Electron Transport Chain
Introduction: Electron Transport Chain
- Oxidation-reduction reactions, also called redox reactions, are chemical processes in which electrons move from one molecule to another.
- The electron-donating molecule is the reducing agent (reductant) and the electron-accepting molecule is the oxidizing agent (oxidant).
- Reducing and oxidizing agents function as conjugate redox pairs.
- The general form of such a reaction is: electron donor → e- + electron acceptor.
- For example, Fe2+ → e- + Fe3+ shows Fe2+ as the electron donor and Fe3+ as the acceptor; together they form a redox pair.
- The citric acid cycle oxidizes oxaloacetate to two molecules of CO2, producing three NADH and one FADH2.
- These reduced coenzymes contain electrons with high transfer potential that enter the mitochondrial electron transport chain.
- The ultimate electron acceptor in aerobic respiration is O2, which is reduced to H2O.
- Mitochondria and chloroplasts use electron transport to create an electrochemical proton gradient that drives ATP synthesis.
- Some archaea, bacteria and anaerobes also gain energy from electron transfers between inorganic molecules.
- During glycolysis and β-oxidation, NADH and FADH2 are oxidized and their electrons are used to generate ATP.
- ATP produced by this mechanism is called chemiosmotic phosphorylation.
- DG'° for electron transfer from NADH to oxygen is -219.2 kJ·mol-1, much larger than DG°' for ATP hydrolysis (-30.5 kJ·mol-1), indicating substantial stored energy in NADH.
- The electron transport pathway conserves this energy so it can drive the synthesis of multiple ATP molecules.
- Electrons from NADH and FADH2 pass through membrane-bound carriers to O2, and this electron flow is coupled to H+ transport across the inner mitochondrial membrane.
- Accumulation of H+ in the intermembrane (perimitochondrial) space creates a proton gradient termed the proton-motive force (PMF).
- The PMF drives ATP synthase to produce ATP from ADP and inorganic phosphate.
Mitochondrial Structure
- A mitochondrion has two membranes: an outer membrane that helps maintain organelle shape and a less permeable inner membrane.
- The outer membrane contains porin, a protein that forms pores allowing molecules under ~10 kDa to diffuse freely.
- The space between the two membranes is the intermembrane space.
- The inner membrane is largely impermeable and restricts free movement of most molecules.
- The inner membrane forms invaginations called cristae that increase membrane surface area.
- The inner and outer membranes differ in lipid and protein composition; cardiolipin is a major phospholipid of the inner membrane and is absent from the outer membrane.
- Phosphatidylinositol and cholesterol are important constituents of the outer membrane but are absent from the inner membrane.
- The outer membrane contains enzymes involved in phospholipid biosynthesis.
- No outer-membrane proteins are encoded by mitochondrial genes; all are encoded in the nucleus.
- Some molecules cross the membranes via specific transporters.
- The TCA cycle enzymes are located in the matrix of the mitochondrion.
- Except for succinate dehydrogenase, the multisubunit complexes of the electron-transport chain and ATP synthase contain proteins encoded by both nuclear and mitochondrial genes.
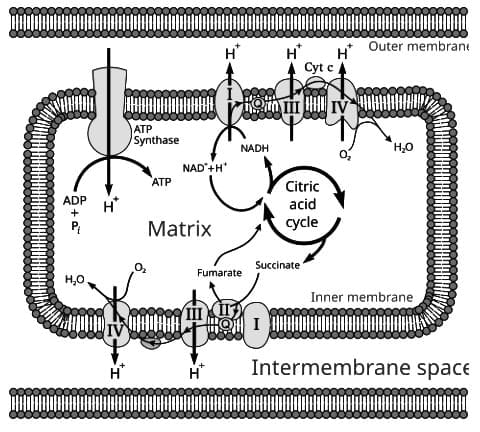
The Components of the Electron Transport Chain
The electron transport chain removes electrons from reduced carriers such as NADH and transfers them to oxygen to form H2O.
NADH
- NADH is produced in the matrix by pyruvate dehydrogenase, isocitrate dehydrogenase, α-ketoglutarate dehydrogenase and malate dehydrogenase.
- The ETC begins by reoxidizing NADH to NAD+ and channeling electrons into the respiratory chain.
- NADH transfers two electrons at a time as a hydride ion.
Flavoproteins
- Flavoproteins contain either FAD or FMN as a prosthetic group.
- They can accept or donate one electron or two electrons, so they often bridge two-electron and one-electron carriers.
- Typical standard reduction potentials for flavoproteins are around 0 V.
Coenzyme Q (CoQ) or Ubiquinone (UQ)
- Coenzyme Q consists of a quinone ring with a long isoprenoid side chain, commonly ten isoprene units in mammals.
- It is highly hydrophobic and diffuses freely within the inner membrane lipid bilayer.
- Like flavoproteins, CoQ can accept or donate one or two electrons.
- Ubiquinones are a family of compounds with the same quinone structure but differing isoprenoid tail lengths (six to ten units).
- Because it is not tightly protein-bound, CoQ serves as a mobile electron carrier between flavoproteins and cytochromes.
- Plastoquinones, the analogous carriers in photosynthesis, differ from ubiquinones in ring substituents and side-chain composition.
Cytochromes
- Cytochromes are proteins that contain a heme prosthetic group and function as one-electron carriers.
- The heme iron cycles between Fe2+ and Fe3+ during electron transfer.
- Cytochromes occur as monomeric proteins or as subunits of larger complexes that organise redox reactions efficiently.
- Cytochromes differ by their light-absorption spectra and are classified as a, b, c, and so on.
- Because spectral order does not match physiological sequence, the cytochromes found between ubiquinol and oxygen include b, c1, c, a and a3.
- Cytochromes b, c and c1 contain iron-protoporphyrin IX (heme).
- In cytochromes c and c1, the heme is covalently attached to the protein by thioether linkages; it is not covalently bound in other cytochromes.
- Heme B is present in cytochrome b; heme C is present in cytochromes c and c1.
- Three cytochrome types are distinguished by prosthetic group: cytochrome a - heme a; cytochrome b - heme b; cytochrome d - a tetrapyrrolic chelate of iron.
A distinct family, cytochrome P450 oxidases, absorbs light at 450 nm and participates in steroid synthesis and detoxification.
Cytochromes a and a3 contain heme a and form the terminal oxidase of the respiratory chain, often termed cytochrome oxidase.
Cytochrome c is the only soluble electron-transport protein that can be dissociated from the inner membrane by gentle treatment.
Iron-Sulphur Proteins
- Iron-sulfur proteins participate in one-electron transfers using Fe2+ and Fe3+ redox states.
- The simplest form is the FeS center, with iron tetrahedrally coordinated by four cysteines.
- Other forms include 2Fe-2S (two irons, two cysteines and two inorganic sulfides), 3Fe-4S and 4Fe-4S centers.
- The iron atoms may be reduced or oxidized, and protein environment tunes their electron affinity widely.
- NADH dehydrogenase (Complex I) contains both 2Fe-2S and 4Fe-4S types of centers.
Copper Proteins
- Copper-bound proteins participate in one-electron transfers involving Cu+ and Cu2+ redox states.
Overview of the Electron Transport Chain
- Electrons flow along the chain from donors to acceptors until they reduce oxygen, the ultimate electron acceptor.
- The standard reduction potentials of carriers range between the NADH/NAD+ couple (-0.315 V) and the O2/H2O couple (0.816 V).
- The principal enzyme complexes are: I NADH-CoQ oxidoreductase; II FADH2-CoQ oxidoreductase; III CoQ-cytochrome c oxidoreductase; IV cytochrome c oxidase.
- ATP synthase functions as Complex V and synthesises ATP using the proton-motive force.
- Complex II does not pump protons and so does not contribute to generating the PMF when transferring electrons from FADH2 to CoQ.
- Electrons move from carriers with lower (more negative) reduction potentials to those with higher (more positive) potentials.
- Oxygen has the most positive standard reduction potential and thus the greatest tendency to accept electrons.
- The order of carriers in the chain was established by spectroscopic methods.
Complex I (NADH-Coenzyme Q Reductase)
- Complex I accepts electrons from NADH and links glycolysis, the citric acid cycle and fatty acid oxidation to the electron transport chain.
- It is the largest respiratory complex, containing over 40 polypeptide chains.
- Complex I oxidises NADH and transfers electrons via FMN and iron-sulfur centres to ubiquinone (CoQ).
- Ubiquinone then delivers electrons to the cytochrome b-c1 complex (Complex III).
- The complex contains flavin mononucleotide (FMN) and multiple iron-sulfur proteins.
- Iron-sulfur centres are present as 2Fe-2S or 4Fe-4S clusters and are bound to cysteine residues on the protein.
- FMN is required for activity, so Complex I is classified as a flavoprotein.
- The complex binds NADH and transfers two electrons as a hydride to FMN, forming NAD+ and FMNH2.
- Subsequent electron transfer proceeds one electron at a time through a series of iron-sulfur centres.
Importance of FMN
- FMN accepts two electrons from NADH and donates electrons one at a time to iron-sulfur clusters.
- FMN and FAD link two-electron carriers and one-electron carriers in many redox pathways.
- The final step in Complex I transfers two electrons, one at a time, to coenzyme Q.
- CoQ is mobile in the membrane because its isoprenoid tail makes it highly hydrophobic.
- Electron transfer from NADH to CoQ is coupled to active transport of protons, moving four H+ per two electrons.
- The intermembrane-side of the inner membrane is the P face (positive), and the matrix side is the N face (negative).
Complex II
- Complex II, or succinate dehydrogenase (FADH2-CoQ oxidoreductase), is a membrane-bound enzyme of the TCA cycle that oxidises succinate to fumarate.
- It generates FADH2, which transfers electrons to CoQ.
- Complexes I and II both produce reduced coenzyme Q (CoQH2), the substrate for Complex III.
- Complex II is the only citric acid cycle enzyme that is an integral membrane protein and consists of four subunits.
- Two subunits are iron-sulfur proteins; the other two bind FAD covalently via a histidine residue and are called FP2.
- Complex II contains three Fe-S centres: one 4Fe-4S cluster, one 3Fe-4S cluster and one 2Fe-2S cluster.
- Succinate binds and transfers a hydride to FAD, producing FADH2 and fumarate.
- FADH2 then transfers electrons one at a time to the Fe-S centres, and finally to CoQ to form CoQH2.
- The free energy change for Complex II is too small to pump protons across the membrane, which explains why FADH2 yields about 1.5 ATP compared with ~2.5 ATP per NADH.
Complex III
- Complex III, or CoQ-cytochrome c oxidoreductase, transfers electrons from CoQH2 to cytochrome c via the Q-cycle.
- Ubiquinone (CoQ) is hydrophobic and freely mobile in the membrane and can carry one or two electrons, picking up a proton with each electron when reduced.
- The cytochrome b-c1 complex contains at least 11 polypeptides and functions as a dimer.
- Each monomer contains three hemes and an iron-sulfur protein and passes electrons from ubiquinone to cytochrome c.
- Cytochrome c is water-soluble and shuttles electrons in the intermembrane space from Complex III to Complex IV.
Q-Cycle
- The Q-cycle begins when CoQH2 binds to the QP site near the intermembrane face.
- One electron from CoQH2 transfers to the Rieske Fe-S protein and then to cytochrome c1, releasing two protons into the intermembrane space.
- The remaining electron converts CoQH2 into a semiquinone radical (CoQH•) still bound at QP; the semiquinone then transfers an electron to the bL heme.
- The bL heme passes the electron to the bH heme near the N face, which then reduces a CoQ at the QN site to form a bound semiquinone radical.
- A second CoQH2 binds at QP and repeats the steps, delivering another electron to cytochrome c1 and two more protons to the intermembrane space.
- Electrons transferred to the semiquinone at QN convert it to CoQH2 by uptake of two protons from the N face, and CoQH2 diffuses away.
- The net result is two electrons moved to cytochrome c and four protons released into the intermembrane space per two CoQH2 oxidised.
- Electrons that reach cytochrome c are carried through the intermembrane space to the CuA site of Complex IV.
Cytochrome c is coordinated by cysteine linkages to the heme and uses methionine and histidine residues to protect the heme iron from inappropriate ligands.
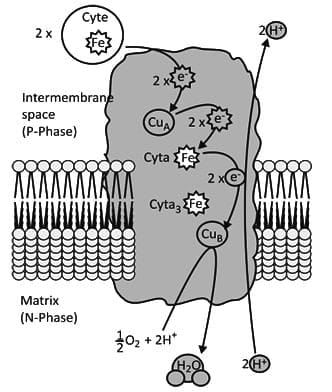
Complex IV
- Cytochrome oxidase (Complex IV) functions as a dimer, with each monomer containing about 13 polypeptide chains, including two cytochromes and two copper centres.
- Complex IV accepts one electron at a time from cytochrome c and passes electrons to oxygen.
- The complex contains cytochromes a and a3 and copper centres CuA and CuB.
- Copper sites cycle between Cu+ and Cu2+ and function as one-electron carriers.
- Cytochrome c transfers an electron to CuA; CuA transfers it to cytochrome a, which transfers it to cytochrome a3 and CuB.
- When the binuclear centre accumulates four electrons, O2 binds between the heme iron and copper and is reduced to water.
- The reduction of O2 by Complex IV requires four electrons; four protons are taken from the matrix and two protons are released into the intermembrane space per O2 reduced.
- By holding O2 until it receives four electrons, cytochrome oxidase prevents formation of reactive intermediates and enables efficient energy release during respiration.
The Mitochondrial Electron-Transport Chain
- Standard reduction potentials of the most mobile components are shown in diagrams and indicate where free energy is harvested to synthesise ATP.
- Complexes I, III and IV do not directly synthesise ATP; they pump protons to generate the proton gradient used by ATP synthase.
Inhibitors of the Electron-Transport Chain
- Several inhibitors have been identified that reveal how the electron-transport chain operates.
- Experimental oxygen electrode traces show changes in O2 concentration when inhibitors or substrates are added to mitochondria with excess ADP and Pi.
- Addition of β-hydroxybutyrate supports NAD+-linked oxidation because it serves as a source of ketone bodies during starvation.
- Rotenone and amytal inhibit NAD+-linked oxidation by blocking electron transfer from Complex I iron-sulfur centres to ubiquinone.
- Inhibition of electron transfer can increase reactive oxygen species (ROS) production, potentially damaging mitochondrial components and DNA.
- Succinate, produced in the TCA cycle, supports FAD-linked oxidation when added to mitochondria.
- Antimycin inhibits FAD-linked oxidation by blocking electron flow within Complex III.
- Cyanide inhibits cytochrome oxidase (Complex IV) by preventing O2 binding and thereby halting ATP production.
FAQs on Electron Transport Chain
| 1. What is the function of the electron transport chain in mitochondria? |  |
| 2. What are the main components of the electron transport chain? |  |
| 3. Why is flavin mononucleotide (FMN) important in the electron transport chain? |  |
| 4. How does the electron transport chain contribute to ATP production? |  |
| 5. What role does oxygen play in the electron transport chain? |  |


