Short Notes: Equilibrium
6.1 Chemical Equilibrium
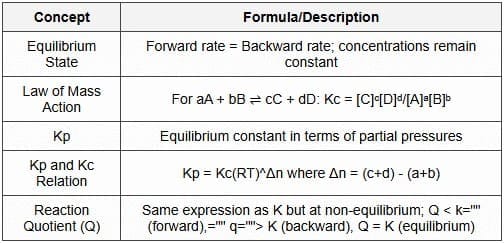
Chemical equilibrium is the state in a closed system where the macroscopic properties (concentrations, pressure, temperature) remain constant with time because the forward and reverse reaction rates are equal. At equilibrium the reaction is dynamic - reactant is converted to product and product to reactant at the same rate.
Law of mass action and equilibrium constants
For a general homogeneous reaction
the equilibrium constant in terms of molar concentrations is given by
For gaseous systems the equilibrium constant in terms of partial pressures is
The relation between Kp and Kc is
where Δn = (c + d) - (a + b), R is the gas constant and T the temperature in kelvin.
Key characteristics
- Reaction quotient (Q): calculated using the same expression as K but with instantaneous concentrations; if Q < K the reaction proceeds forward, if Q > K it proceeds backward, if Q = K the system is at equilibrium.
- Units: Equilibrium constants are ideally dimensionless (activity basis). In practice Kc or Kp may be quoted with units; careful unit handling is required in calculations.
- Heterogeneous equilibria: Concentrations of pure solids and pure liquids are constant and are therefore omitted from the equilibrium expression.
- Temperature dependence: Only temperature changes K (see Le Chatelier and van't Hoff relation); pressure, concentration and catalyst do not change the numerical value of K (except via temperature change).
Example (illustrative)
For the Haber process
the expression for Kc is
6.2 Le Chatelier's Principle
Le Chatelier's principle states: when a system at equilibrium is subjected to a stress (change in concentration, pressure, volume or temperature), the system shifts in the direction that tends to counteract the stress and restore a new equilibrium.
- Concentration change: Adding a reactant or removing a product shifts the equilibrium in the forward direction; adding a product or removing a reactant shifts it in the reverse direction. Example: adding H2 to the Haber equilibrium shifts toward NH3.
- Pressure / volume (gaseous systems): Increasing pressure (decreasing volume) favours the side with fewer moles of gas; decreasing pressure (increasing volume) favours the side with more moles of gas. Example: for N2 + 3H2 ⇌ 2NH3, increasing pressure shifts equilibrium right because 4 moles → 2 moles.
- Temperature: Treat temperature as heat. For an endothermic reaction heat is a reactant; increasing temperature favours the endothermic direction and increases K. For an exothermic reaction heat is a product; increasing temperature favours the reverse direction and decreases K.
- Catalyst: A catalyst increases the rate of both forward and reverse reactions equally, therefore it lowers the time to reach equilibrium but does not change the equilibrium position or the value of K.
- Inert gas addition: At constant volume, adding an inert gas does not change partial pressures and therefore has no effect on equilibrium. At constant pressure it can change partial pressures and may shift equilibrium.
- Limitations: Le Chatelier's principle predicts the direction of shift qualitatively but does not provide the new equilibrium concentrations or the magnitude of shift; for quantitative changes use equilibrium constant expressions.
6.3 Ionic Equilibrium

Ionic equilibrium concerns equilibria involving ions in aqueous solution - acid-base equilibria, salt hydrolysis, precipitation and solubility, and the ionisation of water.
Autoionisation of water and pH scale
Water undergoes autoionisation:
At 25 °C the ionic product of water is
pH is defined as
and pOH = -log[OH-], with pH + pOH = pKw (≈ 14.00 at 25 °C).
Acids and bases
- Strong acids/bases: completely ionise in water (for example HCl, NaOH).
- Weak acids/bases: partially ionise. Acid dissociation constant:
- For a weak base B: B + H2O ⇌ BH+ + OH- and Kb = [BH+][OH-]/[B].
- Relation for conjugate pairs: pKa + pKb = pKw.
- Ostwald's dilution law (approximate, for a weak monoprotic acid):
where α is degree of dissociation and c the analytical concentration (valid when α ≪ 1).
Common ion effect and salt hydrolysis
- Common ion effect: Presence of an ion common to an equilibrium suppresses the ionisation of a weak electrolyte (shifts equilibrium toward the undissociated form). Example: addition of acetate ion to acetic acid reduces its ionisation.
- Salt hydrolysis: Salts can alter pH of their aqueous solutions depending on the strengths of the parent acid and base. Examples:
- Salt of strong acid + strong base → neutral (pH ≈ 7).
- Salt of weak acid + strong base → basic solution (anions hydrolyse producing OH-).
- Salt of weak base + strong acid → acidic solution (cations hydrolyse producing H+).
- Hydrolysis constants relate to Ka and Kb. For an anion A- (conjugate base of weak acid):
6.4 Buffer Solutions
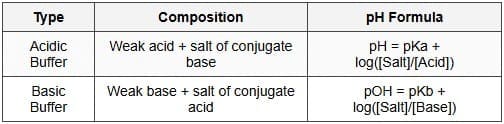
Buffer solutions resist large changes in pH when small amounts of strong acid or base are added. A typical buffer contains a weak acid and its conjugate base, or a weak base and its conjugate acid.
Henderson-Hasselbalch equation
For a buffer composed of a weak acid HA and its conjugate base A-, the pH is given by:
Similarly, for a weak base buffer:
Buffer capacity and range
- Buffer range: Effective pH control is roughly within pKa ± 1.
- Buffer capacity: The amount of strong acid/base the buffer can neutralise without significant pH change; it increases with total concentration of buffer components and is greatest when [HA] ≈ [A-].
- Effect of dilution: Dilution reduces buffer capacity but has little effect on pH when the ratio [A-]/[HA] remains unchanged.
Example (preparation and use)
- Acetate buffer: mixture of acetic acid and sodium acetate. If [A-] = [HA], pH = pKa (for acetic acid pKa ≈ 4.76 at 25 °C).
- Physiological example: bicarbonate buffer system in blood (H2CO3 / HCO3-) helps maintain blood pH ≈ 7.4.
6.5 Solubility Equilibrium
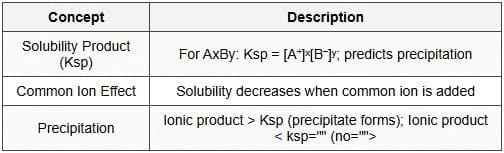
Solubility equilibrium describes the saturation of a sparingly soluble ionic compound in solvent (usually water). The solid is in equilibrium with its dissociated ions in solution.
Solubility product (Ksp)
For a salt that dissociates as
the solubility product is
If the molar solubility of MX is s, then
For salts with other stoichiometries, express Ksp accordingly. Examples:
Common ion effect and precipitation
- Common ion effect: Addition of an ion common to the solubility equilibrium reduces solubility. Example: addition of Cl- reduces solubility of AgCl.
- Precipitation criterion: Compare reaction quotient Qsp with Ksp. If Qsp > Ksp precipitation occurs; if Qsp < Ksp more solid dissolves; if Qsp = Ksp the solution is saturated.
- Selective precipitation: Based on different Ksp values, ions can be separated selectively by controlling concentrations and pH.
Effect of pH, complexation and other equilibria
- pH effect: Solubility of salts containing basic anions increases in acidic medium because the anion is protonated (for example, carbonate salts become more soluble in acid).
- Complex ion formation: Complexation can greatly increase solubility. Example: AgCl(s) dissolves in presence of excess NH3 because Ag+ forms the complex [Ag(NH3)2]+ with formation constant Kf. Combined equilibria reduce free [Ag+] and shift dissolution to the right.
- Combined equilibrium calculation: To find total solubility when complexation occurs, use Ksp together with Kf and mass-balance relationships.
Short worked illustration (conceptual)
To decide if AgCl will precipitate when solutions of AgNO3 and NaCl are mixed, compute Qsp = [Ag+][Cl-] from initial concentrations. Compare Qsp with Ksp(AgCl). If Qsp > Ksp, AgCl precipitates.
Final note: For quantitative problems always write balanced chemical equations, identify the equilibrium expressions, substitute known concentrations or solubilities, and solve algebraically, applying approximations (such as neglecting small x) only when justified. Remember temperature dependence of equilibrium constants and use appropriate values (for example K_w = 1.0×10-14 at 25 °C).
FAQs on Short Notes: Equilibrium
| 1. What is equilibrium in the context of chemical reactions? |  |
| 2. What factors can affect chemical equilibrium? |  |
| 3. How is the equilibrium constant (K) defined? |  |
| 4. What is the significance of the equilibrium position? |  |
| 5. How do catalysts influence chemical equilibrium? |  |





