NEET Exam > NEET Notes > Short Notes > Short Notes: Biomolecules
Short Notes: Biomolecules
Chemical Composition of Living Cells
- Elements in living organisms: C, H, O, N, S, P (major) trace elements (Mg, Ca, Na, K, Fe, Cu, Zn, etc.)
- Biomolecules: Organic and inorganic compounds found in living organisms
- Inorganic compounds: Water, minerals
- Organic compounds: Carbohydrates, Lipids, Proteins, Nucleic acids, Vitamins, Enzymes
Analysis of Chemical Composition
- Acid-soluble pool: Biomolecules with molecular weight < 1000 Da (small molecules)
- Examples: Amino acids, monosaccharides, nucleotides, ions
- Acid-insoluble pool: Biomolecules with molecular weight > 10,000 Da (macromolecules)
- Examples: Polysaccharides, proteins, nucleic acids, lipids
- Lipids: Exception - small molecular weight but present in acid-insoluble fraction (membrane association)
Carbohydrates
- General formula: Cn(H2O)n or (CH2O)n
- Also called: Saccharides or Sugars
- Composition: Carbon, Hydrogen, Oxygen (ratio H:O = 2:1)
Classification of Carbohydrates
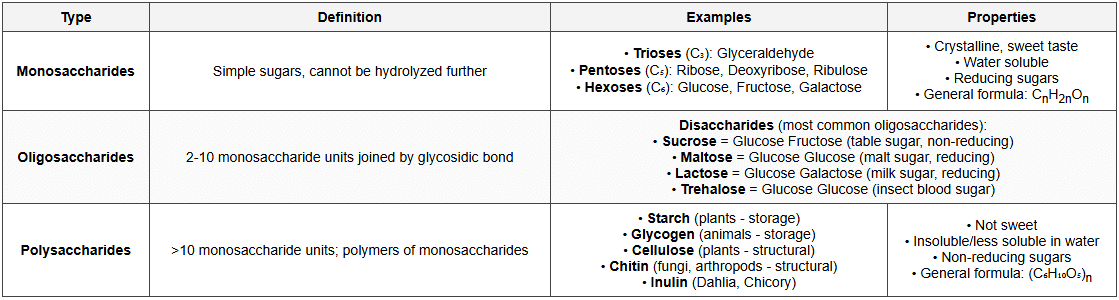
Important Polysaccharides
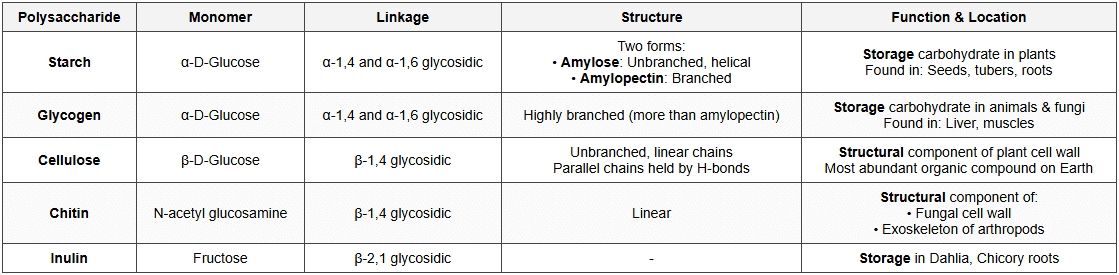
Functions of Carbohydrates
- Energy source: Primary source of energy (4 kcal/g)
- Energy storage: Starch (plants), Glycogen (animals)
- Structural role: Cellulose (cell wall), Chitin (exoskeleton)
- Component of nucleotides: Ribose and deoxyribose in RNA and DNA
- Cell recognition: Glycoproteins and glycolipids on cell surface
Lipids
- Definition: Hydrophobic or amphipathic organic molecules
- Solubility: Insoluble in water, soluble in organic solvents (chloroform, ether, benzene)
- Not true polymers
- Composition: C, H, O (some also have N, P, S)
Classification of Lipids
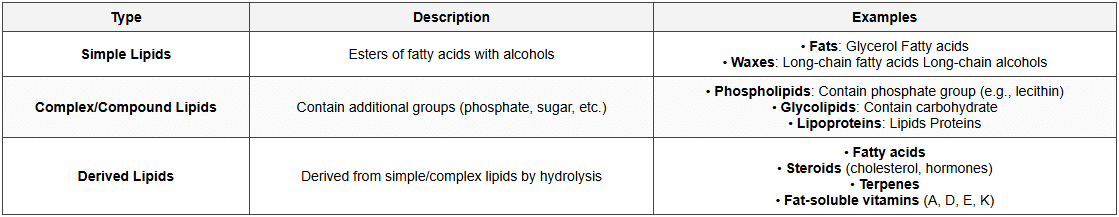
Fatty Acids
- Structure: Carboxylic acid with long hydrocarbon chain (R-COOH)
- Classification:
- Saturated fatty acids: No double bonds (single bonds only)
- Examples: Palmitic acid (C₁₆), Stearic acid (C₁₈)
- Solid at room temperature
- Found in: Animal fats, butter, ghee
- Unsaturated fatty acids: One or more double bonds (C=C)
- Monounsaturated: One double bond (e.g., Oleic acid)
- Polyunsaturated: Multiple double bonds (e.g., Linoleic acid, Linolenic acid)
- Liquid at room temperature (oils)
- Found in: Plant oils, fish oil
- Saturated fatty acids: No double bonds (single bonds only)
- Essential fatty acids: Cannot be synthesized by body, must be obtained from diet
- Examples: Linoleic acid, Linolenic acid, Arachidonic acid
Fats and Oils (Triglycerides)
- Structure: Glycerol (trihydric alcohol) esterified with 3 fatty acids
- Also called: Triacylglycerols
- Fats: Solid at room temperature (saturated fatty acids); animal origin
- Oils: Liquid at room temperature (unsaturated fatty acids); plant origin
- Energy value: 9 kcal/g (highest among biomolecules)
Phospholipids
- Structure: Glycerol 2 Fatty acids Phosphate group ( nitrogen base)
- Amphipathic: Hydrophilic head (phosphate) Hydrophobic tail (fatty acids)
- Examples: Lecithin (phosphatidylcholine), Cephalin
- Function: Major component of cell membranes (lipid bilayer)
Functions of Lipids
- Energy storage: Fats and oils (more efficient than carbohydrates)
- Structural component: Phospholipids in cell membranes
- Insulation: Subcutaneous fat (thermal insulation)
- Protection: Cushioning of organs
- Hormones: Steroid hormones (testosterone, estrogen, cortisol)
- Vitamins: Fat-soluble vitamins (A, D, E, K)
- Waterproofing: Waxes on leaves, fruits, feathers
Proteins
- Most abundant organic compound in cells (10-15% of cell mass)
- Term coined by: Berzelius (means "of first importance")
- Polymers of: Amino acids
- Linkage: Peptide bond (between -COOH of one amino acid and -NH₂ of another)
- Composition: C, H, O, N (always) S (in some)
Amino Acids
- General structure: H₂N-CHR-COOH
- Amino group (-NH₂): Basic
- Carboxyl group (-COOH): Acidic
- R group (side chain): Variable; determines properties
- α-carbon: Central carbon atom
- Amphoteric nature: Can act as both acid and base (zwitterion)
- Number: 20 standard amino acids in proteins
- Essential amino acids: Cannot be synthesized by body (9 in humans)
- Mnemonic: PVT TIM HALL
- Phenylalanine, Valine, Threonine, Tryptophan, Isoleucine, Methionine, Histidine, Arginine, Leucine, Lysine
Classification of Amino Acids (by R group)
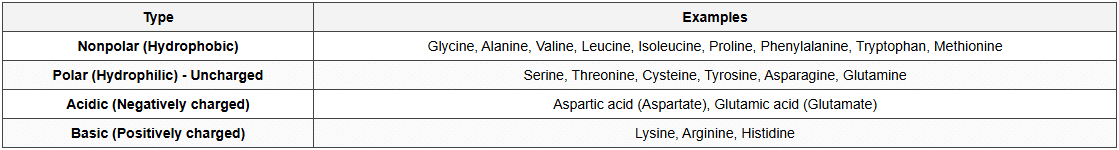
Structure of Proteins
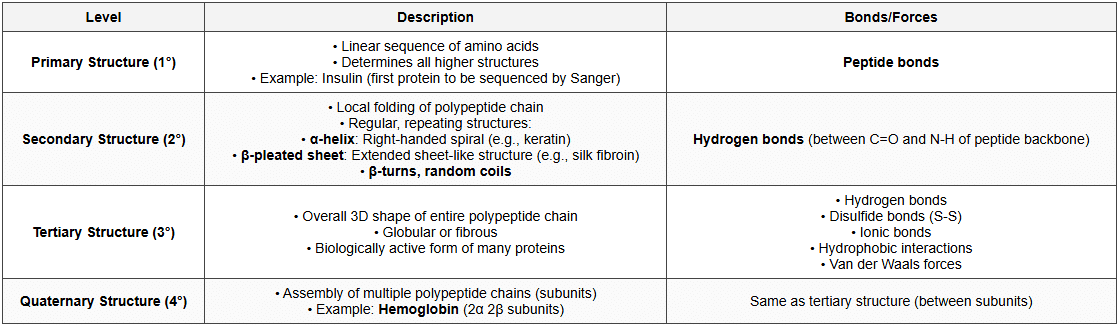
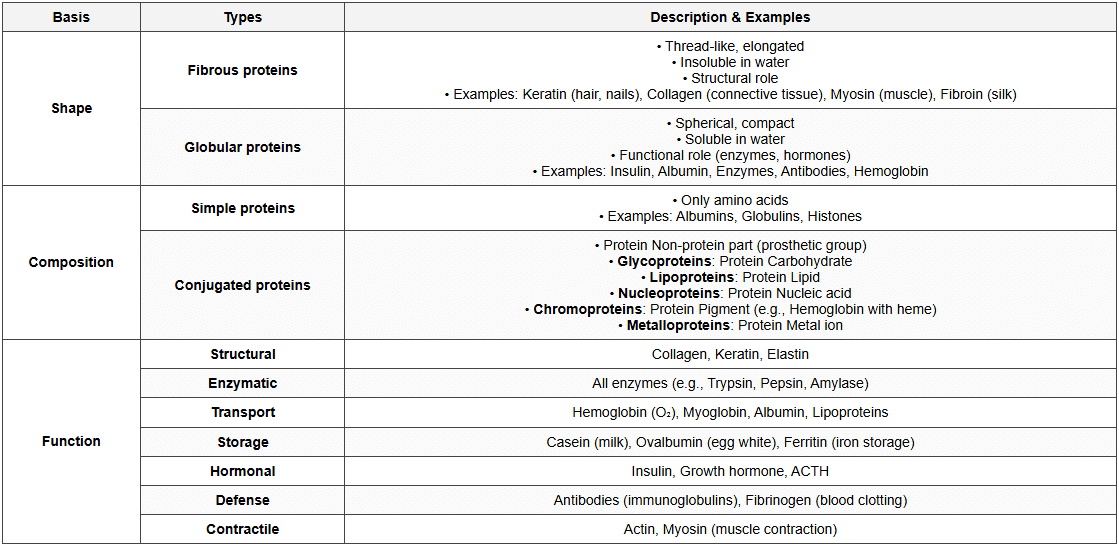
Classification of Proteins
Protein Denaturation
- Definition: Loss of protein's native structure (2°, 3°, 4°) without breaking peptide bonds
- Causes: Heat, pH changes, chemicals, heavy metals, organic solvents
- Result: Loss of biological activity
- May be: Reversible (renaturation) or irreversible
Functions of Proteins
- Structural: Cell structure, tissues (collagen, keratin)
- Enzymatic: Catalyze biochemical reactions
- Transport: Oxygen (hemoglobin), nutrients
- Defense: Antibodies, blood clotting
- Regulatory: Hormones (insulin, growth hormone)
- Movement: Muscle contraction (actin, myosin)
- Storage: Storage of amino acids and minerals
Nucleic Acids
- Discovered by: Friedrich Miescher (1869) - called it "nuclein"
- Polymers of: Nucleotides
- Types: DNA (Deoxyribonucleic acid) and RNA (Ribonucleic acid)
- Function: Storage and expression of genetic information
Nucleotides
- Monomer of nucleic acids
- Components:
- Nitrogenous base:
- Purines (double ring): Adenine (A), Guanine (G)
- Pyrimidines (single ring): Cytosine (C), Thymine (T - only in DNA), Uracil (U - only in RNA)
- Pentose sugar:
- Ribose: In RNA (has -OH at 2' carbon)
- Deoxyribose: In DNA (has -H at 2' carbon)
- Phosphate group: PO₄³⁻
- Nitrogenous base:
- Nucleoside: Base Sugar (without phosphate)
- N-glycosidic bond between base and sugar
- Nucleotide: Base Sugar Phosphate
- Phosphate attached to 5' carbon of sugar
- Linkage between nucleotides: Phosphodiester bond
- Between 3'-OH of one sugar and 5'-phosphate of next sugar
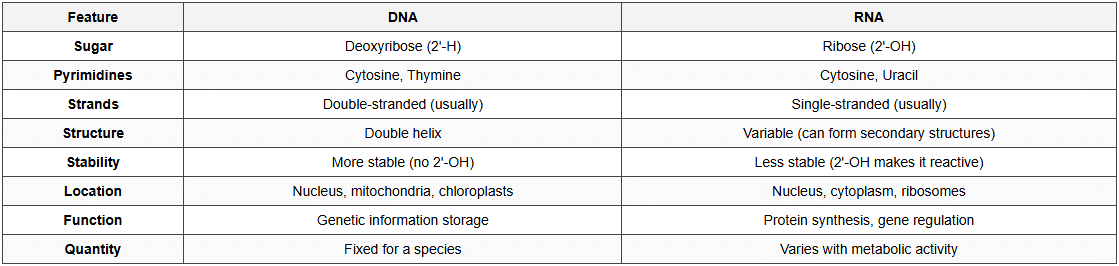
DNA (Deoxyribonucleic Acid)
- Watson & Crick Model (1953): Double helix structure
- Structure:
- Double helix: Two antiparallel polynucleotide chains
- Sugar-phosphate backbone: Outside (hydrophilic)
- Nitrogenous bases: Inside (hydrophobic), stacked
- Base pairing (Chargaff's rule):
- A pairs with T (2 hydrogen bonds)
- G pairs with C (3 hydrogen bonds)
- A G = T C (purines = pyrimidines)
- Antiparallel: One strand 5'→3', other strand 3'→5'
- Right-handed helix: B-DNA (most common form)
- Pitch: 3.4 nm (34 Å)
- Distance between base pairs: 0.34 nm
- Base pairs per turn: 10
- Diameter: 2 nm (20 Å)
- Major and minor grooves
- Functions:
- Storage of genetic information
- Replication (self-duplication)
- Transcription (RNA synthesis)
- Gene expression and regulation
- Heredity and variation
RNA (Ribonucleic Acid)
- Structure:
- Usually single-stranded (except in some viruses)
- Can form secondary structures by intramolecular base pairing (hairpin loops)
- Contains ribose sugar
- Contains Uracil instead of Thymine
- A pairs with U (in RNA)
Types of RNA
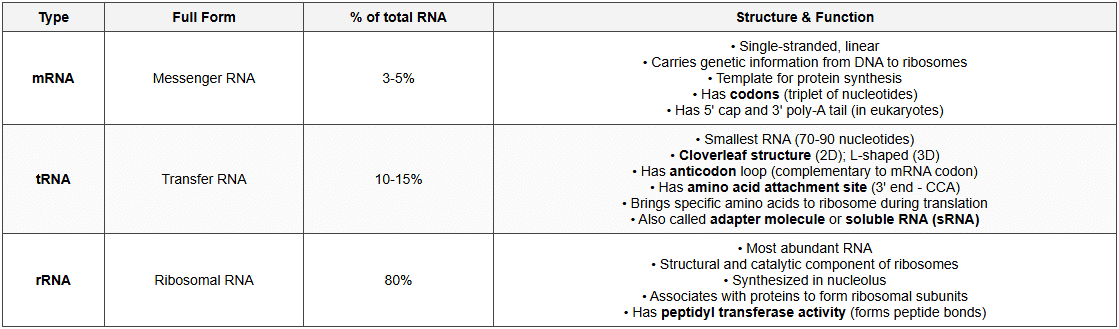
DNA vs RNA
Biology Class 11 NEET Revision - Cheat Sheet
ENZYMES
Definition and Properties
- Enzymes: Biological catalysts, mostly proteins (some RNA - ribozymes)
- Properties:
- Highly specific in action
- Increase rate of reaction without being consumed
- Lower activation energy
- Work at optimum temperature (37°C for human enzymes) and pH
- Denatured by high temperature and extreme pH
- Reversible in action
- Required in minute quantities
Enzyme Structure
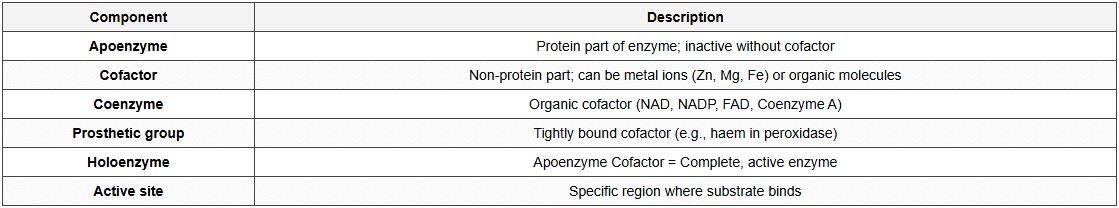
Enzyme Action - Mechanism
- Lock and Key Model(Fischer):
- Enzyme (lock) has specific shape
- Substrate (key) fits precisely into active site
- Rigid, complementary structures
- Induced Fit Model(Koshland):
- Active site is flexible
- Enzyme changes shape when substrate binds
- More widely accepted model
Enzyme Action - Steps
- Substrate binds to active site forming Enzyme-Substrate (ES) complex
- Enzyme catalyzes conversion of substrate to product
- Product is released
- Enzyme is free to bind another substrate molecule
Classification of Enzymes (IUB System)

Nomenclature of Enzymes
- Common names: Usually end with -asesuffix added to substrate name
- Example: Urease (acts on urea), Sucrase (acts on sucrose)
- Systematic names: Based on reaction type and substrate
- Each enzyme has an EC number (Enzyme Commission number) - 4 digit classification code
Factors Affecting Enzyme Activity
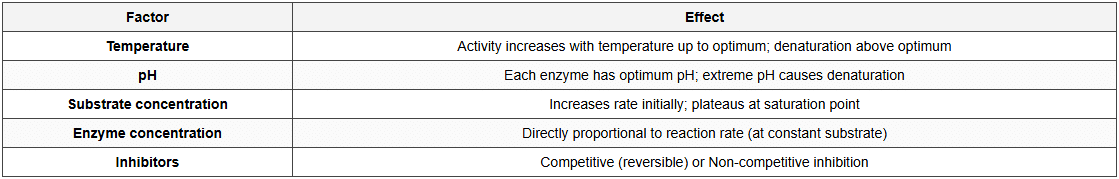
The document Short Notes: Biomolecules is a part of the NEET Course Short Notes for NEET.
All you need of NEET at this link: NEET
FAQs on Short Notes: Biomolecules
| 1. What are the main types of biomolecules? |  |
Ans. The main types of biomolecules include carbohydrates, proteins, lipids, and nucleic acids. Each of these biomolecules plays a crucial role in biological processes. Carbohydrates provide energy, proteins serve as building blocks and enzymes, lipids store energy and form cell membranes, while nucleic acids such as DNA and RNA are essential for genetic information and protein synthesis.
| 2. How do enzymes function as biological catalysts? |  |
Ans. Enzymes function as biological catalysts by lowering the activation energy required for a chemical reaction to occur. They bind to specific substrates at their active sites, forming an enzyme-substrate complex, which facilitates the conversion of substrates into products more efficiently, thereby speeding up metabolic reactions without being consumed in the process.
| 3. What is the structure and function of DNA? |  |
Ans. DNA, or deoxyribonucleic acid, is a double helix structure composed of two strands of nucleotides, which include a sugar, a phosphate group, and nitrogenous bases (adenine, thymine, cytosine, and guanine). Its primary function is to store and transmit genetic information, guiding the synthesis of proteins and ensuring the inheritance of traits in organisms.
| 4. What are the roles of lipids in biological systems? |  |
Ans. Lipids serve several vital roles in biological systems, including energy storage, forming cell membranes, and acting as signalling molecules. They are hydrophobic molecules that can store energy in the form of triglycerides, form the lipid bilayer of cell membranes, and include hormones such as steroids that regulate various physiological processes.
| 5. How do proteins achieve their functional shape? |  |
Ans. Proteins achieve their functional shape through a process called folding, which occurs during and after the synthesis of the polypeptide chain. The sequence of amino acids determines the protein's primary structure, and various interactions such as hydrogen bonds, ionic bonds, and hydrophobic interactions lead to the formation of secondary, tertiary, and sometimes quaternary structures, crucial for their specific functions in the body.
Related Searches
Sample Paper, Short Notes: Biomolecules, practice quizzes, Short Notes: Biomolecules, study material, Previous Year Questions with Solutions, pdf , shortcuts and tricks, Short Notes: Biomolecules, MCQs, Exam, Extra Questions, Free, video lectures, past year papers, Semester Notes, ppt, Objective type Questions, Important questions, Summary, mock tests for examination, Viva Questions;





