Important Diagrams: Elements, Compounds, and Mixtures
Q1: Observe the diagrams showing the particle arrangement of iron, water, and air.
a) Which one is an element, a compound, and a mixture?
b) Give one more example of each.
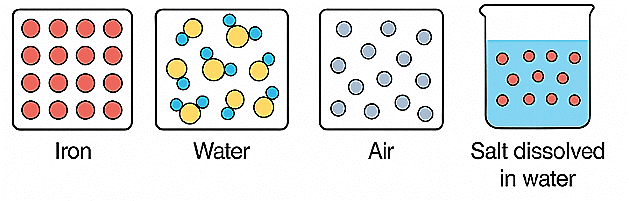
Ans: Elements contain only one type of atom. Compounds contain different elements bonded together chemically. Mixtures contain two or more substances mixed physically, not chemically.
Q2: A diagram shows copper sulfate crystals heated in a test tube. After heating, water droplets appear on the tube walls, and the substance turns white.
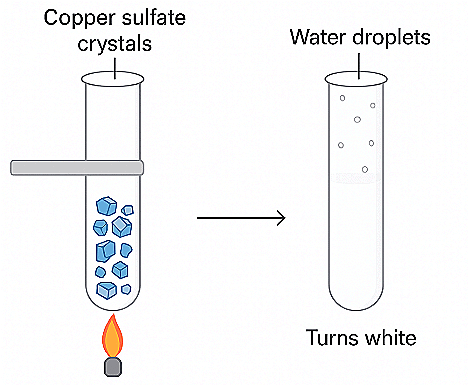
a) What change is happening - physical or chemical?
Ans: Physical change (loss of water of crystallization - dehydration).
Blue copper sulfate crystals are hydrated copper sulfate: CuSO₄·5H₂O (blue). On heating they lose their water of crystallization and become anhydrous CuSO₄ (white). The process can be reversed by adding water - the white anhydrous powder turns blue again - so it is considered a physical change (change in hydration state) rather than a permanent chemical decomposition.
b) What does this tell us about the composition of copper sulfate crystals?
Ans: The crystals contain water molecules incorporated into their crystal structure (water of crystallization). The formula CuSO₄·5H₂O shows this: 5 water molecules are part of each formula unit of the solid.
c) Suggest a method to confirm the presence of water in copper sulfate.
Ans: Use anhydrous copper sulfate (white) as a test: add a drop of water to it - it turns blue immediately.
Alternative tests: collect condensed vapor on a cold surface (water droplets), or use cobalt(II) chloride paper (turns from blue to pink on exposure to water vapour).
Q3: a) What is the difference between the mixture (before heating) and the product (after heating)?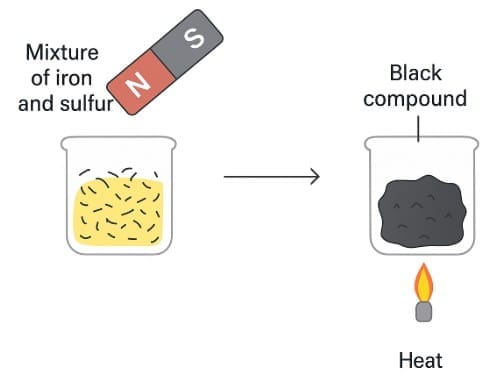 Ans: Before heating: a physical mixture of iron filings and sulfur powder - each retains its own properties (iron is magnetic, sulfur is yellow and non-magnetic).
Ans: Before heating: a physical mixture of iron filings and sulfur powder - each retains its own properties (iron is magnetic, sulfur is yellow and non-magnetic).
After heating: a chemical compound (iron sulfide) is formed - the properties change (black solid, not magnetic).
b) Chemical equation for this change.
Ans: Fe + S → FeS (One atom of iron reacts with one atom of sulfur to form iron(II) sulfide.)
c) Name two observable differences in their properties.
Ans:
Magnetic property: Before heating, iron filings are attracted by a magnet; after heating (FeS) the product is not attracted by a magnet.
Color & appearance: The mixture shows separate yellow (sulfur) and metallic grey/black (iron) particles; the product is a uniform black substance (iron sulfide).
(Other differences: change in hardness, change in smell on burning, cannot separate FeS by simple physical means.)
FAQs on Important Diagrams: Elements, Compounds, and Mixtures
| 1. What are elements? |  |
| 2. How do compounds differ from elements? |  |
| 3. What are mixtures, and how do they differ from compounds? |  |
| 4. Can you provide examples of homogeneous and heterogeneous mixtures? |  |
| 5. Why is it important to understand the differences between elements, compounds, and mixtures? |  |






