Short Notes: Hydrocarbons
9.1 Alkanes (\(C_nH_{2n+2}\))
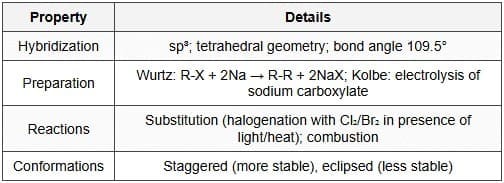
Alkanes are saturated acyclic hydrocarbons containing only single carbon-carbon bonds. Each carbon atom is sp³ hybridised, adopts a tetrahedral geometry and the ideal bond angle is 109.5°. They are also called paraffins.
- General formula: \(C_nH_{2n+2}\).
- Nomenclature: Use prefixes (meth-, eth-, prop-, but-) + suffix -ane. Number the longest chain to give lowest locants for substituents; name substituents as alkyl groups.
- Isomerism: Chain (constitutional) isomerism and positional isomerism of substituents are common; stereoisomerism is not found in simple saturated open-chain alkanes. Number of constitutional isomers increases rapidly with carbon number.
- Physical properties: Alkanes are non-polar, immiscible with water, and soluble in non-polar solvents. Boiling point increases with molecular mass; branching lowers boiling point (less surface area → weaker van der Waals forces).
- Sources and preparation: Major source is petroleum (fractional distillation and catalytic reforming). Laboratory methods include the Wurtz reaction (coupling of alkyl halides with sodium in dry ether) and reduction of alkyl halides using hydrogen with suitable catalysts.
- Chemical reactivity: Relatively inert compared with unsaturated hydrocarbons but undergo important reactions:
- Combustion: Complete combustion: \( \text{C}_x\text{H}_y + \left(x + \tfrac{y}{4}\right)\text{O}_2 \rightarrow x\text{CO}_2 + \tfrac{y}{2}\text{H}_2\text{O} \).
- Free-radical halogenation: \( \text{RH} + \text{X}_2 \xrightarrow{hv} \text{RX} + \text{HX} \) (initiation → propagation → termination). Tertiary hydrogen atoms are most readily substituted (tertiary radical most stable).
- Cracking: Thermal or catalytic cracking converts higher alkanes to smaller alkanes and alkenes.
- Conformation: Rotation about C-C single bonds leads to conformational isomerism; for example, staggered conformation of ethane is more stable than eclipsed. Newman projections are used to visualise conformations.
- Uses: Fuels (methane, propane, petrol), lubricants, feedstock for petrochemical industry.
9.2 Alkenes (\(C_nH_{2n}\))
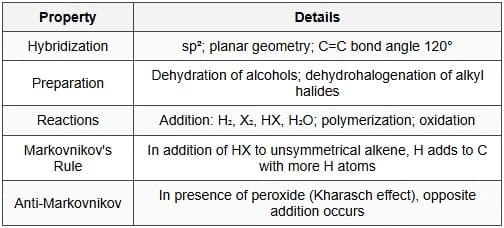
Alkenes are unsaturated hydrocarbons containing at least one carbon-carbon double bond. The carbons in the double bond are sp² hybridised and the geometry around each is trigonal planar with bond angles ≈ 120°. The π bond restricts rotation and gives rise to stereoisomerism.
- General formula: \(C_nH_{2n}\) for acyclic, mono-unsaturated alkenes.
- Nomenclature and stereochemistry: Number the chain to give the double bond the lowest possible number; indicate configuration using cis/trans or E/Z rules when required.
- Preparation: Dehydration of alcohols (acidic), dehydrohalogenation of alkyl halides (base, E2), and by cracking of higher hydrocarbons.
- Characteristic reactions (electrophilic addition):
- Hydrogenation: \( \text{RCH=CHR'} + \text{H}_2 \xrightarrow{\text{Pd/BaSO}_4,\text{H}_2\text{ or Lindlar}} \text{RCH}_2\text{CH}_2\text{R'} \).
- Halogenation: \( \text{RCH=CHR'} + \text{Br}_2 \rightarrow \) vicinal dibromide (anti addition).
- Hydrohalogenation: \( \text{RCH=CHR'} + \text{HBr} \rightarrow \) alkyl bromide (Markovnikov addition; carbocation intermediate can lead to rearrangements).
- Hydration: Acid-catalysed addition of water gives alcohols (Markovnikov); oxymercuration-demercuration avoids rearrangement; hydroboration-oxidation gives anti-Markovnikov alcohols.
- Oxidation: Permanganate oxidation (cold, dilute KMnO4) gives diols; ozonolysis (O3) cleaves double bond to give carbonyl compounds.
- Polymerisation: Alkenes undergo addition polymerisation (free-radical, cationic or coordination polymerisation) to give polymers such as polyethylene.
- Mechanistic notes: Electrophilic attack on the π bond forms a carbocation (or bridged halonium in halogenation) which is then attacked by nucleophile; regiochemistry explained by carbocation stability (Markovnikov's rule).
- Tests: Decolourisation of bromine in CCl4 and disappearance of purple KMnO4 (Baeyer test) indicate unsaturation.
- Applications: Manufacture of alcohols, plastics (polymers), and intermediates in organic synthesis.
9.3 Alkynes (\(C_nH_{2n-2}\))
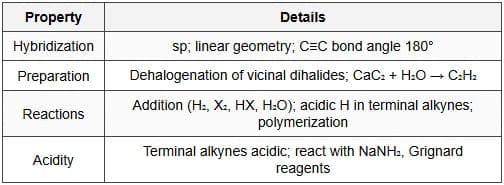
Alkynes contain a carbon-carbon triple bond. The carbons in the triple bond are sp hybridised, adopt a linear geometry and the bond angle is 180°. Terminal alkynes show acidic character (pKa ≈ 25) compared with alkenes and alkanes.
- General formula: \(C_nH_{2n-2}\) for acyclic mono-unsaturated alkynes.
- Preparation: Dehydrohalogenation of vicinal or geminal dihalides using strong base (e.g., NaNH2) to give alkynes; partial dehydrogenation of alkanes and cracking processes in industry.
- Reactions:
- Hydrogenation: Complete hydrogenation gives alkanes; partial hydrogenation with Lindlar catalyst gives cis-alkenes; dissolving metal reduction (Na/NH3) gives trans-alkenes.
- Addition of HX and X2: Follows Markovnikov rule for electrophilic additions; addition to produce dihalides.
- Hydration: Acid-catalysed hydration (HgSO4/H2SO4) of terminal alkynes yields methyl ketones (Markovnikov); hydroboration-oxidation yields aldehydes (anti-Markovnikov) for terminal alkynes.
- Formation of acetylide ions: Terminal alkynes react with strong bases (NaNH2, NaH) to form nucleophilic acetylide ions (RC≡C-) which undergo alkylation with primary alkyl halides.
- Oxidation: Strong oxidants convert alkynes into carboxylic acids (internal) or a mixture including carboxylic acids (terminal → carboxylate + CO2 under strong conditions).
- Uses: Alkynes are used as chemical intermediates and as fuels (acetylene in welding). Terminal alkynes are important building blocks in organic synthesis (C-C bond formation).
9.4 Aromatic Hydrocarbons
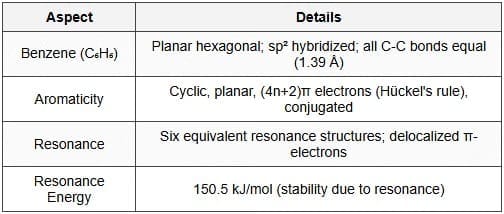
Aromatic hydrocarbons are cyclic, conjugated molecules that obey Hückel's rule of aromaticity: a planar cyclic conjugated system containing \(4n+2\) π electrons (n = 0, 1, 2 ...) is aromatic and particularly stable. The simplest aromatic hydrocarbon is benzene, formula \(C_6H_6\).
- Benzene structure: Six π electrons are delocalised over the ring giving equal C-C bond lengths (~1.39 Å) and extra stabilisation (aromatic stabilisation energy). Benzene resists addition reactions that would destroy aromaticity and prefers electrophilic aromatic substitution (EAS).
- Examples of aromatic systems: Benzene, naphthalene (fused rings), anthracene, phenol (aromatic ring with -OH substituent).
- Chemical behaviour: Aromatic rings undergo substitution reactions (nitration, sulfonation, halogenation, Friedel-Crafts alkylation/acylation) rather than addition under normal conditions.
- Criteria for aromaticity: Planarity, cyclic conjugation and \(4n+2\) π electrons are necessary for aromatic stabilisation.
- Physical properties: Generally non-polar, insoluble in water; many aromatic compounds have characteristic UV and IR absorptions and distinct chemical tests.
9.5 Electrophilic Substitution in Benzene
Electrophilic aromatic substitution (EAS) proceeds by two principal steps: formation of a non-aromatic σ-complex (arenium ion) by electrophilic attack, followed by deprotonation to restore aromaticity.
- General mechanism:
- Generation of a strong electrophile \(E^+\) (e.g., \( \text{NO}_2^+ \) from HNO3/H2SO4, \( \text{Br}^+ \) from Br2/FeBr3).
- Electrophile attacks the π system to form the σ-complex (arenium ion), which is resonance-stabilised but non-aromatic.
- Loss of a proton (base removes H+) from the σ-complex restores the aromatic system and gives the substituted benzene.
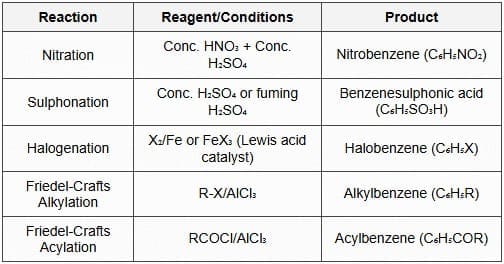
- Typical EAS reactions:
- Nitration: \( \text{C}_6\text{H}_6 + \text{HNO}_3 \xrightarrow{\text{H}_2\text{SO}_4} \text{C}_6\text{H}_5\text{NO}_2 + \text{H}_2\text{O} \). Electrophile: \( \text{NO}_2^+ \).
- Sulfonation: Reversible introduction of -SO3H using fuming sulphuric acid (SO3/H2SO4).
- Halogenation: \( \text{C}_6\text{H}_6 + \text{Br}_2 \xrightarrow{\text{FeBr}_3} \text{C}_6\text{H}_5\text{Br} + \text{HBr} \). Electrophile generated by Lewis acid.
- Friedel-Crafts alkylation/acylation: Alkylation uses R+ (from R-Cl/AlCl3) and acylation uses acylium ions (RCO+); acylation does not suffer from alkyl rearrangement and deactivates the ring (reducing further alkylation).
- Important mechanistic points: Stability of σ-complex and the ease of regeneration of aromaticity control reactivity. Strong deactivating groups (e.g., -NO2) can prevent EAS; highly activated rings require milder conditions.
9.6 Directive Influence
Substituents already present on a benzene ring influence both the rate of further electrophilic substitution and the position at which the new electrophile is introduced. This is described in terms of activation/deactivation and directive influence.
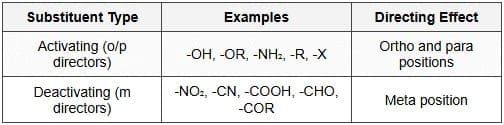
- Activating groups (ortho/para directors): Electron-donating groups that increase ring electron density and direct new electrophiles to the ortho and para positions. Examples: -OH, -OR, -NH2, -NHR, -CH3. Activation may be by resonance (-OH, -NH2) or by hyperconjugation (+I effect, e.g., -CH3).
- Deactivating groups (meta directors): Electron-withdrawing groups that decrease ring electron density and direct electrophiles to the meta position. Examples: -NO2, -CN, -SO3H, -COOR, -COR. These act mainly by -I or -R effects.
- Special case - halogens: Halogens (-F, -Cl, -Br, -I) are deactivating by induction (-I) but direct to ortho/para positions due to a lone-pair resonance contribution.
- Underlying reason: Directing effects arise from a balance of resonance donation/withdrawal and inductive effects; resonance donors stabilise the σ-complex at ortho/para positions, whereas strong withdrawers destabilise ortho/para σ-complexes and make meta substitution comparatively more favourable.
- Practical considerations: Protonation or other changes of substituents under strongly acidic reaction conditions can change their directing influence (for example, protonated -NH2 becomes -NH3+ which is strongly deactivating and meta directing).
- Steric effects: Bulky substituents often favour para substitution because ortho positions are sterically hindered.
Summary (optional): Hydrocarbons are classified by saturation and bonding: alkanes (single bonds, saturated), alkenes (double bonds, unsaturated) and alkynes (triple bonds, linear). Aromatic hydrocarbons (e.g., benzene) possess delocalised π systems and follow characteristic electrophilic substitution chemistry. Understanding structure, hybridisation, mechanism (especially electrophilic addition for unsaturated hydrocarbons and electrophilic substitution for aromatics) and directing effects is central for predicting products and reactivity in organic chemistry.
FAQs on Short Notes: Hydrocarbons
| 1. What are hydrocarbons? |  |
| 2. What are the different types of hydrocarbons? |  |
| 3. How are alkanes and alkenes distinguished? |  |
| 4. What is the significance of hydrocarbons in daily life? |  |
| 5. What is the process of cracking in relation to hydrocarbons? |  |






