Rapid Revision: Magnetic And Heating Effects
Introduction
- This chapter explains two important effects of electric current: the magnetic effect and the heating effect. It also describes how electric cells and batteries produce electricity through chemical reactions and how these effects are used in everyday devices.
- Electric current can create magnetic fields, produce heat, and power many machines and appliances.
Magnetic Effect of Electric Current
- When electric current flows through a conductor such as a wire, it produces a magnetic field around the wire. This phenomenon is called the magnetic effect of electric current.
- A compass needle placed near a current-carrying wire gets deflected, showing that the wire behaves like a magnet while current flows. When the current stops, the magnetic effect disappears.
- The region around a current-carrying wire where magnetic influence is felt is called the magnetic field.
- This discovery was first made by Hans Christian Oersted, who showed that electricity and magnetism are closely related.
Electromagnets
An electromagnet is a temporary magnet produced by passing electric current through a coil of wire. When current flows, the coil behaves like a magnet; when current stops, it loses magnetism.
Electromagnets are stronger when an iron core (such as an iron nail) is placed inside the coil. Compass needle near (a) End A; (b) End B of an electromagnet
Compass needle near (a) End A; (b) End B of an electromagnet
Key characteristics of electromagnets:
- They work only when electric current flows
- They have two poles, North and South
- Their strength can be controlled
- They can be switched on and off
Factors Affecting Strength of an Electromagnet
- Amount of electric current flowing through the coil
- Number of turns of the coil
- Presence of an iron core
- Direction of current (reverses the poles)
Increasing current or number of turns increases the strength of the electromagnet.
Applications of Electromagnets
Electromagnets are widely used because they can be controlled easily.
Common uses include:
- Electric bells
- Electric motors
- Loudspeakers
- Cranes for lifting heavy metal objects
- Magnetic sorting in factories
Lifting electromagnets used in cranes pick up iron and steel objects when current is switched on and release them when switched off.
Heating Effect of Electric Current
When electric current flows through a conductor, the conductor becomes hot. This is known as the heating effect of electric current.
Heat is produced because the conductor offers resistance to the flow of current. Electrical energy is converted into heat energy.
Wires with higher resistance, such as nichrome wire, produce more heat than low-resistance wires like copper.
The amount of heat produced depends on:
- Magnitude of electric current
- Material of the conductor
- Length and thickness of the wire
- Duration for which current flows
Applications of Heating Effect
The heating effect is useful in many household and industrial devices.
Examples of appliances that work on this principle:
- Electric heaters
- Electric irons
- Electric stoves
- Kettles
- Immersion rods
- Hair dryers
- Incandescent bulbs
These devices contain a heating element, usually made of nichrome, which becomes red hot when current passes through it.
Hazards of Heating Effect
Uncontrolled heating can be dangerous.
Possible problems include:
- Energy loss in transmission wires
- Overheating of appliances
- Melting of plugs and sockets
- Electrical fires
Therefore, safety devices and proper wiring are essential in electrical circuits.
Production of Electricity in Cells and Batteries
Electric cells and batteries produce electricity through chemical reactions. They convert chemical energy into electrical energy.
Voltaic (Galvanic) Cell
A Voltaic cell consists of:
- Two different metal electrodes
- An electrolyte (liquid such as acid or salt solution)
- A container
A chemical reaction between the electrodes and electrolyte produces electric current. Over time, the chemicals are used up, and the cell stops working (becomes "dead").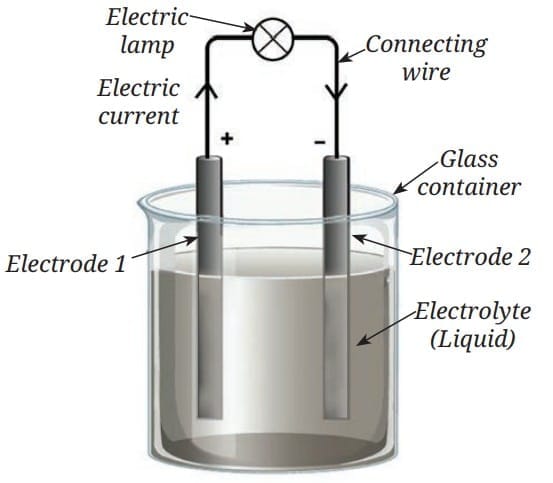 Simple representation of a Voltaic cell
Simple representation of a Voltaic cell
Simple cells can even be made using fruits such as lemons, where the fruit juice acts as the electrolyte. Electric cell made using lemons
Electric cell made using lemons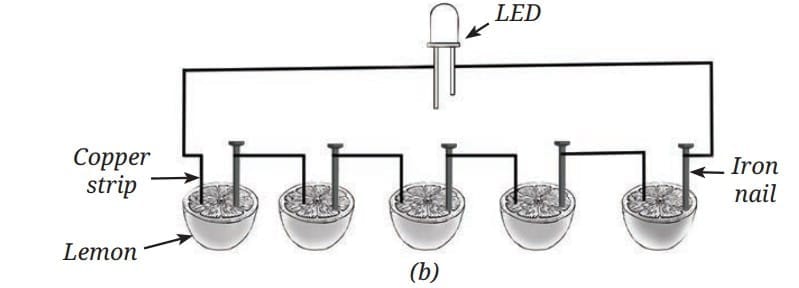 Connections in lemon cell
Connections in lemon cell
Dry Cells
Dry cells are widely used portable sources of electricity. They are called "dry" because the electrolyte is in the form of a moist paste rather than a liquid.
Main parts of a dry cell:
- Zinc container (negative terminal)
- Carbon rod with metal cap (positive terminal)
- Paste electrolyte
Dry cells are single-use cells and must be disposed of after use. (a) Dry cell; (b) Its internal structure
(a) Dry cell; (b) Its internal structure
Rechargeable Batteries
Rechargeable batteries can be recharged and reused multiple times. They are used in many modern devices, from small electronics to vehicles.
Advantages:
- Reduce waste
- Save money over time
- Provide long-term power supply
However, rechargeable batteries gradually lose efficiency after repeated use.
Lithium-ion batteries are the most common type used today.
Environmental Considerations
- Used batteries still contain harmful chemicals and metals. Improper disposal can cause environmental damage and fire hazards.
- Recycling batteries helps recover valuable materials and protects the environment.
FAQs on Rapid Revision: Magnetic And Heating Effects
| 1. What are the magnetic effects of electric current? |  |
| 2. How does the heating effect of electric current work? |  |
| 3. What is resistance, and how does it affect electric current? |  |
| 4. What are electric cells and batteries? |  |
| 5. How do magnetic and heating effects of electric current relate to each other? |  |






