Important Diagrams: Particulate Nature of Matter
Q1: a) Observe the diagram showing containers A, B, and C of different shapes. Water is filled up to 200 mL in Container A and then transferred to B and C. What changes and what remains the same when the water is transferred?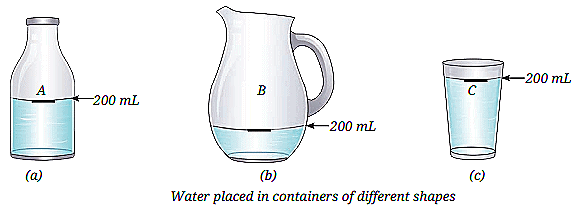
Ans: The shape of water changes according to the shape of each container. But the volume (200 mL) of water remains the same in all containers.
b) Based on the diagram, what does this activity prove about the shape and volume of liquids?
Ans: This activity proves that liquids do not have a fixed shape but they have a fixed volume.
c) Why was 200 mL level marked in each container before starting the experiment?
Ans: The 200 mL level was marked to measure the exact quantity of water and to observe whether the same amount of water is present after transferring it to other containers.
d) The water level looks different in B and C even though the amount is the same. Why does the water level appear higher in a narrow container and lower in a wide one?
Ans: Because liquids spread according to the shape of the container.
In a narrow container, water rises higher.
In a wide container, water spreads out and appears lower.
e) Fill in the blanks using observations from the diagram: Liquids do not have a fixed ________ but they have a fixed ________.
Ans: Liquids do not have a fixed shape but they have a fixed volume.
Q2: a) Study the diagram showing three particles arrangements labeled A, B, and C. Identify the state of matter of each arrangement.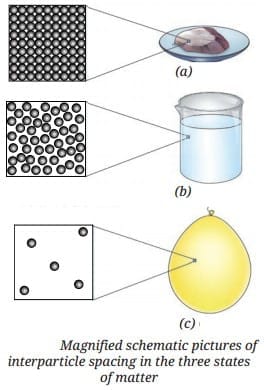
Ans:
- A - Solid: particles tightly packed
- B - Liquid: particles loosely packed but close
- C - Gas: particles far apart and moving freely
b) In the diagram, the particles in a solid are shown very close together. Why do solids have a fixed shape and volume?
Ans: Solids have a fixed shape and volume because the particles are tightly packed and cannot move freely.
c) In the diagram, the particles in a gas are very far apart. What can you say about the compressibility of gases?
Ans: Gases are highly compressible because there is a lot of space between their particles.
d) Imagine ice melting into water. Which change of state is shown? Explain why this happens.
Ans: The change of state is melting (solid → liquid).
It happens because heat energy is absorbed, which makes the particles move more freely.
e) Fill in the blanks using the diagram of states of matter: _________ have a fixed volume but no fixed shape, while _________ have neither fixed shape nor fixed volume.
Ans: Liquids have a fixed volume but no fixed shape, while gases have neither fixed shape nor fixed volume.
Q3 a) What do you observe in image (a) and image (b) after adding potassium permanganate to water?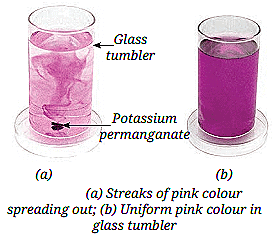
Ans:
In (a), streaks of pink colour are spreading.
In (b), the pink colour becomes uniform, showing complete mixing.
b) Why does the colour spread in water even without stirring?
Ans: Because particles of liquid are always moving, allowing potassium permanganate particles to spread and mix on their own.
c) Which property of matter is demonstrated in this activity?
Ans: The property shown is diffusion, which proves that particles of matter are in constant motion.
d) In which condition will diffusion happen faster-hot water or cold water? Give reason.
Ans: Diffusion happens faster in hot water because particles move more quickly at higher temperatures.
e) What does this experiment tell us about the arrangement of particles in liquids?
Ans: It shows that liquid particles are not tightly packed and can move freely, allowing mixing to occur naturally.
FAQs on Important Diagrams: Particulate Nature of Matter
| 1. What is the particulate nature of matter? |  |
| 2. How do particles behave in solids, liquids, and gases? |  |
| 3. What are the key differences between the three states of matter? |  |
| 4. What role does temperature play in the behaviour of particles? |  |
| 5. What is diffusion, and how is it related to the particulate nature of matter? |  |





