CBSE Class 8 > Class 8 Notes > Science > Infographics: Matter in Our Surroundings
Infographics: Matter in Our Surroundings

The document Infographics: Matter in Our Surroundings is a part of the Class 8 Course Science Class 8.
All you need of Class 8 at this link: Class 8
FAQs on Infographics: Matter in Our Surroundings
| 1. What are the three states of matter? |  |
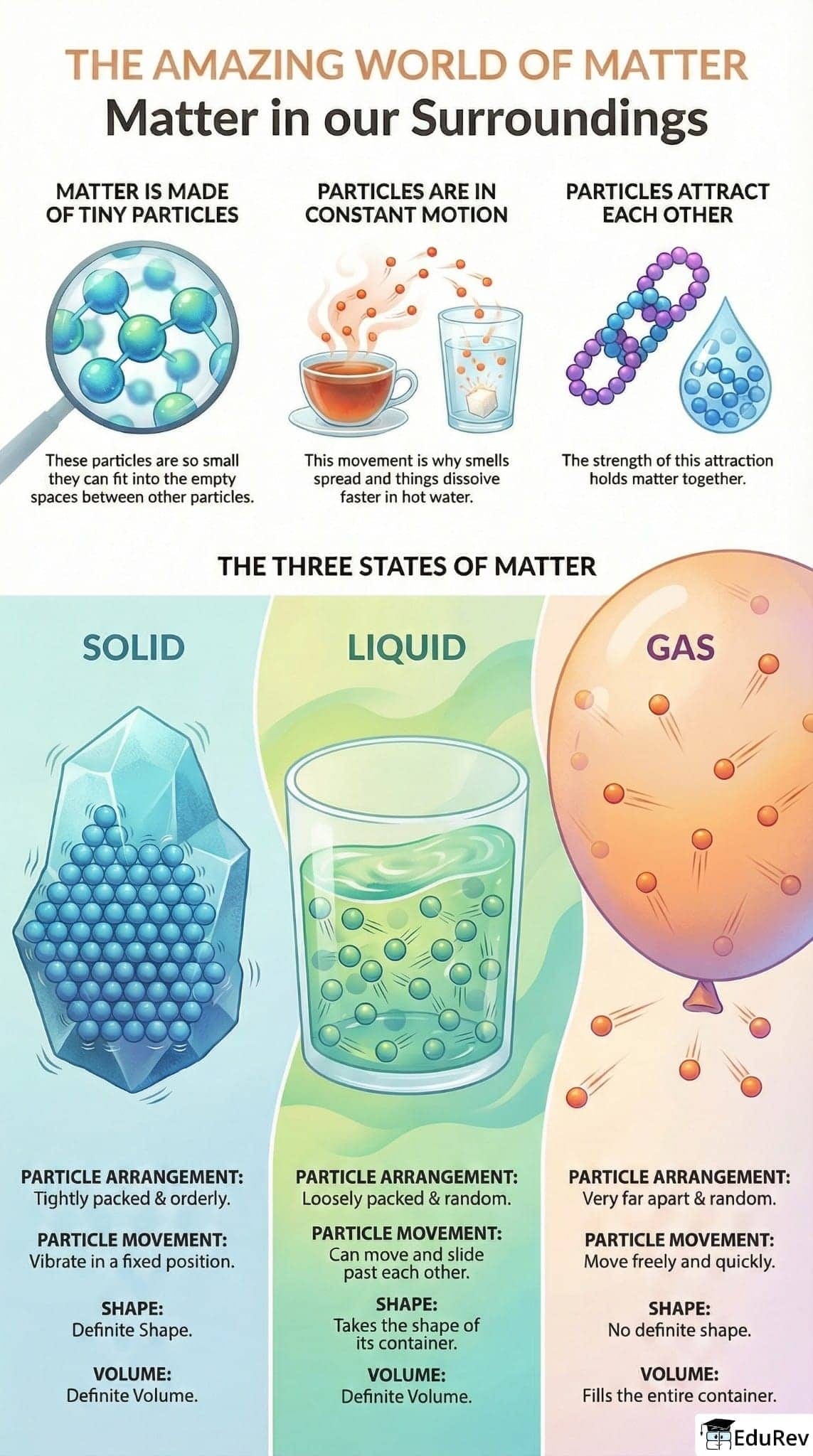
Ans. The three states of matter are solid, liquid, and gas. In solids, particles are closely packed together and vibrate in fixed positions, giving them a definite shape and volume. In liquids, particles are still close but can move past one another, allowing liquids to take the shape of their container while maintaining a definite volume. In gases, particles are far apart and move freely, resulting in no definite shape or volume.
| 2. How does temperature affect the state of matter? |  |
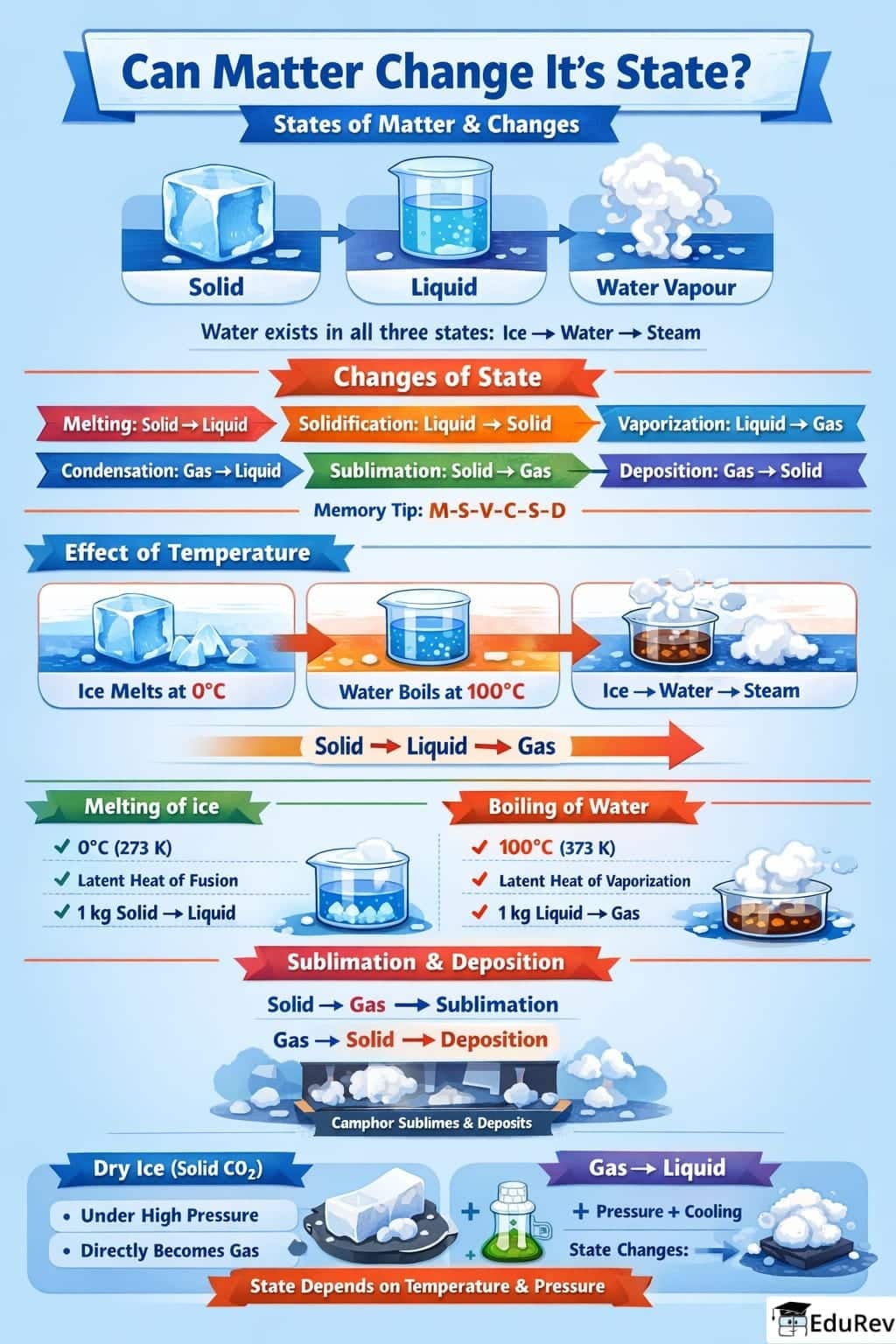
Ans. Temperature significantly affects the state of matter. When a substance is heated, the increase in temperature causes the particles to gain energy and move more vigorously. This can lead to a change in state, such as solid to liquid (melting) or liquid to gas (vapourisation). Conversely, cooling a substance can cause gas to turn into liquid (condensation) or liquid to solid (freezing).
| 3. What is the difference between a pure substance and a mixture? |  |
Ans. A pure substance consists of only one type of particle and has a uniform composition and distinct properties, such as boiling and melting points. Examples include elements like oxygen and compounds like water (H₂O). A mixture, on the other hand, contains two or more different substances that are physically combined. Mixtures can be homogeneous, where the components are evenly distributed, or heterogeneous, where the components remain distinct.
| 4. What is the role of intermolecular forces in determining the state of matter? |  |
Ans. Intermolecular forces are the forces of attraction or repulsion between molecules. These forces play a crucial role in determining the state of matter. In solids, strong intermolecular forces keep particles tightly packed, leading to a fixed shape. In liquids, these forces are weaker, allowing particles to slide past one another while maintaining volume. In gases, intermolecular forces are very weak, enabling particles to move freely and occupy the available space.
| 5. How can we separate the components of a mixture? |  |
Ans. The components of a mixture can be separated using various physical methods, depending on the properties of the substances involved. Techniques include filtration, where solids are separated from liquids, distillation, which separates liquids based on different boiling points, and chromatography, which separates substances based on their movement through a medium. Each method exploits specific physical characteristics of the components to achieve separation.
Related Searches
Semester Notes, shortcuts and tricks, Infographics: Matter in Our Surroundings, Objective type Questions, pdf , study material, Sample Paper, mock tests for examination, Infographics: Matter in Our Surroundings, Summary, ppt, Extra Questions, video lectures, past year papers, Important questions, Previous Year Questions with Solutions, MCQs, practice quizzes, Exam, Infographics: Matter in Our Surroundings, Viva Questions, Free;





