NCERT Solutions: Combustion & Flame
Q1. List conditions under which combustion can take place.
Ans: The conditions required for combustion to take place are:

- Presence of a fuel. A fuel is any substance that can burn, for example coal, wood, petrol or gas.
- Air (or oxygen). Oxygen is needed to react with the fuel and keep the burning going.
- Ignition temperature (minimum temperature at which a substance catches fire). The fuel must be heated to this temperature so that combustion starts.
Q2. Fill in the blanks:
(a) Burning of wood and coal causes ................of air.
Ans: Pollution
(b) A liquid fuel, used in homes is ................
Ans: LPG
(c) Fuel must be heated to its ............ ................before it starts burning.
Ans: Ignition temperature
(d) Fire produced by oil cannot be controlled by ................
Ans: Water
Q3. Explain how the use of CNG in automobiles has reduced pollution in our cities.
Ans:
- Combustion of fuels like petroleum causes formation of un-burnt carbon particles along with carbon monoxide gas. These harmful pollutants enter the air and cause respiratory diseases.
- Compressed Natural Gas (CNG) burns more completely than petrol or diesel and produces much smaller quantities of harmful gases and soot particles. It emits lower amounts of carbon monoxide, hydrocarbons and particulate matter, and virtually no smoke. For these reasons, using CNG in vehicles reduces roadside air pollution and improves air quality in cities.
Q4. Compare LPG and wood as fuels.
Ans:
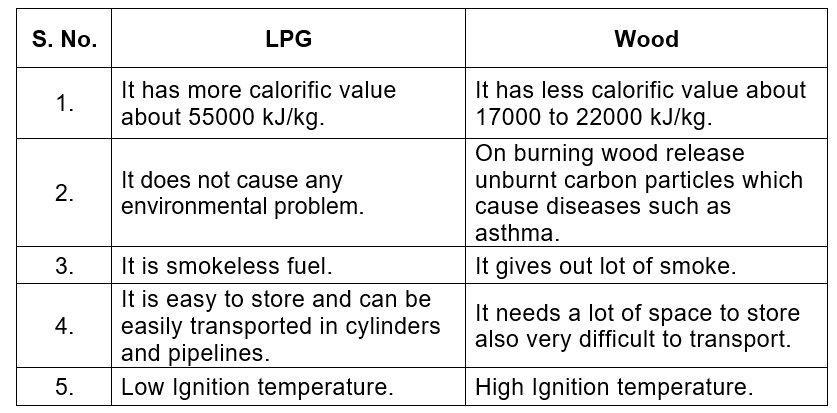
- Cleanliness: LPG is a much cleaner fuel - it burns without smoke or soot. Wood produces smoke and unburnt carbon particles.
- Heat output: LPG has a higher calorific value, so it gives more heat per kilogram than wood.
- Control and convenience: LPG burns with an easily controlled flame and is convenient to use; wood requires more effort to light and control.
- Storage and supply: LPG is stored in cylinders and is readily available for homes; wood needs space to store and is less convenient.
- Environmental impact: Burning wood releases more smoke and pollutants; LPG causes less air pollution when used properly.
Q5. Give reasons:
(a) Water is not used to control fires involving electrical equipment.
Ans: Water is a conductor of electricity. Using water on an electrical fire may cause electric shocks and can make the fire worse by causing short circuits. Therefore water must not be used to control fires involving electrical equipment.
(b) LPG is a better domestic fuel than wood.
Ans: LPG is better because it burns cleanly without smoke or soot, gives more heat for the same amount of fuel, and is easier to control and use in stoves. This reduces indoor air pollution and the risk of respiratory problems.
(c) Paper by itself catches fire easily whereas a piece of paper wrapped around an aluminium pipe does not.
Ans: Paper has a low ignition temperature and catches fire easily. When wrapped around an aluminium pipe, the metal conducts heat away from the paper, lowering the paper's temperature so it does not reach its ignition point and therefore does not catch fire.
Q6. Make a labelled diagram of a candle flame.
Ans:
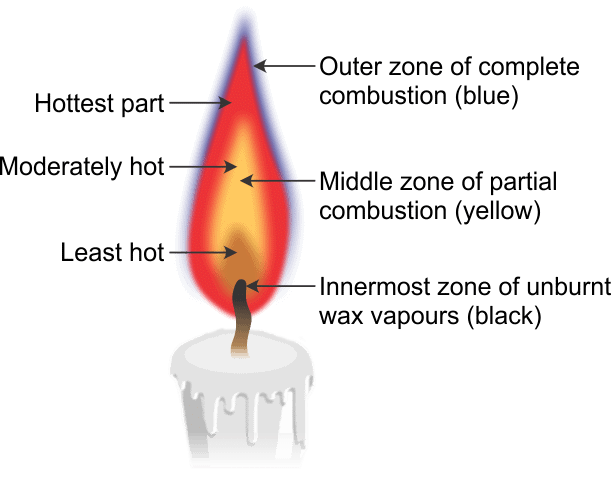
Labels to include on the diagram:
- Inner dark zone (centre) - contains unburnt wax vapour and is relatively cool.
- Bright yellow zone (luminous zone) - glows due to tiny carbon particles and is moderately hot.
- Outer non-luminous zone - where complete combustion occurs and temperature is highest.
Q7. Name the unit in which the calorific value of a fuel is expressed.
Ans: The calorific value of a fuel is expressed in kilojoules per kilogram (kJ/kg).
Q8. Explain how CO2 is able to control fires.
Ans: Being heavier than oxygen, CO2 settles over the burning material and forms a blanket that cuts off the supply of oxygen. Carbon dioxide also does not support combustion. By preventing contact between the fuel and oxygen, CO2 extinguishes the fire.
Q9. It is difficult to burn a heap of green leaves but dry leaves catch fire easily. Explain.
Ans: Green leaves contain a lot of moisture. Water in them absorbs heat and prevents the leaves from reaching ignition temperature, so they do not burn easily. Dry leaves have little or no moisture, so they heat up quickly to their ignition temperature and catch fire readily.
Q10. Which zone of a flame does a goldsmith use for melting gold and silver and why?
Ans: A goldsmith uses the outermost zone of the flame because this zone is the hottest. The high temperature there helps to melt gold and silver quickly and efficiently.
Q11. In an experiment 4.5 kg of a fuel was completely burnt. The heat produced was measured to be 180,000 kJ. Calculate the calorific value of the fuel.
Ans: Total mass of fuel = 4.5 kg
Heat produced by burning the given mass of fuel = 180,000 kJ
We know that calorific value of a fuel = (Heat produced) ÷ (Mass of fuel)
Calorific value = 180,000 kJ ÷ 4.5 kg
Calorific value = 40,000 kJ/kg
Q12. Can the process of rusting be called combustion? Discuss.

Rusted Iron Object
Ans:
Rusting is not the same as combustion. The main differences are:
- Speed: Combustion is a rapid reaction with oxygen that gives off heat and often light (flame). Rusting is a very slow reaction.
- Appearance: Combustion usually produces flames and visible heat; rusting produces no flame and little noticeable heat.
- Nature of change: Combustion is a burning process; rusting is a type of corrosion in which iron reacts slowly with oxygen in the presence of water.
Both are oxidation reactions and both release some heat, but because rusting is slow and produces no flame it is not called combustion.
Q13. Abida and Ramesh were doing an experiment in which water was to be heated in a beaker. Abida kept the beaker near the wick in the yellow part of the candle flame. Ramesh kept the beaker in the outermost part of the flame. Whose water will get heated in a shorter time?
Ans: The water in Ramesh's beaker will heat in a shorter time. The outermost zone of the flame is the hottest, while the yellow (luminous) zone is cooler, so placing the beaker in the outer zone transfers heat more quickly.
FAQs on NCERT Solutions: Combustion & Flame
| 1. What is combustion and how does it differ from burning? |  |
| 2. Why do some materials catch fire easily while others need higher temperatures? |  |
| 3. What exactly is the difference between complete combustion and incomplete combustion? |  |
| 4. How does a flame form, and why do different flames have different colours? |  |
| 5. What are fuels and how do we classify them for CBSE combustion topics? |  |






