JEE Exam > JEE Notes > Chemistry Main & Advanced > Infographic: Equilibrium
Infographic: Equilibrium
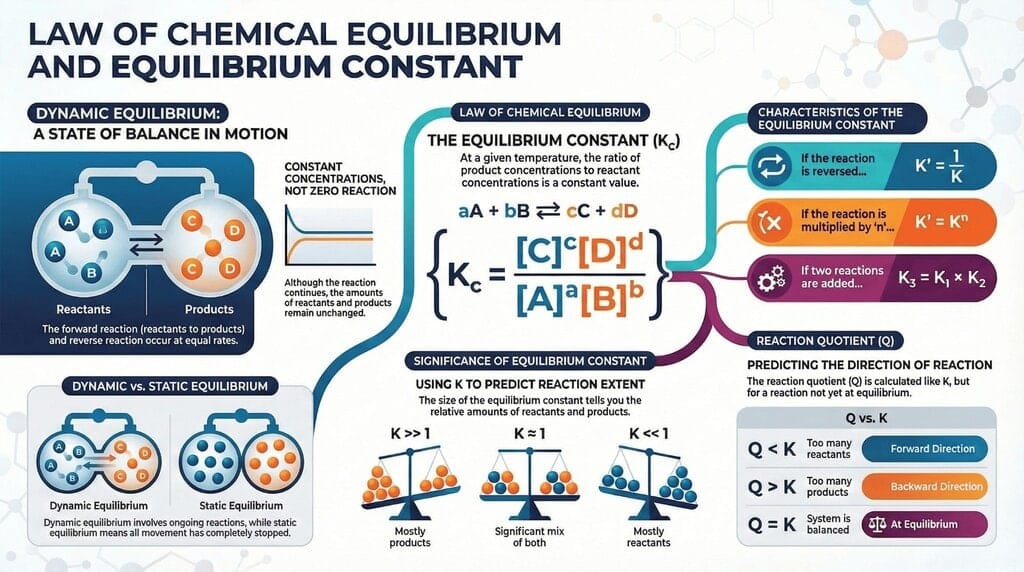
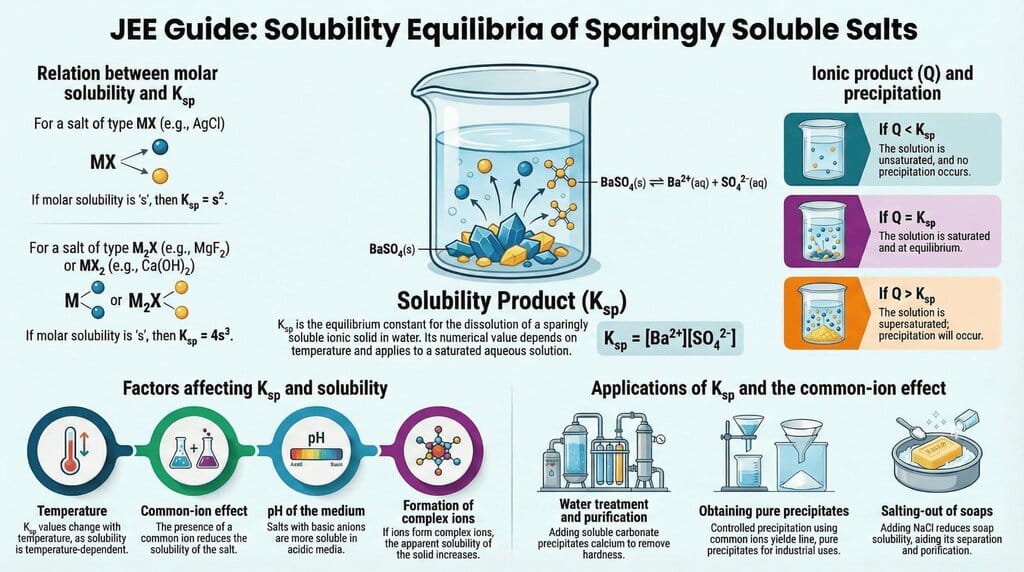
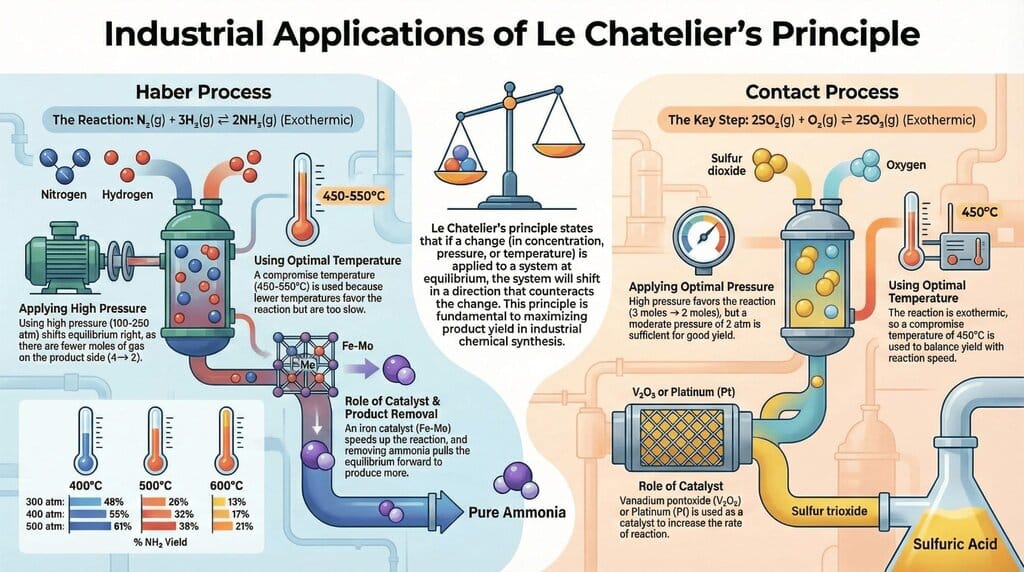
The document Infographic: Equilibrium is a part of the JEE Course Chemistry for JEE Main & Advanced.
All you need of JEE at this link: JEE
FAQs on Infographic: Equilibrium
| 1. What is the concept of equilibrium in chemistry? |  |
Ans. Equilibrium in chemistry refers to the state in a reversible reaction where the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products over time. This state can be dynamic, meaning the reactions still occur, but there is no net change in the concentrations of the substances involved.
| 2. How is the equilibrium constant (K) expressed? |  |
Ans. The equilibrium constant (K) is expressed as the ratio of the concentrations of the products raised to their coefficients to the concentrations of the reactants raised to their coefficients, according to the balanced chemical equation. For a reaction aA + bB ⇌ cC + dD, the expression for K is K = [C]ⁿ[D]ᵐ / [A]ˡ[B]ᵇ, where square brackets denote molarity.
| 3. What factors can affect the position of equilibrium? |  |
Ans. The position of equilibrium can be affected by changes in concentration, temperature, and pressure. According to Le Chatelier's principle, if a system at equilibrium is subjected to a change, the system adjusts to counteract that change and restore a new equilibrium. For instance, increasing the concentration of reactants shifts the equilibrium to favour the formation of products.
| 4. What is the significance of the reaction quotient (Q) in relation to equilibrium? |  |
Ans. The reaction quotient (Q) is a ratio similar to the equilibrium constant (K) but is calculated using the current concentrations of reactants and products at any point in time. By comparing Q to K, one can determine the direction in which the reaction will proceed to reach equilibrium: if Q < k,="" the="" reaction="" will="" shift="" right="" to="" produce="" more="" products;="" if="" q=""> K, it will shift left to produce more reactants.
| 5. Can equilibrium be reached in all chemical reactions? |  |
Ans. Not all chemical reactions can reach equilibrium. Some reactions are irreversible, meaning they go to completion and do not have a reversible nature. Equilibrium is typically associated with reversible reactions where both forward and reverse processes can occur, allowing for a balance between reactants and products.
Related Searches
Sample Paper, video lectures, Exam, Important questions, mock tests for examination, Semester Notes, shortcuts and tricks, practice quizzes, Infographic: Equilibrium, study material, Free, pdf , Viva Questions, Summary, Infographic: Equilibrium, MCQs, past year papers, Previous Year Questions with Solutions, Infographic: Equilibrium, Objective type Questions, Extra Questions, ppt;





