CBSE Previous Year Questions: Biomolecules
CBSE Previous Year Questions 2025
Multiple Choice Type Questions
(A) It is an aldohexose.
(B) On heating with HI it forms n-hexane.
(C) It exists in furanose form.
(D) It does not give Schiff's test.
Ans: (C) It exists in furanose form.
Sol: Glucose is an aldohexose. It reacts with HI to form n-hexane, confirming its six-carbon straight chain structure. While it does not give the Schiff's test despite having an aldehyde group, glucose primarily exists in the pyranose form (a six-membered ring), not the furanose form (a five-membered ring), which is more characteristic of fructose.
Q2: Pyranose ring of glucose is formed due to the reaction between: [1 Mark]
(A) C₁ and C₃
(B) C₁ and C₅
(C) C₁ and C₄
(D) C₁ and C₂
Ans: (B) C₁ and C₅
Sol: The pyranose (six-membered ring) form of glucose is formed by intramolecular hemiacetal formation between the -CHO at C₁ and -OH at C₅.
Q3: Furanose ring of fructose is formed due to reaction between: [1 Mark]
A) C\(_1\) and C\(_5\)
(B) C\(_2\) and C\(_5\)
(C) C\(_1\) and C\(_4\)
(D) C\(_1\) and C\(_2\)
Ans: (B) - C\(_2\) and C\(_5\)
Sol: In fructose, the keto group is at C\(_2\). Intramolecular hemiketal formation between C\(_2\) (keto group) and the -OH at C\(_5\) gives a five-membered furanose ring.
Q4: \(\alpha\)-D-glucose and \(\beta\)-D-glucose differ from each other with respect to the: [1 mark]
(A) size of the hemiacetal ring
(B) configuration at the C₂ carbon
(C) number of -OH groups
(D) configuration at the C₁ carbon
Ans: (D) configuration at the C₁ carbon
Sol: α- and β-D-glucose are anomers. They differ only in the configuration of the hydroxyl group at the anomeric carbon (C₁). In α-D-glucose, the -OH at C₁ is below the plane, while in β-D-glucose it is above the plane.
Q5: \(\alpha\)-helix structure refers to: [1 Mark]
(A) primary structure of protein
(B) secondary structure
(C) tertiary structure
(D) quaternary structure
Ans: (B) secondary structure of protein
Sol: The α-helix is a type of secondary structure of proteins formed by coiling of the polypeptide chain. It is stabilized by intramolecular hydrogen bonds between the C=O group of one peptide bond and the N-H group of another peptide bond.
Q6: Assertion (A) : Vitamin K can be stored in our body.
Reason (R) : Vitamin K is a water soluble vitamin. [1 Mark]
(A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
(B) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
(C) Assertion (A) is true, but Reason (R) is false.
(D) Assertion (A) is false, but Reason (R) is true.
Ans: (C) A is true, R is false.
Sol: Vitamin K is stored in fatty tissues and liver - A true. But Vitamin K is fat-soluble, not water-soluble - R false.
Q7: 'Scurvy' is caused due to the deficiency of: [1 Mark]
(A) Vitamin A
(B) Vitamin B2
(C) Vitamin C
(D) Vitamin D
Ans: (C) Vitamin C (Ascorbic acid)
Sol: Scurvy is caused by deficiency of Vitamin C, leading to bleeding gums and poor wound healing.
Q8: Assertion (A) : Maltose is a reducing sugar.
Reason (R) : One of the two glucose units can open to expose free aldehydic group in solution. [1 Mark]
(A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
(B) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
(C) Assertion (A) is true, but Reason (R) is false.
(D) Assertion (A) is false, but Reason (R) is true.
Ans: (A) - Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation.
Sol: Maltose consists of two glucose units linked by an α(1→4) glycosidic bond. One glucose unit retains a free anomeric carbon that can open to form a free aldehyde group in solution, making maltose a reducing sugar. Thus, both Assertion and Reason are true, and Reason correctly explains the Assertion.
Q9: Deficiency of vitamin B1 causes the disease: [1 Mark]
(A) Convulsions
(B) Beri-Beri
(C) Fissuring at corners of mouth and lips (Cheilosis)
(D) Muscular weakness
Ans: (B) Beri-Beri
Sol: Deficiency of vitamin B1 (thiamine) causes Beri-Beri, which affects the nervous system and cardiovascular system.
Q10: Assertion (A) : Vitamin C cannot be stored in our body.
Reason (R) : Vitamin C is water soluble and excreted in urine. [1 Mark]
(A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
(B) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
(C) Assertion (A) is true, but Reason (R) is false.
(D) Assertion (A) is false, but Reason (R) is true.
Ans: (A) - Both A and R are true and R is the correct explanation of A.
Sol: Vitamin C is a water-soluble vitamin. It dissolves in body fluids and excess amounts are excreted in urine. Therefore, it is not stored in the body. Hence, both Assertion and Reason are true, and Reason correctly explains the Assertion.
Very Short Answer Type Questions
Q11: Write the reactions involved when D-glucose is treated with the following reagents: [2 Marks]
(a) HCN
(b) \(Br_{2}\) water
Ans:
(a) With HCN:
Glucose reacts with HCN to form a cyanohydrin by addition of HCN across the carbonyl group.
This confirms the presence of a carbonyl group in glucose.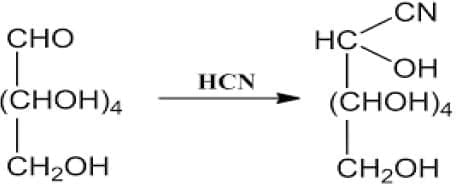
(b) With Br₂ water:
Bromine water oxidises the aldehyde group of glucose to a carboxylic acid forming gluconic acid.
Since Br₂ water is a mild oxidising agent that oxidises aldehydes but not alcohol groups, this confirms that glucose contains an aldehydic group.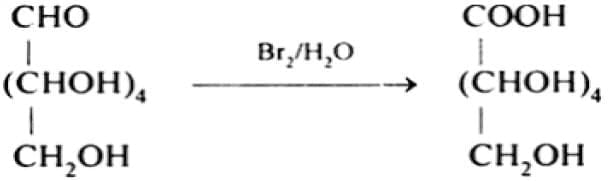
Q12: How do you explain the following? [2 Marks]
(a) Presence of an aldehydic group in glucose.
(b) Presence of five -OH groups in glucose.
Ans:
(a) Aldehydic group in glucose: Glucose gets oxidised to a six-carbon carboxylic acid (gluconic acid) on reaction with a mild oxidising agent like bromine water. This indicates the presence of an aldehydic (-CHO) group in glucose.
\( \underbrace{CHO\text{-(CHOH)}_4\text{-CH}_2OH}_{\text{Glucose}} \xrightarrow{Br_2/H_2O} \underbrace{COOH\text{-(CHOH)}_4\text{-CH}_2OH}_{\text{Gluconic acid}} \)
(b) Five -OH groups in glucose: Acetylation of glucose with acetic anhydride gives glucose pentaacetate. Since five acetyl groups are introduced, this confirms the presence of five -OH groups in glucose.
\( \text{Glucose} \xrightarrow{(CH_3CO)_2O} \text{Glucose pentaacetate} \)
Q13: Write two differences between DNA and RNA. [2 Marks]
Ans:
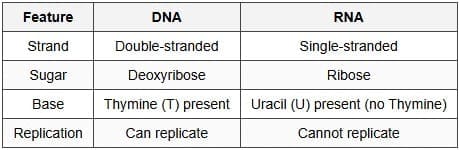
(Any two differences are sufficient.)
Q14: The two strands in DNA are not identical but complementary. Explain. What products would be formed when DNA is hydrolysed? [2 Marks]
The two DNA strands are complementary because specific base pairing occurs:
- Adenine pairs with Thymine (2 H-bonds)
- Guanine pairs with Cytosine (3 H-bonds)
Thus, the sequence of one strand determines the other.
On hydrolysis, DNA gives:
- 2-Deoxyribose sugar
- Nitrogenous bases (A, T, G, C)
- Phosphoric acid
(Any TWO)
Short Answer Type Questions
Q16: (a) What is the difference between native protein and denatured protein? [3 Marks]
(b) Which one of the following is a disaccharide ?
Glucose, Lactose, Amylose, Fructose
(c) Which vitamin is responsible for the coagulation of blood?
Ans:
(a)
- Native Protein: It refers to the protein in its biologically active form with its specific three-dimensional structure intact.
- Denatured Protein: When a protein is subjected to physical or chemical changes (like heat or pH change), its higher-order structures (secondary and tertiary) are destroyed, leading to a loss of biological activity.
Q17: Differentiate between: [3 Marks]
(a) Essential amino acids and Non-essential amino acids
(b) Peptide linkage and Glycosidic linkage
(c) Fibrous protein and Globular protein
Ans:
(a) Amino acids that cannot be synthesised in the body and must come from diet = essential amino acids. Those synthesised in the body = non-essential amino acids.
(b)
- Peptide linkage: -CO-NH- bond joining two amino acids.
- Glycosidic linkage: bond formed via oxygen joining two monosaccharide units.
(c)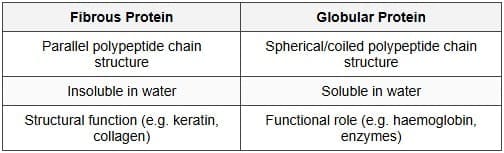
Case-Based Questions
Q18: Read the following passage and answer the questions: [5 Marks]
The α-amino acids are the building blocks of proteins. All α-amino acids exist as zwitter ion due to which they show amphoteric behaviour. All amino acids are joined through peptide bond. Proteins are broadly classified as globular proteins and fibrous proteins. Globular proteins are water soluble, whereas fibrous proteins are not. The complete structure of protein is discussed at four different levels i.e. primary, secondary, tertiary and quaternary structures. Protein loses its biological activity in denatured form.
(a) Define the following:
(i) Peptide linkage
(ii) Denatured protein
(b) Why do amino acids show amphoteric behaviour?
(c) (i) How can you differentiate between Fibrous protein and Globular protein?
(ii) Write the names of two different secondary structures of proteins.
Ans:
(a) (i) Peptide linkage: A peptide bond is a linkage (-CO-NH-) which joins amino acids together. It is formed by the condensation of the amino group of one amino acid with the carboxyl group of another, with loss of a water molecule.
(ii) Denatured protein: When a protein in its native form is subjected to a physical change (like change in temperature) or a chemical change (like change in pH), it loses its biological activity. This phenomenon is called denaturation.
(b) Amino acids show amphoteric behaviour due to the formation of zwitter ions. The amino group (-NH₂) can accept a proton (acts as base) and the carboxyl group (-COOH) can donate a proton (acts as acid). Due to the presence of both carboxylic and amino groups, they can react with both acids and bases.
(c) (i)
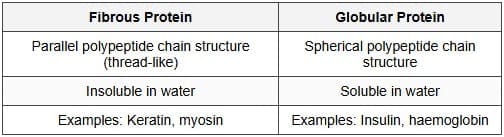
(ii) Two secondary structures of proteins: α-helix and β-pleated sheet
Q19: The following questions are case-based questions. Read the case carefully and answer the questions that follow.
Ribose and 2-deoxyribose have important roles in biology. Their most important derivatives are those in which a phosphate group is attached to the 5-position. The mono-, di-, and tri-phosphate forms as well as 3-5 cyclic monophosphate are important. Purines and pyrimidines form a major class of compounds with ribose and deoxyribose. When these purine and pyrimidine derivatives are coupled with ribose sugar, they are called nucleosides. [4 Marks]
(a) What products are formed when DNA is hydrolysed? How is DNA different from RNA in terms of structure?
Ans: Products of Hydrolysis: Complete hydrolysis of DNA yields a pentose sugar (2-deoxy-D-ribose), phosphoric acid, and nitrogen-containing heterocyclic bases (Adenine, Guanine, Cytosine, and Thymine).
Structural Difference:
(b) Differentiate between a Nucleotide and a Nucleoside.
Ans:
- Nucleoside: A unit formed by the attachment of a base to 1' position of sugar is known as nucleoside. (Base + Sugar)
- Nucleotide: When a nucleoside is linked to phosphoric acid at 5'-position of sugar moiety, we get a nucleotide. (Base + Sugar + Phosphate).
(c) (i) Mention two important functions of nucleic acids.
Ans: To preserve genetic information and Protein synthesis
OR
(c) (ii) Name the linkage joining two nucleotides. Name the base which is found in a nucleotide of RNA but not in DNA.
Ans: Linkage: Phosphodiester linkage joins two nucleotides.
Base: Uracil is found in RNA but not in DNA.
FAQs on CBSE Previous Year Questions: Biomolecules
| 1. What are biomolecules? |  |
| 2. What is the significance of carbohydrates in living organisms? |  |
| 3. How do enzymes function as proteins in biological systems? |  |
| 4. What are lipids and their functions in biological systems? |  |
| 5. What role do nucleic acids play in genetics? |  |





