Grade 7 Exam > Grade 7 Notes > Natural Sciences > Worskheet -- Acids, bases and neutral substances
Worskheet -- Acids, bases and neutral substances
# Grade 7 Natural Sciences Worksheet ## Section A: Multiple Choice **1.** Which of the following is the correct definition of a **pure substance**? **[2]** A. A material made of two or more substances mixed together B. A material that contains only one type of particle and has fixed properties C. A substance that can be separated by filtration D. A mixture of elements and compounds **2.** Thandi heats a solid substance and it changes directly into a gas without becoming a liquid first. What is this process called? **[2]** A. Melting B. Evaporation C. Sublimation D. Condensation **3.** Which statement best describes an **element**? **[2]** A. It is made of two or more atoms chemically bonded together B. It consists of only one type of atom and cannot be broken down into simpler substances C. It is a mixture of different substances D. It can be separated into simpler substances by physical methods **4.** Sipho observes that when salt dissolves in water, the water becomes salty throughout. What type of mixture has been formed? **[2]** A. Heterogeneous mixture B. Suspension C. Homogeneous mixture D. Colloid --- ## Section B: True or False **1.** The boiling point of a substance is the temperature at which it changes from a solid to a liquid. **[1]** **2.** In a **compound**, atoms of different elements are chemically bonded together in fixed proportions. **[1]** **3.** Filtration is a separation method used to separate a dissolved solid from a liquid. **[1]** **4.** The kinetic theory of matter states that particles in a gas move faster than particles in a solid. **[1]** --- ## Section C: Match the Column Match each term in **Column A** with the correct description in **Column B**. Write only the letter of the correct answer next to the number. **[4]** **Column A:** 1. Diffusion 2. Freezing 3. Distillation 4. Atom **Column B:** A. The process where a liquid changes into a solid B. The smallest particle of an element that still has the properties of that element C. The movement of particles from an area of high concentration to an area of low concentration D. A separation technique used to separate liquids with different boiling points --- ## Section D: Fill in the Blanks Use the words from the word bank to complete the sentences below. Write only the word next to the number. **Word Bank:** solute, evaporation, molecule, physical **1.** A ________ is the smallest particle of a compound that still has the properties of that compound. **[1]** **2.** The substance that dissolves in a solvent to form a solution is called the ________. **[1]** **3.** When water changes from a liquid to a gas at the surface, the process is called ________. **[1]** **4.** A change that does not produce a new substance and can be reversed is called a ________ change. **[1]** --- ## Section E: Short Questions **1.** Define the term **chemical change** and give one example. **[3]** **2.** Explain why the particles in a liquid can flow and take the shape of their container. **[2]** **3.** Describe how you would use the process of **crystallisation** to obtain salt crystals from a salt solution. **[3]** **4.** State two differences between a mixture and a compound. **[4]** --- ## Section F: Scenario Question Lerato is investigating the properties of different substances in her science class. She has three beakers: - **Beaker 1** contains pure water - **Beaker 2** contains sugar dissolved in water - **Beaker 3** contains sand mixed with water She observes the contents and records her findings. **1.** Identify which beaker contains a **pure substance**. Explain your answer. **[2]** **2.** Which beaker contains a **solution**? Give a reason for your answer. **[3]** **3.** Lerato wants to separate the sand from the water in Beaker 3. Suggest a suitable separation method and explain how it works. **[3]** **4.** If Lerato heats Beaker 2 until all the water evaporates, what will remain in the beaker? Explain why this happens. **[3]** --- ## Section G: Long Question Bongani and Zanele are investigating the effect of temperature on the rate of dissolving. They take two beakers of water: one contains cold water at 10 °C and the other contains hot water at 60 °C. They add one teaspoon of sugar to each beaker at the same time and stir both at the same rate. They observe how long it takes for the sugar to dissolve completely in each beaker. **1.** State the **independent variable** and the **dependent variable** in this investigation. **[2]** **2.** Predict which beaker will have the sugar dissolve faster. Explain your prediction using the **kinetic theory of matter**. **[4]** **3.** Identify **two factors** that Bongani and Zanele kept constant (controlled variables) to make their investigation fair. **[2]** **4.** After conducting the experiment, they found that the sugar dissolved faster in the hot water. Evaluate whether their results support the kinetic theory of matter and justify your conclusion. **[4]** --- **[Grand Total: 60]** --- ## Answer Key Hello, well done for completing the worksheet! Before you check your answers, make sure you have attempted all the questions on your own. Use this answer key to see how well you understood the concepts and learn from any mistakes. Keep up the great effort! --- ### Section A - Question 1 **Answer: B** - A material that contains only one type of particle and has fixed properties A pure substance is made of only one kind of particle, so it has the same composition and properties throughout. Examples include pure water or pure iron. --- ### Section A - Question 2 **Answer: C** - Sublimation Sublimation is the process where a solid changes directly into a gas without passing through the liquid state. Dry ice (solid carbon dioxide) is a good example of this process. --- ### Section A - Question 3 **Answer: B** - It consists of only one type of atom and cannot be broken down into simpler substances An element is a pure substance that contains only one type of atom. It cannot be broken down into anything simpler by chemical methods. Examples are oxygen, gold, and carbon. --- ### Section A - Question 4 **Answer: C** - Homogeneous mixture A homogeneous mixture (also called a solution) is one where the substances are evenly mixed and you cannot see the separate parts. Salt water is a solution because the salt dissolves completely and spreads evenly through the water. --- ### Section B - Question 1 **FALSE** **Corrected statement:** The boiling point of a substance is the temperature at which it changes from a liquid to a gas. The original statement is wrong because it describes the **melting point**, not the boiling point. Boiling happens when a liquid turns into a gas. --- ### Section B - Question 2 **TRUE** This statement is correct. A compound is formed when atoms of different elements join together chemically in fixed ratios. For example, water (H₂O) always has two hydrogen atoms for every one oxygen atom. --- ### Section B - Question 3 **FALSE** **Corrected statement:** Filtration is a separation method used to separate an insoluble solid from a liquid. Filtration works only when the solid does not dissolve. To separate a dissolved solid, you would need evaporation or crystallisation instead. --- ### Section B - Question 4 **TRUE** This is correct. According to the kinetic theory of matter, particles in a gas have more energy and move much faster and more freely than particles in a solid, where they only vibrate in fixed positions. --- ### Section C - Question 1 **Answer: C** - The movement of particles from an area of high concentration to an area of low concentration Diffusion happens when particles spread out naturally from where there are many of them to where there are fewer. An example is the smell of perfume spreading across a room. --- ### Section C - Question 2 **Answer: A** - The process where a liquid changes into a solid Freezing is the change of state from liquid to solid. This happens when a liquid loses energy and its particles slow down enough to form a solid structure, like water turning to ice. --- ### Section C - Question 3 **Answer: D** - A separation technique used to separate liquids with different boiling points Distillation uses the fact that different liquids boil at different temperatures. You heat the mixture, and the liquid with the lower boiling point evaporates first, then it is cooled and collected separately. --- ### Section C - Question 4 **Answer: B** - The smallest particle of an element that still has the properties of that element An atom is the basic building block of all matter. Each element is made of a unique type of atom, and that atom has all the chemical properties of the element. --- ### Section D - Question 1 **Answer: molecule** A molecule is made when two or more atoms bond together chemically. If the atoms are from different elements, the molecule is part of a compound (like H₂O). --- ### Section D - Question 2 **Answer: solute** The solute is the substance that dissolves. In a cup of sugar water, the sugar is the solute and the water is the solvent. --- ### Section D - Question 3 **Answer: evaporation** Evaporation is when a liquid turns into a gas at temperatures below its boiling point, usually from the surface. It happens because some particles have enough energy to escape into the air. --- ### Section D - Question 4 **Answer: physical** A physical change does not create a new substance. Examples include melting, freezing, dissolving, and cutting. The material can usually be changed back. --- ### Section E - Question 1 **Model Answer:** A **chemical change** is a change in which one or more new substances are formed. *(1 mark)* The new substances have different properties from the original substances. *(1 mark)* An example is burning wood, which produces ash and smoke. *(1 mark)* **Key points to include:** definition that new substance is formed, mention of different properties, correct example. --- ### Section E - Question 2 **Model Answer:** Particles in a liquid are close together but not fixed in position. *(1 mark)* They can slide past each other, which allows the liquid to flow and take the shape of its container. *(1 mark)* **Key points:** particles can move past each other, this allows flow and shape change. --- ### Section E - Question 3 **Model Answer:** To obtain salt crystals by **crystallisation**, first heat the salt solution gently to evaporate some of the water. *(1 mark)* This makes the solution more concentrated. *(1 mark)* Then allow the solution to cool slowly, and salt crystals will form as the water continues to evaporate. *(1 mark)* **Key points:** heating/evaporating water, cooling slowly, crystals form. --- ### Section E - Question 4 **Model Answer:** **Difference 1:** A mixture contains two or more substances that are **not chemically bonded**, while a compound contains two or more elements that **are chemically bonded**. *(2 marks)* **Difference 2:** The substances in a mixture can be separated by **physical methods** like filtration or evaporation, but a compound can only be separated by **chemical methods**. *(2 marks)* **Key points:** bonding difference (2 marks), separation method difference (2 marks). --- ### Section F - Question 1 **Answer:** Beaker 1 contains a pure substance. *(1 mark)* Pure water is made of only one type of particle (H₂O molecules) and has fixed properties. *(1 mark)* --- ### Section F - Question 2 **Answer:** Beaker 2 contains a solution. *(1 mark)* A solution is a homogeneous mixture where one substance (the sugar/solute) is completely dissolved in another (the water/solvent). *(1 mark)* The sugar particles are evenly spread throughout the water and cannot be seen. *(1 mark)* --- ### Section F - Question 3 **Answer:** Lerato should use **filtration**. *(1 mark)* She pours the mixture through filter paper in a funnel. *(1 mark)* The sand particles are too large to pass through the tiny holes in the filter paper, so they are trapped, while the water passes through as the filtrate. *(1 mark)* --- ### Section F - Question 4 **Answer:** Sugar crystals will remain in the beaker. *(1 mark)* This is because evaporation removes only the water (which changes to water vapour), but the sugar was dissolved, not evaporated. *(1 mark)* The sugar stays behind as a solid because it does not evaporate at the temperature used. *(1 mark)* --- ### Section G - Question 1 **Answer:** **Independent variable:** Temperature of the water (or 10 °C vs 60 °C). *(1 mark)* **Dependent variable:** Time taken for the sugar to dissolve completely. *(1 mark)* --- ### Section G - Question 2 **Model Answer:** The sugar will dissolve faster in the hot water (60 °C). *(1 mark)* According to the kinetic theory of matter, particles in hot water have more energy and move faster than particles in cold water. *(1 mark)* This means the water particles collide with the sugar particles more often and with more energy. *(1 mark)* These collisions help break the sugar particles apart and spread them through the water more quickly. *(1 mark)* **Mark breakdown:** correct prediction (1), mention of kinetic theory/energy (1), explanation of faster particle movement (1), link to collisions and dissolving (1). --- ### Section G - Question 3 **Model Answer:** **Two controlled variables:** - The amount of sugar added to each beaker (one teaspoon). *(1 mark)* - The rate of stirring in both beakers. *(1 mark)* Other acceptable answers: volume of water, type of sugar used. --- ### Section G - Question 4 **Model Answer:** Yes, their results **support** the kinetic theory of matter. *(1 mark)* The kinetic theory predicts that higher temperatures give particles more kinetic energy, causing them to move faster. *(1 mark)* Because the sugar dissolved faster in hot water, this shows that the faster-moving water particles increased the rate of dissolving, which matches the theory. *(1 mark)* Therefore, the experiment provides evidence that particle movement and energy affect how quickly substances dissolve. *(1 mark)* **Full-mark answer must include:** clear statement of support, reference to kinetic energy and temperature, explanation linking faster particles to faster dissolving, concluding statement about evidence. **Partial answer (2 marks):** states support and mentions faster particles but does not fully explain the link. --- ### Mark Allocation Summary Table 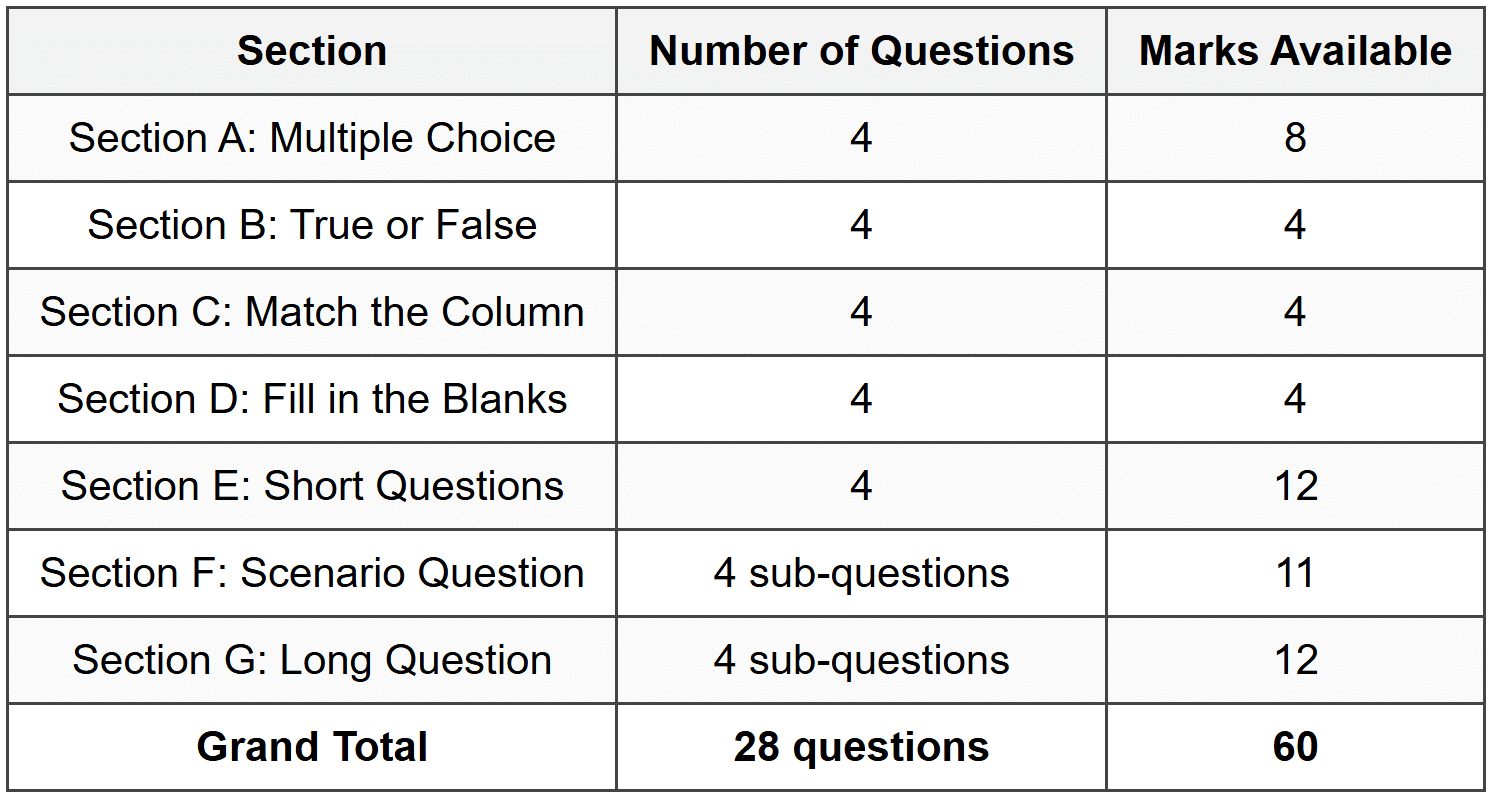

The document Worskheet -- Acids, bases and neutral substances is a part of the Grade 7 Course Natural Sciences Grade 7.
All you need of Grade 7 at this link: Grade 7
Related Searches
Extra Questions, Worskheet -- Acids, past year papers, bases and neutral substances, video lectures, Semester Notes, Worskheet -- Acids, Important questions, study material, pdf , Worskheet -- Acids, bases and neutral substances, Viva Questions, Exam, Summary, Objective type Questions, Free, mock tests for examination, bases and neutral substances, shortcuts and tricks, MCQs, Sample Paper, Previous Year Questions with Solutions, ppt, practice quizzes;





