) Evaporation
(d) Crystallization
Ans: (b)
Chromatography separates components based on different solubilities/affinities.
Q5. A suspension differs from a solution because its particles:
(a) Are smaller than 1 nm
(b) Do not scatter light
(c) Settle on standing
(d) Pass through filter paper
Ans: (c)
Suspended particles are large and settle on standing.
Short Question/Answer (Q&A)
Q1. What is a pure substance (in scientific terms)?
Ans: A pure substance contains only one kind of particle and has uniform composition and properties throughout.
Q2. What is meant by concentration of a solution?
Ans: It is the amount of solute present in a given amount of solution or solvent (e.g., mass percent).
Q3. Give one difference between mixture and compound.
Ans: Mixture has variable composition and components retain properties; compound has fixed ratio and new properties.
Q4. Why can't filtration separate solute from a true solution?
Ans: Solute particles are too small (<1 nm) and pass through filter paper pores.
Q5. What is the Tyndall effect? Give one daily-life example.
Ans: Scattering of light by colloidal particles; visible sunbeams in a dusty room.
Activity-Based Questions
Q1. Shine a torch through three beakers: salt solution, milk, and copper sulphate solution. In which beaker is the light path visible and why?
Ans: The light path is visible in the beaker containing milk. Milk is a colloid (emulsion) in which tiny fat/protein droplets act as colloidal particles. These particles are large enough to scatter the incident light-a phenomenon called the Tyndall effect-so the beam becomes visible along its path. In contrast, salt solution and copper sulphate solution are true solutions with particle sizes smaller than 1 nm; such particles do not scatter light, so the path of the beam is not visible in those beakers.
Q2. Prepare chalk-water (suspension) and allow it to stand. What happens? How will you obtain clear water quickly?
Ans: Chalk-water forms a suspension. The chalk particles are relatively large and insoluble, so when the mixture is left undisturbed, the particles settle at the bottom due to gravity-this is sedimentation-leaving comparatively clearer water above. To obtain clear water quickly, you can:
- Centrifuge the mixture to force the heavier chalk particles to settle rapidly, producing a clear supernatant,
or - Allow sedimentation and then carefully decant (pour off) the clear supernatant, or
- Add a suitable coagulant/flocculant (if permitted in the experiment) to help fine particles clump together and settle faster, followed by decantation/filtration.
These methods exploit the large particle size and instability of suspensions to separate the solid from the liquid efficiently.
Research-Based Question
Q1. Research and submit a research paper: How do stirring and particle size affect the rate of dissolution of sugar in water?
Ans: Title: Effect of Stirring and Particle Size on the Rate of Dissolution
Objective:
To investigate how (i) stirring and (ii) particle size (granulated vs powdered sugar) affect the time taken for a fixed amount of sugar to dissolve in water at room temperature.
Background Theory (Short):
- Dissolution occurs when solute particles disperse uniformly among solvent particles.
- Rate of dissolution increases with increased surface area (finer particles) and stirring (enhances contact and reduces concentration gradient around solute).
- Temperature also affects rate, so it must be kept constant for fair comparison.
Hypotheses:
H1: Stirring will decrease the time required for sugar to dissolve.
H2: Powdered sugar (smaller particle size → larger surface area) will dissolve faster than granulated sugar.
Variables:
- Independent variables: (a) Stirring vs no stirring, (b) Particle size (granulated vs powdered).
- Dependent variable: Time taken (seconds) for complete dissolution of a fixed amount of sugar.
- Controlled variables: Temperature of water (room temp), volume/mass of water, mass of sugar, same beakers, same stirring speed (when used).
Materials/Apparatus:
Beakers (3-4), measuring balance, teaspoon/spatula, stopwatch, glass rod, thermometer, granulated sugar, powdered sugar, 100 mL measuring cylinder, room-temperature water.
Safety & Handling:
Handle glassware carefully; avoid spilling water near electrical points; do not taste chemicals in lab.
Procedure:
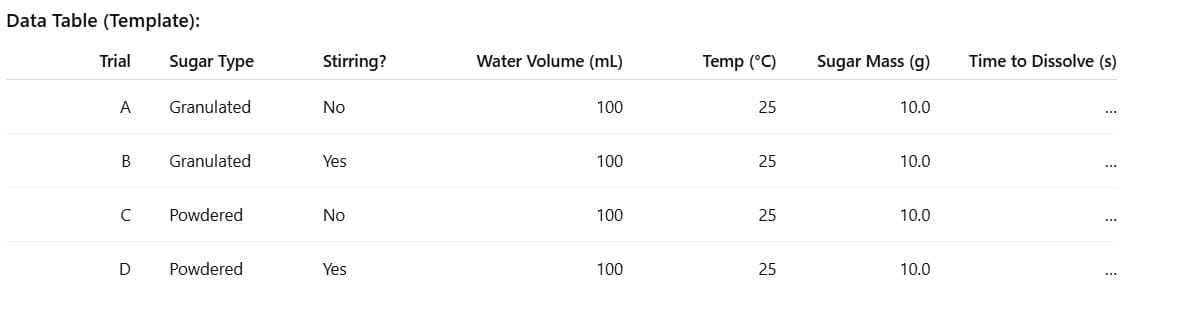
- Measure 100 mL of room-temperature water (note temperature, e.g., 25 °C) into each of 4 beakers.
- Weigh 10.0 g of granulated sugar and 10.0 g of powdered sugar.
- Trial A (Granulated, No Stirring): Add 10.0 g granulated sugar to beaker A. Start stopwatch. Do not stir. Stop timing when no visible crystals remain. Record time.
- Trial B (Granulated, With Stirring): Add 10.0 g granulated sugar to beaker B. Stir gently but steadily (same speed throughout). Record time to complete dissolution.
- Trial C (Powdered, No Stirring): Repeat step 3 using powdered sugar. Record time.
- Trial D (Powdered, With Stirring): Repeat step 4 using powdered sugar. Record time.
- Keep temperature constant (same room, similar timing between trials). If temperature changes, note it.
Calculations/Processing:
Compute % decrease in time due to stirring for the same sugar type:
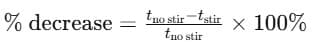
Compare times for granulated vs powdered (with the same stirring condition).
Observations:
- Note clarity of solution at the end.
- Note if any lumps formed and broke down over time.
- Keep a note of consistency of stirring.
Results (Sample Wording):
- With stirring, dissolution was faster than without stirring for both sugar types.
- Powdered sugar dissolved faster than granulated sugar under identical conditions.
Conclusion:
Both stirring and smaller particle size (greater surface area) increase the rate of dissolution. This supports the hypotheses and aligns with particle-level explanations of mixing and diffusion.
Think & Explain
Q1. Lemonade tastes the same throughout. What does this indicate about particle distribution?
Ans: It indicates that the solute particles (sugar, citric acid and other lemon components) are uniformly distributed among the water particles, forming a homogeneous solution. Because the composition is uniform at every point, any sip taken from the top, middle, or bottom has the same taste and concentration.
The dissolved particles are very small (solution-level, < 1 nm), so they do not settle, do not scatter light (no Tyndall effect), and pass through filter paper. This uniform distribution explains why the properties-like taste, colour, and sweetness-remain consistent throughout the lemonade, unlike heterogeneous mixtures (e.g., oil and water) where different parts can taste or look different.
Crossword Puzzle (with answers)


Ans:

FAQs on Assignment: Is Matter Around Us Pure?
| 1. What is meant by 'pure matter'? |  |
| 2. How can we determine if a substance is pure or a mixture? |  |
| 3. What are the different types of mixtures, and how do they differ from pure substances? |  |
| 4. Why is it important to understand whether matter is pure or a mixture? |  |
| 5. Can you provide examples of pure substances and mixtures found in everyday life? |  |






