NEET Exam > NEET Notes > Subject-Wise Mind Maps > Mind Map: Haloalkanes and Haloarenes
Mind Map: Haloalkanes and Haloarenes
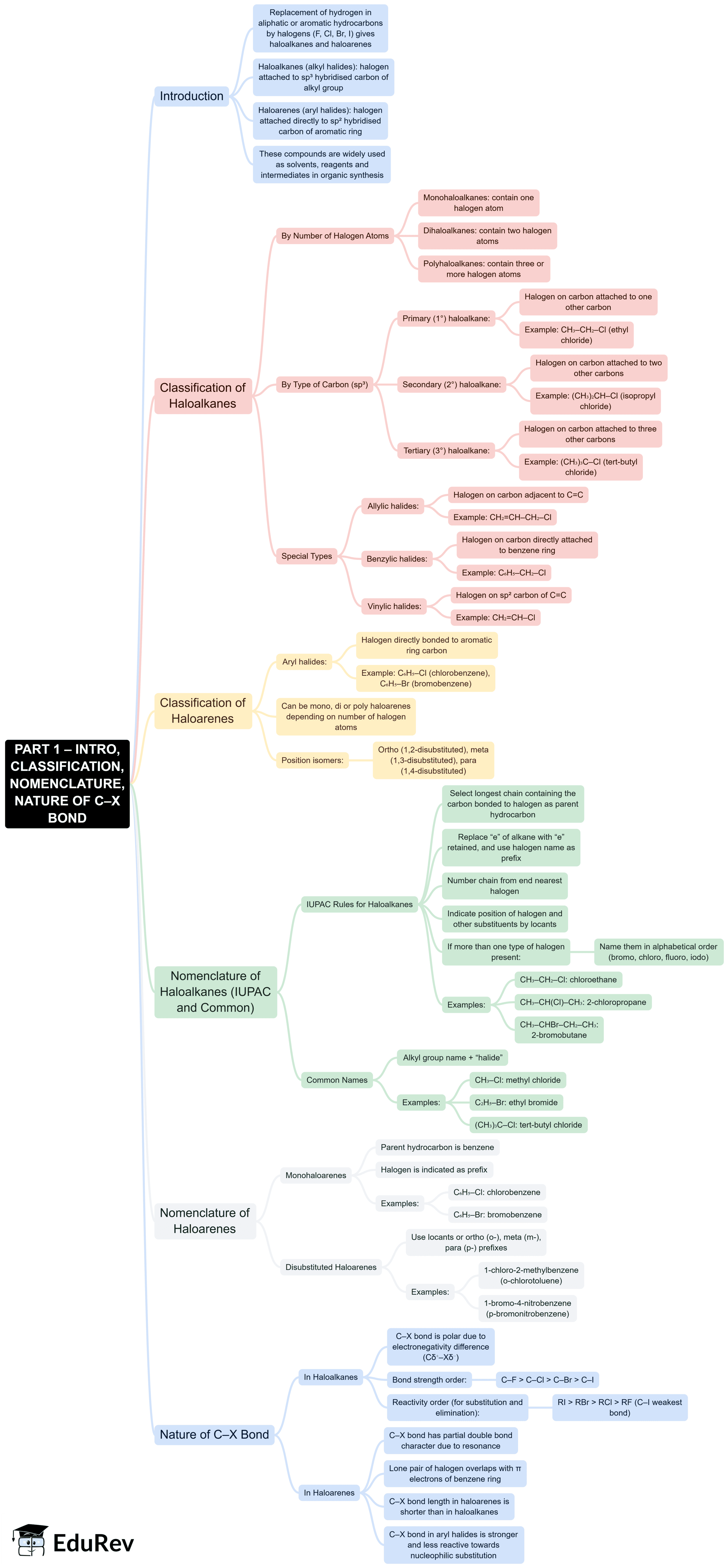
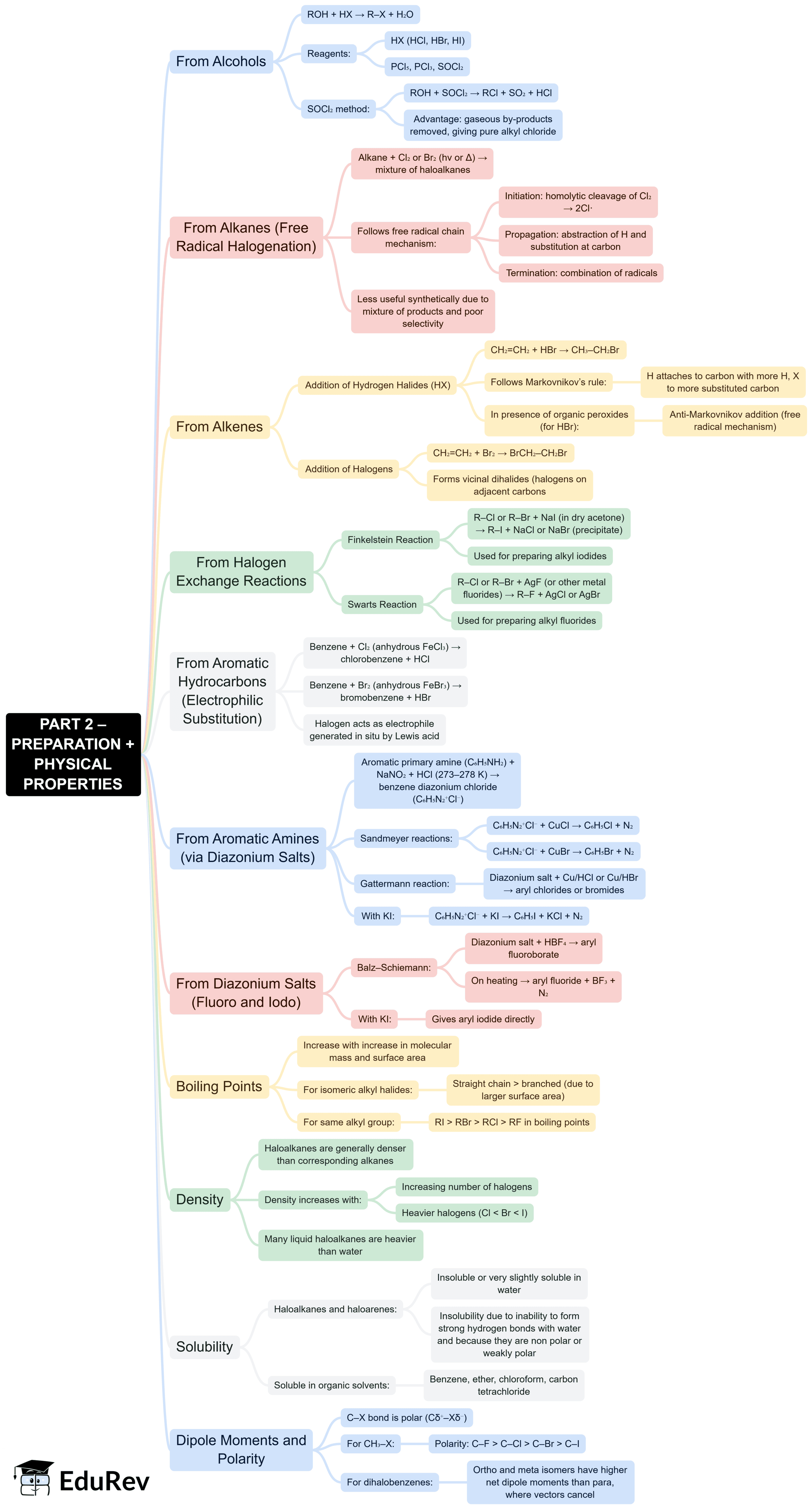
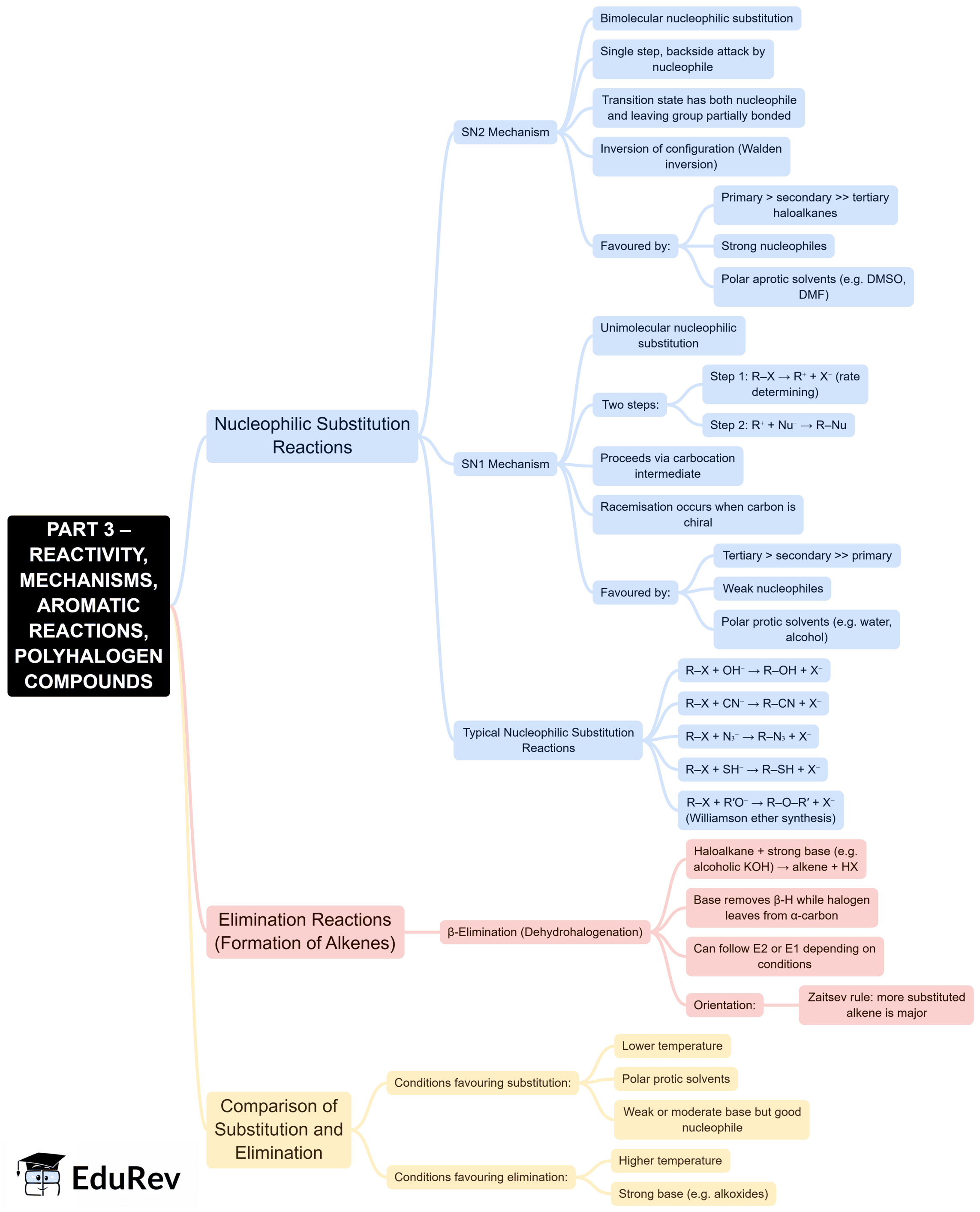
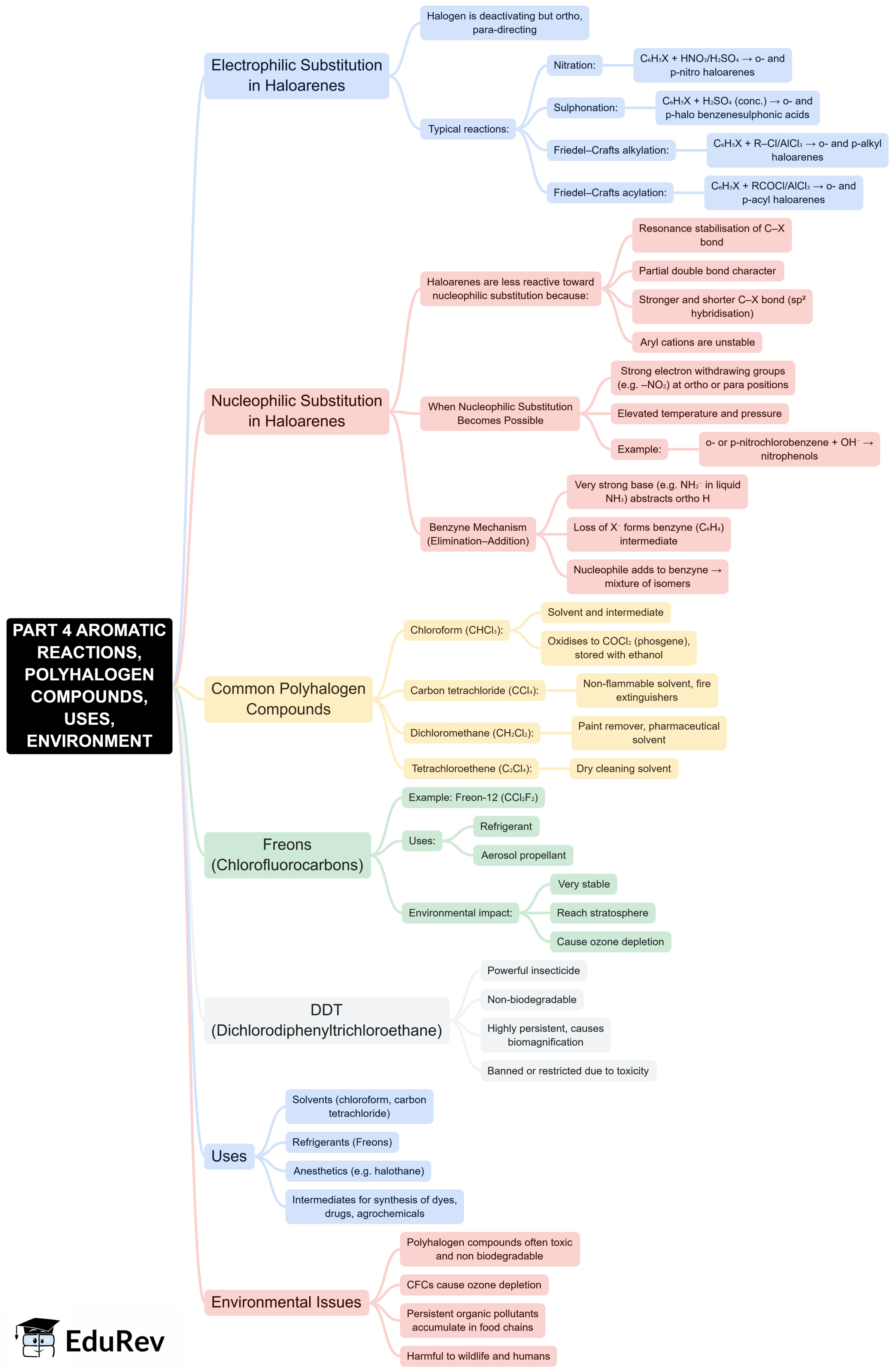
The document Mind Map: Haloalkanes and Haloarenes is a part of the NEET Course Subject-Wise Mind Maps for NEET.
All you need of NEET at this link: NEET
FAQs on Mind Map: Haloalkanes and Haloarenes
| 1. What are haloalkanes and how are they classified? |  |
Ans. Haloalkanes, also known as alkyl halides, are organic compounds containing carbon, hydrogen, and halogen atoms. They are classified based on the number of alkyl groups attached to the carbon bearing the halogen. The main classifications are primary (1°), secondary (2°), and tertiary (3°) haloalkanes. In primary haloalkanes, the carbon atom bonded to the halogen is attached to only one other carbon atom; in secondary haloalkanes, it is attached to two; and in tertiary haloalkanes, it is attached to three carbon atoms.
| 2. What are the physical properties of haloalkanes? |  |
Ans. Haloalkanes generally exhibit physical properties such as higher boiling and melting points compared to their corresponding alkanes due to the presence of polar C-X (carbon-halogen) bonds. They are typically colourless liquids or solids and are insoluble in water but soluble in organic solvents. The boiling point increases with the increase in the molecular weight of the haloalkane and the number of halogen atoms present.
| 3. How do haloalkanes undergo nucleophilic substitution reactions? |  |
Ans. Haloalkanes undergo nucleophilic substitution reactions through two primary mechanisms: the SN1 and SN2 mechanisms. In the SN1 mechanism, the reaction proceeds via the formation of a carbocation intermediate, making it a two-step process, and is favoured by tertiary haloalkanes. In the SN2 mechanism, the nucleophile attacks the haloalkane in a single concerted step, leading to the inversion of configuration, and is favoured by primary haloalkanes due to steric hindrance.
| 4. What are haloarenes and how do they differ from haloalkanes? |  |
Ans. Haloarenes, or aryl halides, are compounds where a halogen atom is attached to an aromatic ring. The key difference between haloarenes and haloalkanes lies in their structure and reactivity. Haloarenes typically have a more stable structure due to resonance in the aromatic ring, which makes them less reactive towards nucleophilic substitution compared to haloalkanes. Their reactivity often involves electrophilic aromatic substitution rather than straightforward nucleophilic substitution.
| 5. What are the applications of haloalkanes and haloarenes in industry? |  |
Ans. Haloalkanes and haloarenes have various applications in the industrial sector. Haloalkanes are used as solvents, in the production of pharmaceuticals, and as refrigerants. Haloarenes, on the other hand, serve as intermediates in the synthesis of dyes, pesticides, and pharmaceuticals. Additionally, some haloalkanes and haloarenes are utilised in making agrochemicals and as starting materials for organic synthesis.
Related Searches
Mind Map: Haloalkanes and Haloarenes, MCQs, Summary, Exam, ppt, past year papers, Previous Year Questions with Solutions, Important questions, Objective type Questions, Free, pdf , mock tests for examination, video lectures, shortcuts and tricks, Viva Questions, Mind Map: Haloalkanes and Haloarenes, practice quizzes, Sample Paper, Extra Questions, study material, Semester Notes, Mind Map: Haloalkanes and Haloarenes;





