CBSE Class 8 > Class 8 Notes > Science Curiosity - New NCERT > Mnemonics : Nature of Matter: Elements, Compounds, and Mixtures
Mnemonics : Nature of Matter: Elements, Compounds, and Mixtures
Mixtures
1. Examples of Mixtures
In science, a mixture is formed when different substances are put together but do not chemically react. Each part keeps its own properties and can usually be separated.
Mnemonic: PALS
- P = Poha
- A = Air
- L = Lemonade
- S = Sprouts
2. Properties of Mixtures
Mixture properties are unchanged.Mnemonic: No Silly Rules.
- No= No fixed ratio
- Silly= Separable
- Rules= Retain properties
Types of Mixtures
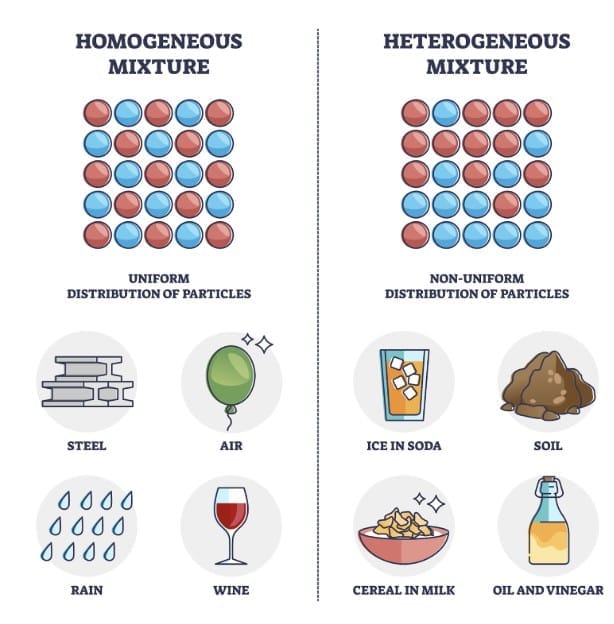
1. Non-Uniform Mixture ( Hetrogeneous) Examples
Different parts are visible.Mnemonic: POT
- P = Poha
- O = Oil & water
- T = Toys in box
2. Uniform Mixture ( Homogeneous) Examples
Looks same throughout.Mnemonic: SALT
- S = Sugar water
- A = Air
- L = Lemonade
- T = Tea
Alloys
1. Alloys Examples

Alloys are mixtures of metals.
What students need to learn:Common examples of alloys: Brass, Bronze, and Stainless steel
Mnemonic: SBB
- S = Stainless steel
- B = Brass
- B = Bronze
2. Stainless Steel Composition
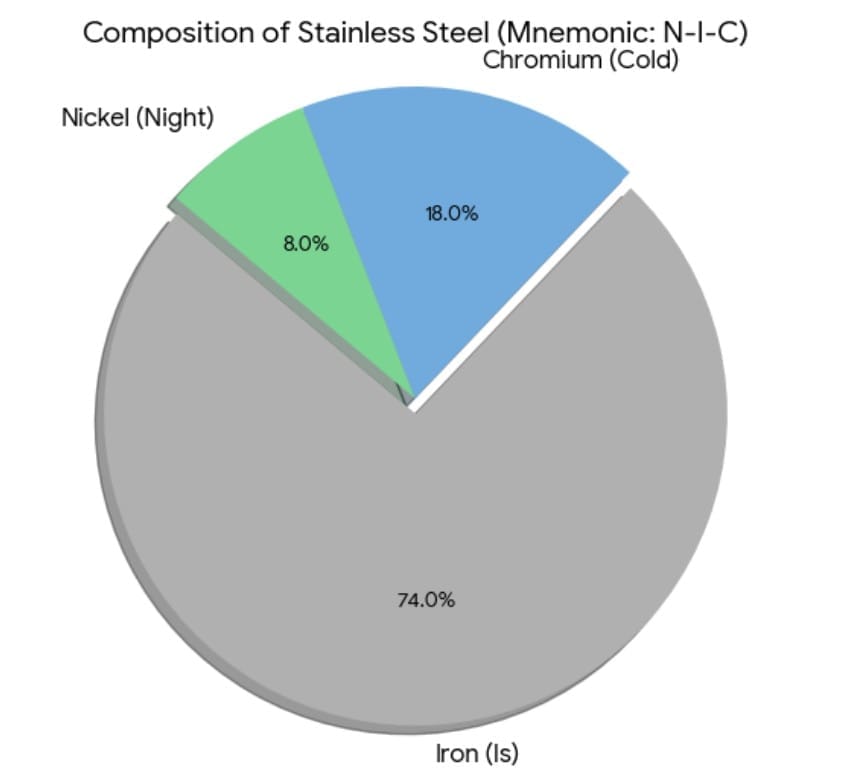
Stainless steel resists rusting.
Mnemonic: Night Is Cold.
- Night= Nickel
- Is= Iron
- Cold= Chromium
Adulteration
Adulteration reduces quality.
What students need to learn: Adulteration is the act of adding cheap substances to increase profit. This process lowers the nutritional value of food and can lead to health risks for the consumer.
Mnemonic: CHEAT
- C = Cheap addition
- H = Harmful
- E = Extra profit
- A = Adulteration
- T = Trust lost
The document Mnemonics : Nature of Matter: Elements, Compounds, and Mixtures is a part of the Class 8 Course Science Curiosity Class 8 - New NCERT.
All you need of Class 8 at this link: Class 8
FAQs on Mnemonics : Nature of Matter: Elements, Compounds, and Mixtures
| 1. What are mixtures and how do they differ from pure substances? |  |
Ans. Mixtures are combinations of two or more substances that retain their individual properties and can be separated by physical methods. In contrast, pure substances consist of only one type of particle and have a uniform composition, such as elements and compounds. Mixtures can be homogeneous (uniform composition) or heterogeneous (distinct components), while pure substances are always homogeneous.
| 2. What are the different types of mixtures? |  |
Ans. Mixtures are classified into two main types: homogeneous mixtures and heterogeneous mixtures. Homogeneous mixtures, also known as solutions, have a uniform composition throughout, such as salt water. Heterogeneous mixtures consist of visibly different substances or phases, such as a salad or a mixture of sand and iron filings. Each type of mixture behaves differently when it comes to separation and interaction of components.
| 3. What are alloys and how are they formed? |  |
Ans. Alloys are homogeneous mixtures of two or more metals, or a metal and a non-metal, which are combined to enhance certain properties like strength, ductility, or resistance to corrosion. Alloys are formed by melting the constituent metals together and allowing them to solidify, resulting in a material with characteristics that differ from the individual components. Common examples include steel (iron and carbon) and bronze (copper and tin).
| 4. What is purity in the context of mixtures, and how can it be assessed? |  |
Ans. Purity refers to the extent to which a substance is free from contaminants or other substances. In the context of mixtures, purity can be assessed through various methods including melting point determination, boiling point determination, and chromatography. A pure substance has a sharp melting or boiling point, while mixtures may have a range of values due to the presence of multiple components.
| 5. What is adulteration, and why is it significant in mixtures? |  |
Ans. Adulteration is the process of adding impurities or inferior substances to a pure substance, often to increase quantity or reduce production costs. This is significant in mixtures because it can compromise the quality, safety, and efficacy of products, especially in food and pharmaceuticals. Identifying and preventing adulteration is crucial for consumer protection and maintaining standards in various industries.
Related Searches
Important questions, Mnemonics : Nature of Matter: Elements, Extra Questions, Free, mock tests for examination, study material, and Mixtures, Mnemonics : Nature of Matter: Elements, Viva Questions, past year papers, Mnemonics : Nature of Matter: Elements, Compounds, practice quizzes, MCQs, video lectures, Exam, Previous Year Questions with Solutions, and Mixtures, shortcuts and tricks, pdf , ppt, Compounds, Summary, Compounds, and Mixtures, Semester Notes, Sample Paper, Objective type Questions;





