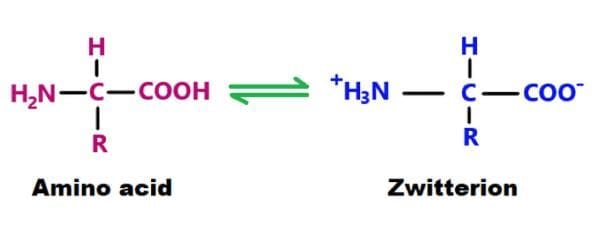
Introduction - Amino Acids
Amino acids are molecules that contain an amino group, a carboxylate group and a side chain (R group) attached to an alpha carbon.
- The alpha carbon is chiral when it is bonded to four different groups.
- All amino acids show this signature characteristic of a chiral carbon.
- Each amino acid has a distinct R group.
- At physiological pH, amino acids exist as zwitterions so the backbone does not determine the protein's net charge.
- The primary function of amino acids is to act as monomers for protein synthesis.
- Proteins are highly versatile biomolecules in structure and function.
- This versatility results from the combination of all amino acids.
- Unlike other biological monomers, there are twenty amino acids with different side chains.
- Each side chain gives an amino acid its distinguishing structural and functional properties.
Major Groups Of Amino Acids
There are seven major types of side chains found in amino acids.
Carboxylate Group
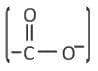
- This group occurs in two amino acids: aspartate and glutamate.
- These amino acids are acidic and carry a negative charge at physiological pH.
- The negative charge can form salt bridges with positively charged amino acids.
- Negatively charged side chains can also bind metal ions; for example, glutamate binds zinc.
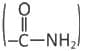
- Asparagine and glutamine have amide groups in their side chains.
- They are neutral and polar at physiological pH.
- The presence of both oxygen and nitrogen lets them act as hydrogen-bond acceptors and donors.
- They readily form hydrogen bonds with water, increasing solubility.
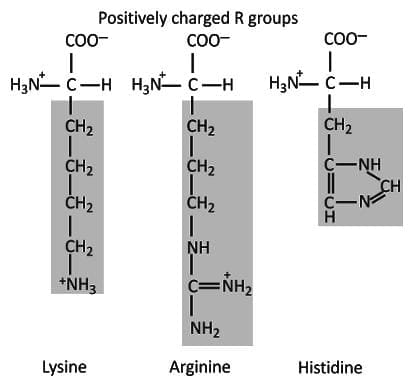
- Lysine, arginine and histidine have basic amine side chains.
- These amino acids are positively charged at physiological pH.
- Both acidic and basic amino acids increase protein and peptide solubility.
- Negatively charged and positively charged side chains can pair to form salt bridges.
- A large proportion of amino acids have aliphatic structures.
- Out of twenty-five amino acids, those with aliphatic side chains include alanine, valine, leucine, isoleucine and methionine.
- Aliphatic side chains are hydrophobic and often reside in the cores of globular proteins.
- They are also common in membrane-spanning regions of proteins.
- Phenylalanine, tyrosine and tryptophan contain aromatic rings.
- The aromatic ring absorbs light around 260 nm.
- This absorbance property is used in protein quantification by spectrophotometry.
- Serine, threonine and tyrosine contain hydroxyl groups in their side chains.
- Tyrosine has an -OH group attached to a benzene ring.
- Cysteine contains a thiol (-SH) side chain.
- Cysteine and methionine are sulphur-containing amino acids.
- Only cysteine forms disulphide bonds with another cysteine residue.
- Glycine and proline form a distinct category because of their unusual structures.
- Glycine lacks a side chain and has only a hydrogen atom as its side group.
- Because glycine's side chain is minimal, it contributes little to tertiary folding constraints.
- Proline is a nonpolar aliphatic amino acid with a cyclic pyrrolidine side chain.
- The cyclic structure of proline makes the peptide chain more rigid, unlike glycine which increases flexibility.
- Proline contains an amino group, a carboxyl group and a pyrrolidine side chain.
- Proline is classified as an imino acid.
- It slows the rate of peptide bond formation when present between two residues.
- The nitrogen of proline cannot serve as a hydrogen-bond donor but can act as an acceptor.
- For this reason, the proline-proline peptide bond is among the slowest to form.
- Proline imparts conformational rigidity to the peptide chain; thermophilic organisms have relatively high proline content in their proteins.
Biologically Active Amino Acids
In addition to forming proteins, amino acids serve several other biological roles.
- A neurotransmitter is a chemical messenger that transmits signals across a chemical synapse.
- Some neurotransmitters are alpha-amino acids or their derivatives.
- Examples include lycine, glutamate, gamma-aminobutyric acid (GABA, a glutamate derivative) and serotonin and melatonin (tryptophan derivatives).
Thyroxine, a tyrosine derivative produced in animal thyroid glands, and indole acetic acid, a tryptophan derivative found in plants, are hormones.
- Amino acids are precursors for nucleotides, heme and chlorophyll.
- Standard and non-standard amino acids act as metabolic intermediates.
- For example, arginine, citrulline and ornithine participate in the urea cycle.
Modified Amino Acids
- After translation, residues in a polypeptide are often enzymatically modified.
- These changes are called post-translational modifications.
- Prothrombin contains a calcium-binding residue known as gamma-carboxyglutamic acid.
- Collagen contains 4-hydroxyproline and 5-hydroxylysine as important structural components.
- Phosphorylation is the most common post-translational modification.
- Phosphorylation of serine, threonine and tyrosine often regulates protein activity.
Titration Of Amino Acids
- Amino acids have ionizable groups whose ionic forms depend on pH.
- Titration can be used to study the effect of pH on amino acid structure.
- Titration also helps determine the reactivity of amino acid side chains.
- The simplest amino acid for study is alanine, which has two titratable groups.
- In strongly acidic solution, alanine exists with an un-ionized carboxyl group and a protonated ammonium group, giving a net charge of +1.
- As OH- concentration increases during titration, the carboxyl group loses a proton to form a negatively charged carboxylate.
- Further addition of OH- removes a proton from the ammonium group.
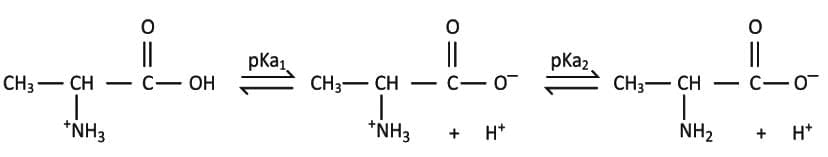
- After the carboxyl group loses its proton, alanine can be electrically neutral.
- The pH at which an amino acid has no net charge is called the isoelectric point (pI).
- The pI for alanine may be calculated from its pKa values as shown below.

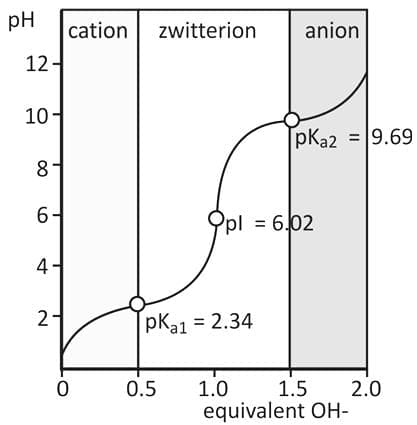
The pI Of Amino Acids With An Ionizable Side Chain
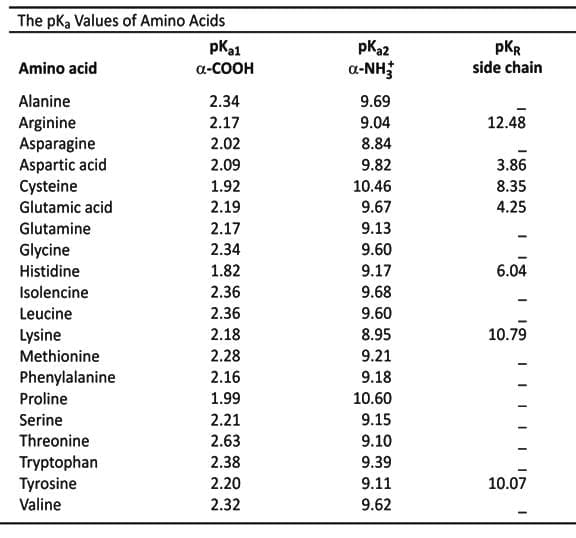
- In a polyprotic acid, protons are first lost from the group with the lowest pKa.
- The pI of an amino acid is the average of the pKa values of the groups that ionize in similar ways.
- For lysine, the pI is the average of the pKa values of the groups that are positively charged in their acidic form.
- For glutamate, the pI is the average of the pKa values of groups that are uncharged in acidic form but negatively charged in basic form.
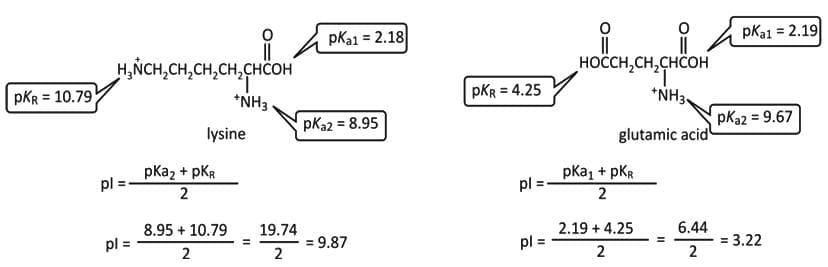
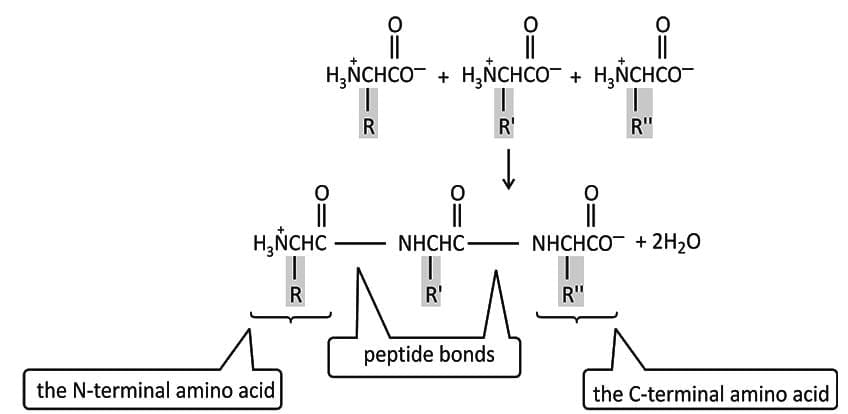
Peptide Bonds
Peptide bonds are the covalent amide links that join amino acid residues in a chain.
By convention, peptide sequences are written with the free amino group (N terminus) on the left and the free carboxyl group (C terminus) on the right.
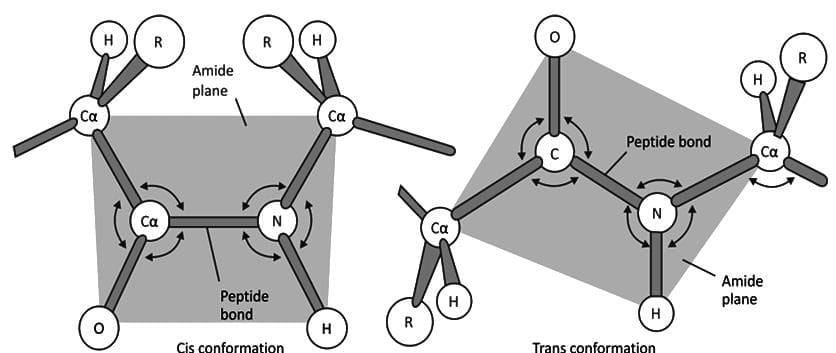
Cis And Trans Peptide Bonds
- In naturally occurring proteins, most peptide bonds are in the trans configuration.
- When any amino acid is followed by proline (X-proline), the peptide bond may adopt a cis configuration.
- A peptide bond has partial double-bond character, about 40% double bond, due to electron delocalization.
- The trans configuration is generally more stable than cis because of steric hindrance.
- Consequently, the alpha-carbons of adjacent residues are usually trans to each other.
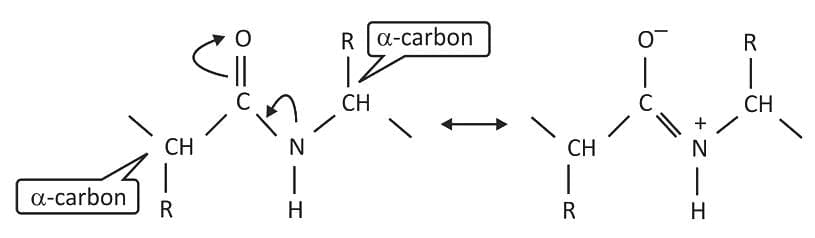
- The partial double-bond character prevents free rotation of the peptide bond.
- The carbon and nitrogen atoms of the peptide bond and their attached atoms lie rigidly in a plane.
- This planarity constrains how a polypeptide chain can fold and influences three-dimensional structure.
- The omega (ω) torsion angle is close to 0° for a cis peptide bond and about 180° for a trans bond.
- Thus X-proline peptide bonds commonly occur in the cis form.
Examples Of Peptides
- Dipeptide: Aspartame, a sweetening agent used as a sugar replacement, is composed of aspartic acid and phenylalanine.
- Tripeptide: GSH, formed from glutamic acid, cysteine and glycine, helps amino acid absorption and protects red blood cells by breaking H2O2.
- Octapeptides: Hormones such as oxytocin and vasopressin (ADH) are composed of eight amino acids.
- Polypeptides: Chains of 10-50 amino acids include examples such as insulin.


