NEET Exam > NEET Notes > Chemistry Class 11 > Cheat Sheet: Classification of Elements & Periodicity in Properties
Cheat Sheet: Classification of Elements & Periodicity in Properties
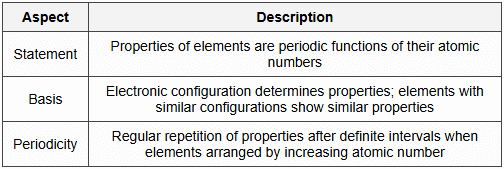
1. Modern Periodic Law
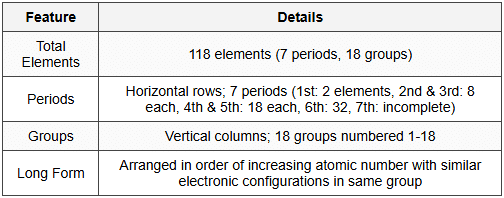
2. Present Form of Periodic Table
2.1 Structure
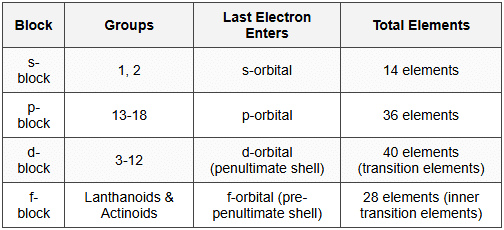
2.2 Block Classification
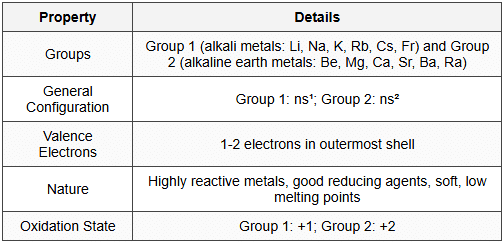
3. s-Block Elements
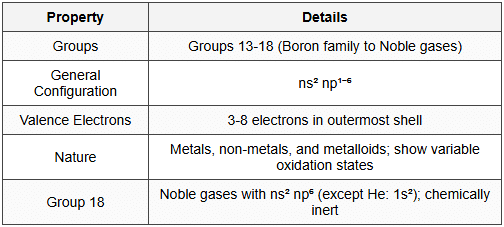
4. p-Block Elements
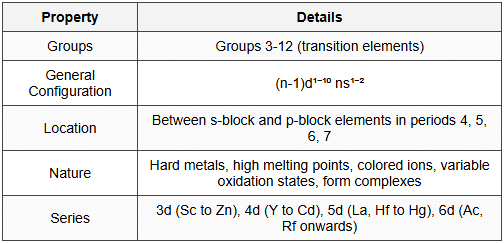
5. d-Block Elements
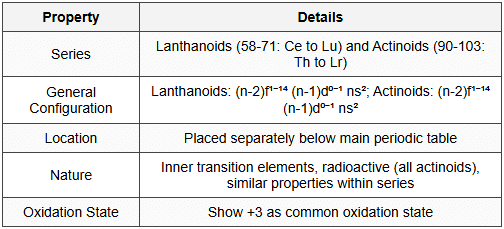
6. f-Block Elements
7. Atomic Radius
7.1 Definition and Types

7.2 Periodic Trends

8. Ionic Radius
8.1 Definition

8.2 Periodic Trends

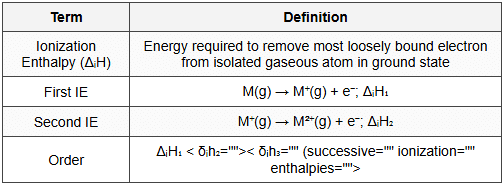
9. Ionization Enthalpy
9.1 Definition
9.2 Factors Affecting IE
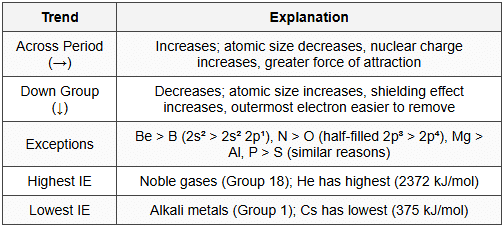
- Atomic size: Larger size, lower IE (easier to remove electron farther from nucleus)
- Nuclear charge: Higher nuclear charge, higher IE (stronger attraction)
- Shielding effect: Greater shielding, lower IE (inner electrons reduce nuclear attraction)
- Penetration effect: s > p > d > f; more penetration means higher IE
- Electronic configuration: Stable configurations (half-filled, fully-filled) have higher IE
9.3 Periodic Trends
10. Electron Gain Enthalpy
10.1 Definition

10.2 Factors Affecting EGE
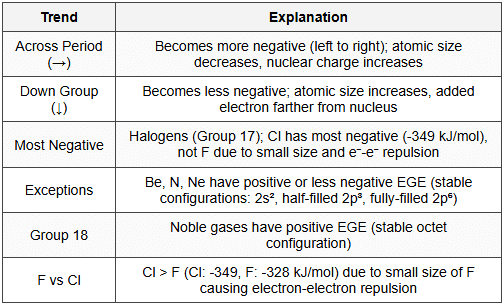
- Atomic size: Smaller atom, more negative EGE (electron closer to nucleus, stronger attraction)
- Nuclear charge: Higher nuclear charge, more negative EGE
- Electronic configuration: Stable configurations (half-filled, fully-filled) have less negative or positive EGE
10.3 Periodic Trends
11. Electronegativity
11.1 Definition
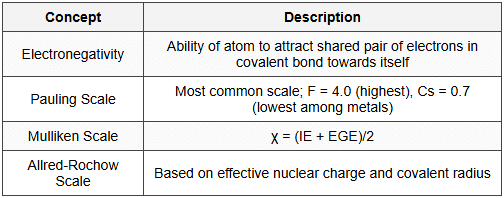
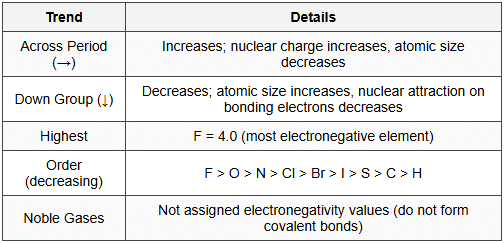
11.2 Periodic Trends
12. Valence and Oxidation States
12.1 Valence
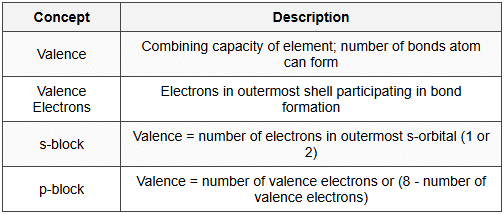
12.2 Oxidation States
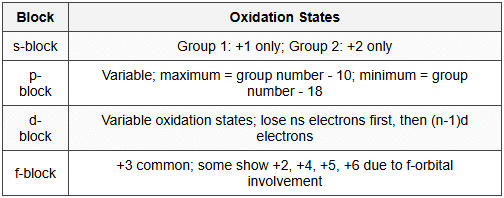
12.3 Periodic Trends in Oxidation States
- Across period: Maximum oxidation state increases up to group 17, then decreases
- Group 15-17: Show negative oxidation states (-3, -2, -1 respectively) in addition to positive states
- Inert pair effect: In heavier p-block elements, ns² electrons resist participation; lower oxidation states become stable (Tl⁺, Pb²⁺, Bi³⁺)
- Transition elements: Show multiple oxidation states due to close energy of ns and (n-1)d orbitals
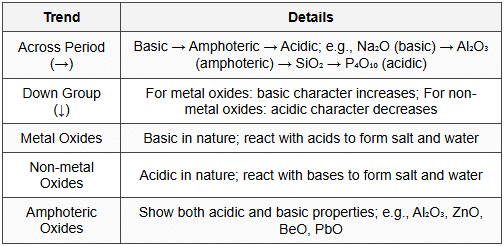
13. Chemical Reactivity
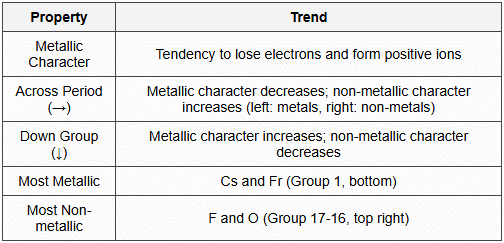
13.1 Metallic and Non-metallic Character
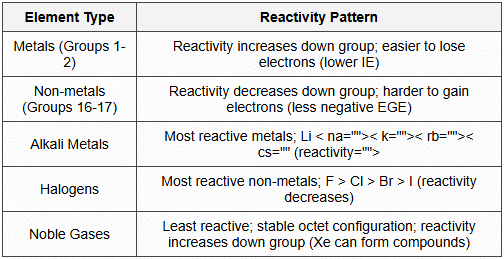
13.2 Reactivity Trends
13.3 Acidic and Basic Character of Oxides
14. Important Comparative Data
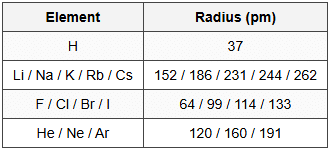
14.1 Atomic Radii (pm)
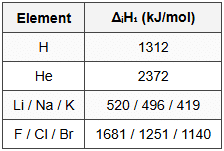
14.2 Ionization Enthalpy (kJ/mol)
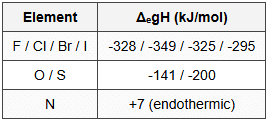
14.3 Electron Gain Enthalpy (kJ/mol)
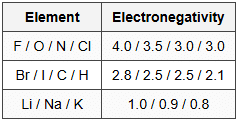
14.4 Electronegativity (Pauling Scale)
15. Key Anomalies and Special Cases
15.1 Exceptions in Periodic Trends
- IE: Be > B (stable 2s² vs 2s² 2p¹); N > O (stable half-filled 2p³ vs 2p⁴); Mg > Al; P > S
- EGE: Cl > F (electron repulsion in small F atom); Group 2 and 18 have positive values
- Atomic radius: Noble gases have largest radii in their periods (van der Waals radii measured)
- d-block contraction: Atomic radii of 5d series similar to 4d series due to lanthanoid contraction
15.2 Diagonal Relationship

15.3 Lanthanoid Contraction
- Steady decrease in atomic and ionic radii from La to Lu
- Caused by poor shielding of 4f electrons
- Results in similar radii of 4d and 5d series elements (e.g., Zr-Hf, Nb-Ta)
- Affects properties of 5d transition elements
15.4 Inert Pair Effect
- Reluctance of ns² electrons to participate in bonding in heavier p-block elements
- Stability of lower oxidation state increases down group
- Examples: Tl⁺ more stable than Tl³⁺; Pb²⁺ more stable than Pb⁴⁺; Bi³⁺ more stable than Bi⁵⁺
- Caused by poor shielding of d and f electrons, increasing effective nuclear charge on s electrons
The document Cheat Sheet: Classification of Elements & Periodicity in Properties is a part of the NEET Course Chemistry Class 11.
All you need of NEET at this link: NEET
Related Searches
Free, Exam, Important questions, past year papers, Extra Questions, practice quizzes, Summary, shortcuts and tricks, mock tests for examination, Cheat Sheet: Classification of Elements & Periodicity in Properties, Previous Year Questions with Solutions, Sample Paper, video lectures, pdf , study material, Semester Notes, Cheat Sheet: Classification of Elements & Periodicity in Properties, Viva Questions, Cheat Sheet: Classification of Elements & Periodicity in Properties, Objective type Questions, ppt, MCQs;






