Acids, bases and neutral substances
Section A: Multiple Choice
Choose the correct answer from the options A-D.
Question 1
Which of the following is a characteristic property of acids?
- A. They taste bitter
- B. They feel slippery to touch
- C. They turn blue litmus paper red
- D. They have a pH greater than 7
[2]
Question 2
Thandi tested a substance and found that it has a pH of 7. This substance is:
- A. A weak acid
- B. A strong base
- C. A neutral substance
- D. A corrosive substance
[2]
Question 3
Which indicator turns pink or red in a basic solution?
- A. Red litmus paper
- B. Blue litmus paper
- C. Phenolphthalein
- D. Methyl orange
[2]
Question 4
Lemon juice contains citric acid. What would you expect to happen when lemon juice is added to a solution of sodium bicarbonate?
- A. The mixture will become more acidic
- B. The mixture will produce bubbles of gas
- C. The mixture will turn purple
- D. Nothing will happen
[2]
Section B: True or False
State whether each statement is TRUE or FALSE.
Question 1
Bases turn red litmus paper blue. [1]
Question 2
All acids are dangerous and can burn your skin. [1]
Question 3
A substance with a pH of 10 is acidic. [1]
Question 4
Distilled water is a neutral substance. [1]
Section C: Match the Column
Match each item in Column A with the correct description in Column B. Write only the letter of the correct match.
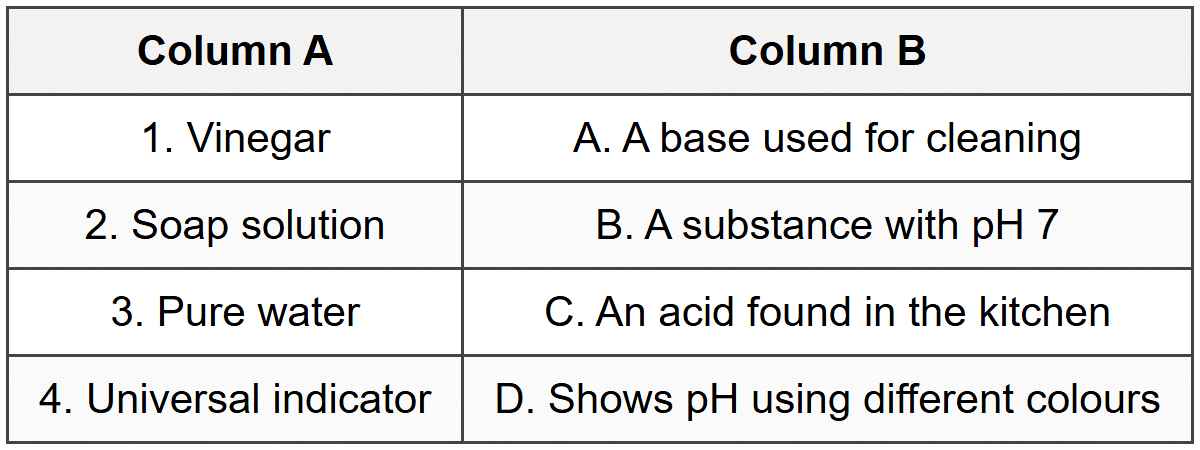
[4]
Section D: Fill in the Blanks
Complete the following sentences using the words from the word bank below.
Word Bank: sour, slippery, neutralisation, indicator, corrosive
Question 1
Acids have a ________ taste. [1]
Question 2
When an acid reacts with a base, the process is called ________. [1]
Question 3
An ________ is a substance that changes colour in acids and bases. [1]
Question 4
Bases feel ________ when touched with wet fingers. [1]
Section E: Short Questions
Question 1
Name two common household substances that are acidic. [2]
Question 2
Explain why it is important to know whether a substance is an acid or a base. [3]
Question 3
Describe what happens to the colour of universal indicator when it is added to an alkaline solution. [2]
Question 4
Sipho spilled some acid on the laboratory bench. Suggest a safe substance he could use to neutralise the acid. [2]
Section F: Scenario Question
Kagiso is doing an investigation for his Natural Sciences project. He collects five different liquids from around his home: orange juice, milk of magnesia, tap water, cola drink, and bleach. He wants to classify them as acids, bases, or neutral substances. Kagiso uses red and blue litmus paper to test each liquid and records his observations in a table.
Question 1
What colour will red litmus paper turn when dipped into bleach, which is a base? [2]
Question 2
Kagiso finds that both litmus papers stay the same colour when dipped into tap water. What does this tell him about the tap water? [2]
Question 3
The cola drink turns blue litmus paper red but does not change the colour of red litmus paper. Explain what type of substance cola drink is and why this result supports your answer. [3]
Question 4
Suggest one way Kagiso could improve his investigation to get more detailed information about how acidic or basic each substance is. [2]
Section G: Long Question
Amahle and Bongani are investigating neutralisation reactions. They pour 50 mL of dilute hydrochloric acid into a beaker and add a few drops of universal indicator. The solution turns red. They slowly add sodium hydroxide solution, which is a base, drop by drop while stirring. They observe that the colour of the solution gradually changes from red to orange, then to yellow, then to green.
Question 1
What does the red colour of the universal indicator tell Amahle and Bongani about the hydrochloric acid solution at the start of the experiment? [2]
Question 2
Explain what is happening chemically when the sodium hydroxide solution is added to the acid. [3]
Question 3
At which point in the colour change would the solution be neutral? Explain your answer. [3]
Question 4
Evaluate what would happen if Amahle and Bongani continued to add more sodium hydroxide solution after the solution turned green. Describe the colour change and the pH change you would expect, and explain why. [4]
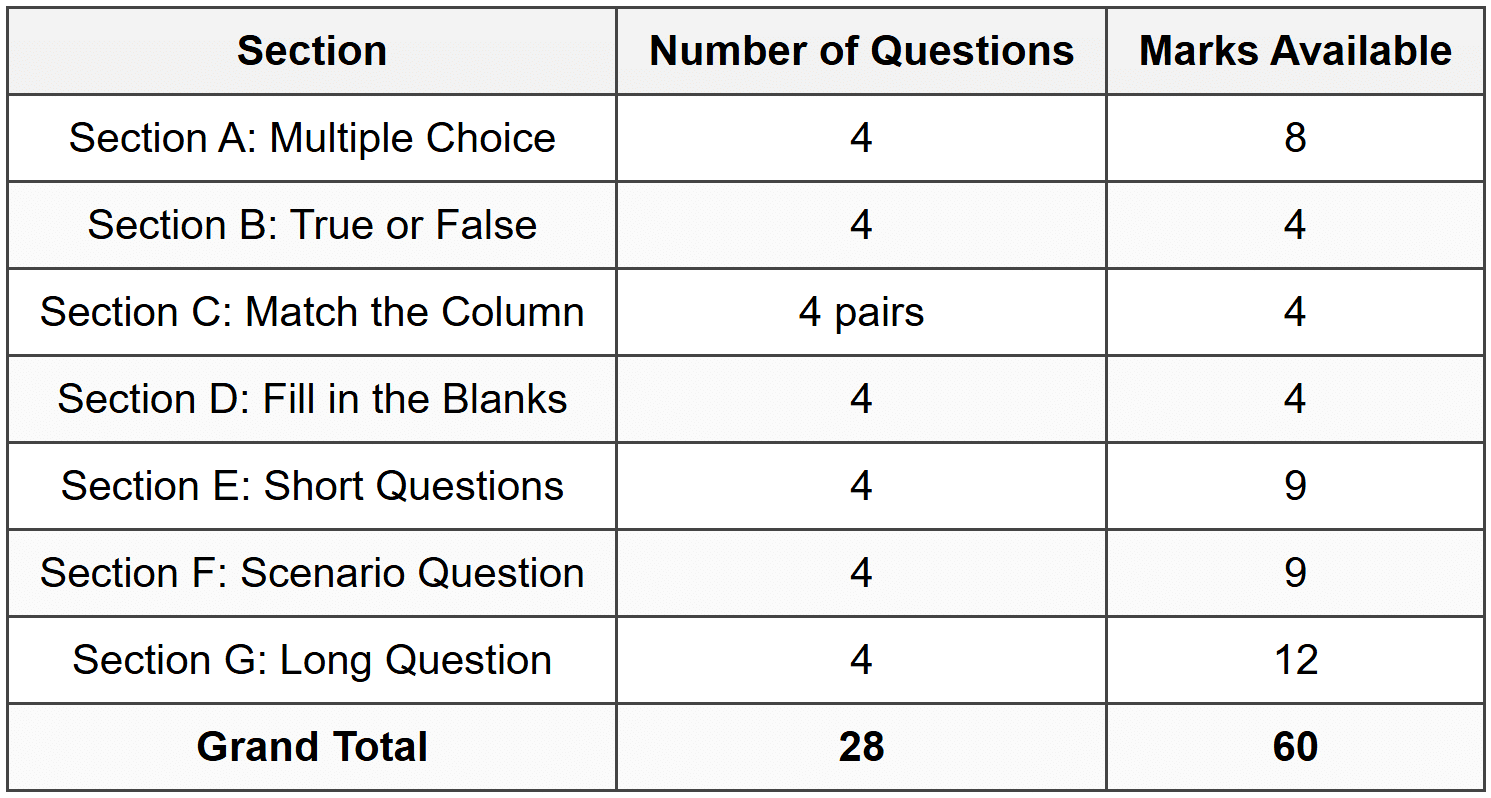
Grand Total: [60]
Answer Key
Well done for completing the worksheet! Remember to read through all your answers carefully and check that you have answered every part of each question. Use this answer key to mark your work honestly and learn from any mistakes. Keep up the great effort!
Section A - Question 1
Answer: C. They turn blue litmus paper red
This is correct because acids have the property of turning blue litmus paper red. This is a standard test used to identify acidic substances. Acids taste sour (not bitter), and they do not feel slippery.
Section A - Question 2
Answer: C. A neutral substance
A substance with a pH of 7 is neutral, meaning it is neither acidic nor basic. Pure water is a common example of a neutral substance with pH 7. Acids have pH less than 7, and bases have pH greater than 7.
Section A - Question 3
Answer: C. Phenolphthalein
Phenolphthalein is an indicator that is colourless in acidic and neutral solutions but turns pink or red in basic (alkaline) solutions. This makes it very useful for detecting bases.
Section A - Question 4
Answer: B. The mixture will produce bubbles of gas
When an acid like lemon juice reacts with sodium bicarbonate (a base), a neutralisation reaction occurs. This reaction produces carbon dioxide gas, which appears as bubbles. This is why baking soda fizzes when you add vinegar or lemon juice to it.
Section B - Question 1
TRUE
Bases turn red litmus paper blue. This is one of the key tests to identify a basic or alkaline substance.
Section B - Question 2
FALSE
Corrected statement: Some acids are weak and not dangerous, such as citric acid in oranges and acetic acid in vinegar.
While strong acids like concentrated sulfuric acid are corrosive and dangerous, many acids we encounter daily are weak and safe to consume in food. Not all acids burn your skin.
Section B - Question 3
FALSE
Corrected statement: A substance with a pH of 10 is basic (alkaline).
The pH scale ranges from 0 to 14. Substances with pH less than 7 are acidic, pH equal to 7 is neutral, and pH greater than 7 is basic. A pH of 10 is clearly on the basic side of the scale.
Section B - Question 4
TRUE
Distilled water is neutral with a pH of 7 because it contains equal amounts of hydrogen ions and hydroxide ions. It is neither acidic nor basic.
Section C - Match the Column
1 → C: Vinegar contains acetic acid, which is a common acid found in the kitchen used for cooking and preserving food.
2 → A: Soap solution is basic (alkaline) and is commonly used for cleaning because bases can break down grease and oils.
3 → B: Pure water has a pH of 7, making it a neutral substance.
4 → D: Universal indicator is a mixture of indicators that shows different colours at different pH values, allowing us to determine how acidic or basic a solution is.
Section D - Question 1
Answer: sour
Acids have a characteristic sour taste. Examples include lemon juice and vinegar, which both taste sour due to the acids they contain.
Section D - Question 2
Answer: neutralisation
When an acid reacts with a base, they neutralise each other to form a salt and water. This process is called neutralisation.
Section D - Question 3
Answer: indicator
An indicator is a substance that changes colour depending on whether it is in an acidic or basic solution. Litmus paper and universal indicator are examples.
Section D - Question 4
Answer: slippery
Bases feel slippery or soapy to touch when wet. This is because they react with the oils in your skin, which is why soap (which is basic) feels slippery.
Section E - Question 1
Model answer: Two common household acids are vinegar (which contains acetic acid) and lemon juice (which contains citric acid). Other acceptable answers include orange juice, cola drinks, or battery acid.
Marking: One mark for each correct example [1 + 1 = 2 marks]
Section E - Question 2
Model answer: It is important to know whether a substance is an acid or a base because this affects how we handle it safely. Strong acids and bases can be corrosive and cause burns. Knowing the nature of a substance also helps us understand how it will react with other substances, which is important in cooking, cleaning, and science experiments.
Marking: Safety reason [1], understanding reactions [1], example or elaboration [1] = 3 marks
Section E - Question 3
Model answer: When universal indicator is added to an alkaline solution, it will turn blue or purple, depending on how strong the base is. The stronger the base (higher pH), the darker purple the colour becomes.
Marking: Correct colour (blue/purple) [1], reference to strength or pH [1] = 2 marks
Section E - Question 4
Model answer: Sipho could use sodium bicarbonate (baking soda) or sodium carbonate to neutralise the acid safely. These are weak bases that will react with the acid to form harmless products like salt, water, and carbon dioxide.
Marking: Correct base named [1], explanation of neutralisation [1] = 2 marks
Section F - Question 1
Model answer: Red litmus paper will turn blue when dipped into bleach because bleach is a base, and bases turn red litmus paper blue.
Marking: Correct colour (blue) [1], explanation that bleach is a base [1] = 2 marks
Section F - Question 2
Model answer: If both red and blue litmus papers stay the same colour in tap water, this tells Kagiso that the tap water is neutral. Neutral substances do not change the colour of either red or blue litmus paper.
Marking: Conclusion that water is neutral [1], explanation about no colour change [1] = 2 marks
Section F - Question 3
Model answer: Cola drink is an acid. This result supports the answer because acids turn blue litmus paper red, which is exactly what happened. The red litmus paper stayed red because acids do not change the colour of red litmus paper. This behaviour is characteristic of acidic substances.
Marking: Identifying cola as acid [1], explanation of blue to red change [1], reference to red staying red [1] = 3 marks
Section F - Question 4
Model answer: Kagiso could use universal indicator or pH paper to test each substance. This would give him a numerical pH value or a range of colours showing exactly how acidic or basic each substance is, rather than just knowing if it is an acid, base, or neutral.
Marking: Suggesting universal indicator or pH paper [1], explanation of more detailed information [1] = 2 marks
Section G - Question 1
Model answer: The red colour of the universal indicator tells them that the hydrochloric acid solution is strongly acidic and has a low pH (around pH 1-3). Red is the colour universal indicator shows in the presence of strong acids.
Marking: Identifying solution as strongly acidic [1], reference to low pH [1] = 2 marks
Section G - Question 2
Model answer: When sodium hydroxide solution is added to the acid, a neutralisation reaction occurs. The hydroxide ions from the base react with the hydrogen ions from the acid to form water. At the same time, a salt (sodium chloride) is formed. As more base is added, the solution becomes less acidic and the pH increases.
Marking: Identifying neutralisation reaction [1], explaining ions reacting to form water [1], mentioning salt formation [1] = 3 marks
Section G - Question 3
Model answer: The solution would be neutral when the universal indicator turns green. This is because green indicates a pH of 7, which is neutral. At this point, the acid and base have completely neutralised each other, and there are equal amounts of hydrogen ions and hydroxide ions in the solution.
Marking: Identifying green colour [1], stating pH 7 [1], explaining complete neutralisation [1] = 3 marks
Section G - Question 4
Model answer: If they continue adding sodium hydroxide after the solution turns green, the colour will change from green to blue and then to purple. This happens because there is now excess base in the solution, making it alkaline. The pH will increase above 7, becoming progressively higher (more basic) as more sodium hydroxide is added. The solution goes from neutral to weakly basic (blue) to strongly basic (purple). This shows that neutralisation is complete and the solution now contains more base than acid.
Marking: Describing colour change to blue/purple [1], explaining excess base [1], stating pH increases above 7 [1], explaining why this happens [1] = 4 marks
Mark Allocation Summary
Remember, practice makes perfect! Review any questions you found difficult and make sure you understand the concepts. Keep up the excellent work in Natural Sciences!






