CBSE Class 8 > Class 8 Notes > Science > Infographics: Nature of Matter: Elements, Compounds, and Mixtures
Infographics: Nature of Matter: Elements, Compounds, and Mixtures
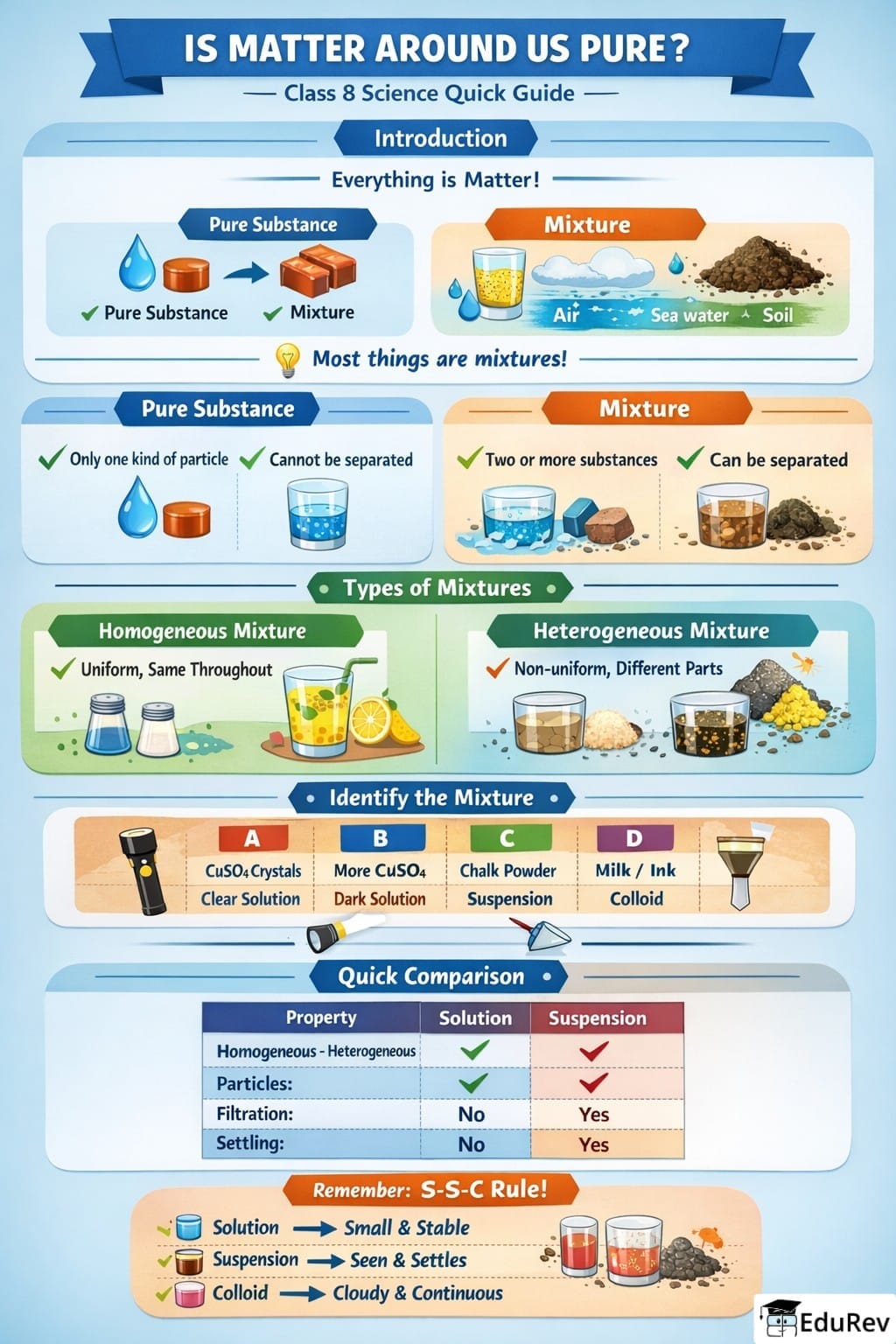

The document Infographics: Nature of Matter: Elements, Compounds, and Mixtures is a part of the Class 8 Course Science Class 8.
All you need of Class 8 at this link: Class 8
FAQs on Infographics: Nature of Matter: Elements, Compounds, and Mixtures
| 1. What are the basic building blocks of matter? |  |
Ans. The basic building blocks of matter are atoms. An atom is the smallest unit of an element and consists of a nucleus containing protons and neutrons, surrounded by electrons in various energy levels. Atoms combine to form molecules, which are the foundation of compounds and mixtures.
| 2. How do elements differ from compounds? |  |
Ans. Elements are pure substances that consist of only one type of atom, while compounds are substances formed when two or more different elements chemically combine in a fixed ratio. For example, oxygen (O) is an element, while water (H₂O) is a compound made up of hydrogen and oxygen atoms.
| 3. What is the difference between a homogeneous mixture and a heterogeneous mixture? |  |
Ans. A homogeneous mixture has a uniform composition throughout, meaning that its components are evenly distributed and not distinguishable, such as saltwater. In contrast, a heterogeneous mixture consists of visibly different substances or phases, such as a salad or sand and iron filings, where the individual components can be seen and separated easily.
| 4. Can mixtures be separated? If so, how? |  |
Ans. Yes, mixtures can be separated using physical methods based on differences in their physical properties. Common techniques include filtration (for separating solids from liquids), distillation (for separating liquids with different boiling points), and chromatography (for separating substances based on their movement through a medium).
| 5. Why are compounds considered to have different properties than their constituent elements? |  |
Ans. Compounds have different properties from their constituent elements because the chemical bonding and arrangement of atoms in a compound result in new characteristics. For example, sodium (Na) is a highly reactive metal and chlorine (Cl) is a poisonous gas, but when they combine to form sodium chloride (NaCl), commonly known as table salt, it is safe and essential for human consumption.
Related Searches
Compounds, Infographics: Nature of Matter: Elements, Infographics: Nature of Matter: Elements, MCQs, Viva Questions, Summary, past year papers, and Mixtures, Exam, ppt, Objective type Questions, Sample Paper, Compounds, pdf , and Mixtures, Important questions, study material, Semester Notes, Compounds, practice quizzes, Previous Year Questions with Solutions, mock tests for examination, Free, shortcuts and tricks, Extra Questions, video lectures, Infographics: Nature of Matter: Elements, and Mixtures;





