Grade 10 Exam > Grade 10 Notes > Physical Sciences > Electromagnetic Radiation
Electromagnetic Radiation
# Physical Sciences Grade 10 Worksheet
A. Energy that requires a medium to travel through
B. Energy that travels as transverse waves at the speed of light in a vacuum
C. Energy that can only travel through solids
D. Energy that is always visible to the human eye 2. The SI unit for frequency is:
A. metre (m)
B. second (s)
C. hertz (Hz)
D. joule (J) 3. Which type of electromagnetic radiation has the longest wavelength?
A. Gamma rays
B. Visible light
C. Ultraviolet radiation
D. Radio waves 4. The speed of light in a vacuum is approximately:
A. 3 × 106 m·s⁻¹
B. 3 × 108 m·s⁻¹
C. 3 × 1010 m·s⁻¹
D. 3 × 1012 m·s⁻¹ 5. Calculate the frequency of an electromagnetic wave with a wavelength of 5.0 × 10-7 m travelling through a vacuum.
A. 1.5 × 10-15 Hz
B. 1.67 × 10-1 Hz
C. 6.0 × 1014 Hz
D. 1.5 × 1015 Hz 6. Which statement about the electromagnetic spectrum is correct?
A. All electromagnetic waves travel at different speeds in a vacuum
B. Higher frequency electromagnetic waves have longer wavelengths
C. All electromagnetic waves are transverse waves
D. Only visible light can transfer energy 7. An electromagnetic wave has a frequency of 2.0 × 109 Hz. What is its wavelength in a vacuum?
A. 0.15 m
B. 0.67 m
C. 6.0 m
D. 6.0 × 1017 m 8. Which region of the electromagnetic spectrum is used in remote controls for televisions?
A. Microwaves
B. Infrared
C. Ultraviolet
D. X-rays 9. If the wavelength of an electromagnetic wave decreases, what happens to its frequency?
A. It decreases
B. It increases
C. It remains constant
D. It becomes zero 10. Calculate the wavelength of a radio wave with a frequency of 95.0 MHz (95.0 × 106 Hz).
A. 3.16 × 10-15 m
B. 3.16 m
C. 285 m
D. 2.85 × 1015 m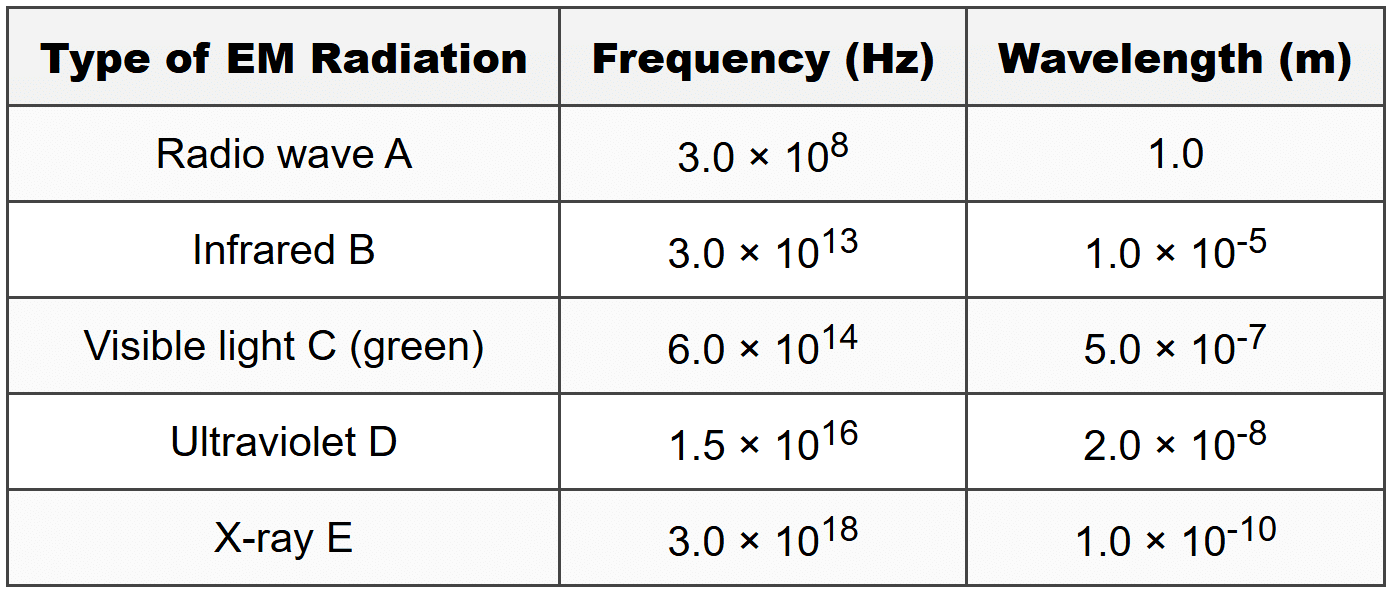 (a) Identify which type of electromagnetic radiation in the table has the highest frequency. (b) Using the data for Radio wave A, verify whether the relationship \( c = f \lambda \) holds true. Show all your working. (c) Calculate the ratio of the frequency of X-ray E to the frequency of Visible light C. Show all working. (d) By analyzing the data in the table, describe the relationship between frequency and wavelength for electromagnetic radiation. Explain whether this relationship is consistent across all the types of radiation shown, and suggest what this tells us about the nature of electromagnetic waves.
(a) Identify which type of electromagnetic radiation in the table has the highest frequency. (b) Using the data for Radio wave A, verify whether the relationship \( c = f \lambda \) holds true. Show all your working. (c) Calculate the ratio of the frequency of X-ray E to the frequency of Visible light C. Show all working. (d) By analyzing the data in the table, describe the relationship between frequency and wavelength for electromagnetic radiation. Explain whether this relationship is consistent across all the types of radiation shown, and suggest what this tells us about the nature of electromagnetic waves.
Electromagnetic radiation is a form of energy that travels as transverse waves through space (it does not need a medium). All electromagnetic waves travel at the speed of light in a vacuum, which is approximately 3 × 108 m·s⁻¹.
The SI unit for frequency is the hertz (Hz), which is equivalent to one cycle per second or s⁻¹.
In the electromagnetic spectrum, radio waves have the longest wavelength and the lowest frequency, while gamma rays have the shortest wavelength and the highest frequency.
The speed of light in a vacuum is a fundamental constant, denoted by c, and its value is approximately 3 × 108 m·s⁻¹.
Using the formula \( c = f \lambda \), we rearrange to \( f = \frac{c}{\lambda} \).
\( f = \frac{3 \times 10^8}{5.0 \times 10^{-7}} = 6.0 \times 10^{14} \) Hz.
This calculation shows that a wave with a wavelength of 5.0 × 10-7 m (which is in the visible light range) has a frequency of 6.0 × 1014 Hz.
All electromagnetic waves are transverse waves, meaning the oscillations of the electric and magnetic fields are perpendicular to the direction of wave propagation. They all travel at the same speed in a vacuum and can all transfer energy.
Using \( c = f \lambda \), we rearrange to \( \lambda = \frac{c}{f} \).
\( \lambda = \frac{3 \times 10^8}{2.0 \times 10^9} = 0.15 \) m.
This wave is in the microwave region of the electromagnetic spectrum.
Remote controls for televisions and other electronic devices typically use infrared radiation. These waves have wavelengths slightly longer than visible light and can carry signals over short distances.
According to the relationship \( c = f \lambda \), if the wavelength decreases, the frequency must increase to keep the speed of light constant in a vacuum. Frequency and wavelength are inversely proportional.
Using \( \lambda = \frac{c}{f} \):
\( \lambda = \frac{3 \times 10^8}{95.0 \times 10^6} = \frac{3 \times 10^8}{9.5 \times 10^7} = 3.16 \) m.
Radio waves used in FM broadcasting have wavelengths in the range of a few metres.
Step 2: Substitute: \( \lambda = \frac{3 \times 10^8 \text{ m·s}^{-1}}{2.45 \times 10^9 \text{ Hz}} \)
Step 3: Calculate:
\( \lambda = \frac{3 \times 10^8}{2.45 \times 10^9} \)
\( \lambda = 1.224 \times 10^{-1} \) m
\( \lambda = 0.122 \) m or 12.2 cm
Step 4: Final answer: λ = 0.122 m or 12.2 cm
Note: The unit (m or cm) is required for full marks.
Radio waves, Microwaves, Infrared, Visible light, Ultraviolet, X-rays, Gamma rays
Step 2: Substitute: \( f = \frac{3 \times 10^8 \text{ m·s}^{-1}}{1.0 \times 10^{-10} \text{ m}} \)
Step 3: Calculate:
\( f = 3 \times 10^8 \times 10^{10} \)
\( f = 3 \times 10^{18} \) Hz
Step 4: Final answer: f = 3.0 × 1018 Hz
Note: The unit Hz is required for full marks.
This can be identified directly from the table by comparing the frequency values.
Step 2: Substitute values for Radio wave A:
Frequency \( f = 3.0 \times 10^8 \) Hz
Wavelength \( \lambda = 1.0 \) m
Speed of light \( c = 3 \times 10^8 \) m·s⁻¹
Step 3: Calculate:
\( f \lambda = (3.0 \times 10^8 \text{ Hz}) \times (1.0 \text{ m}) \)
\( f \lambda = 3.0 \times 10^8 \text{ m·s}^{-1} \)
Step 4: This equals the speed of light \( c = 3 \times 10^8 \) m·s⁻¹.
Conclusion: Yes, the relationship \( c = f \lambda \) holds true for Radio wave A because the product of frequency and wavelength equals the speed of light.
Step 2: Substitute values:
\( \text{Ratio} = \frac{3.0 \times 10^{18} \text{ Hz}}{6.0 \times 10^{14} \text{ Hz}} \)
Step 3: Calculate:
\( \text{Ratio} = \frac{3.0}{6.0} \times 10^{18-14} \)
\( \text{Ratio} = 0.5 \times 10^{4} \)
\( \text{Ratio} = 5.0 \times 10^{3} \) or 5000
Step 4: Final answer: The ratio is 5.0 × 103 or 5000:1
This means X-ray E has a frequency 5000 times greater than Visible light C.
Full marks: A clear statement of inverse relationship, reference to specific data, mention of consistency, and explanation of the constant speed of light.
Partial marks: Identification of inverse relationship without full explanation or data reference.
This is the variable that Lebohang deliberately changed in her experiment by using radio waves, visible light, and X-rays, each with different wavelengths.
This is the variable that Lebohang measured or observed in response to changing the wavelength.
This must be kept constant because if the thickness varied, it would affect how much radiation could penetrate through the material, making it impossible to fairly compare the effect of wavelength alone on penetration ability. Other acceptable controlled variables include: the type of material (wood), the intensity of the radiation source, or the distance between the source and the board.
If the wavelength of electromagnetic radiation decreases, then the ability of the radiation to penetrate the wooden board will increase, because shorter wavelength (higher frequency) electromagnetic waves carry more energy and can interact more effectively with the atomic structure of materials, allowing some to pass through.
Or:
If electromagnetic radiation with shorter wavelengths is used, then more of the radiation will be detected on the other side of the wooden board, because higher energy radiation can penetrate materials more effectively than lower energy radiation.
Note: The hypothesis must be in the correct format and include a scientific reason based on the relationship between wavelength/frequency and energy.
The results show that the ability of electromagnetic radiation to penetrate the wooden board varies with wavelength, but not in a simple linear manner. Radio waves (longest wavelength, lowest frequency and energy) passed through easily, X-rays (shortest wavelength, highest frequency and energy) also passed through but with reduced intensity, while visible light (intermediate wavelength) did not pass through at all. The hypothesis is partially supported. While X-rays did penetrate better than visible light, suggesting that higher energy radiation can penetrate materials, the fact that radio waves (with the lowest energy) penetrated most easily contradicts the hypothesis. This can be explained scientifically: the interaction of electromagnetic radiation with matter depends not only on energy, but also on the size of the wavelength relative to the structure of the material. Radio waves have wavelengths much larger than the gaps and structures in wood, so they pass through without much interaction. Visible light has wavelengths comparable to the cellular structures in wood, causing scattering and absorption, blocking transmission. X-rays have wavelengths much smaller than atomic structures, allowing them to pass between atoms, though some energy is still absorbed. Therefore, penetration depends on both the energy of the radiation and how its wavelength compares to the physical structure of the material.
Full marks: States whether hypothesis is supported (with justification), references the specific results, provides scientific explanation involving both energy and wavelength-structure interaction.
Partial marks: States support for hypothesis without full scientific reasoning, or discusses results without clear conclusion.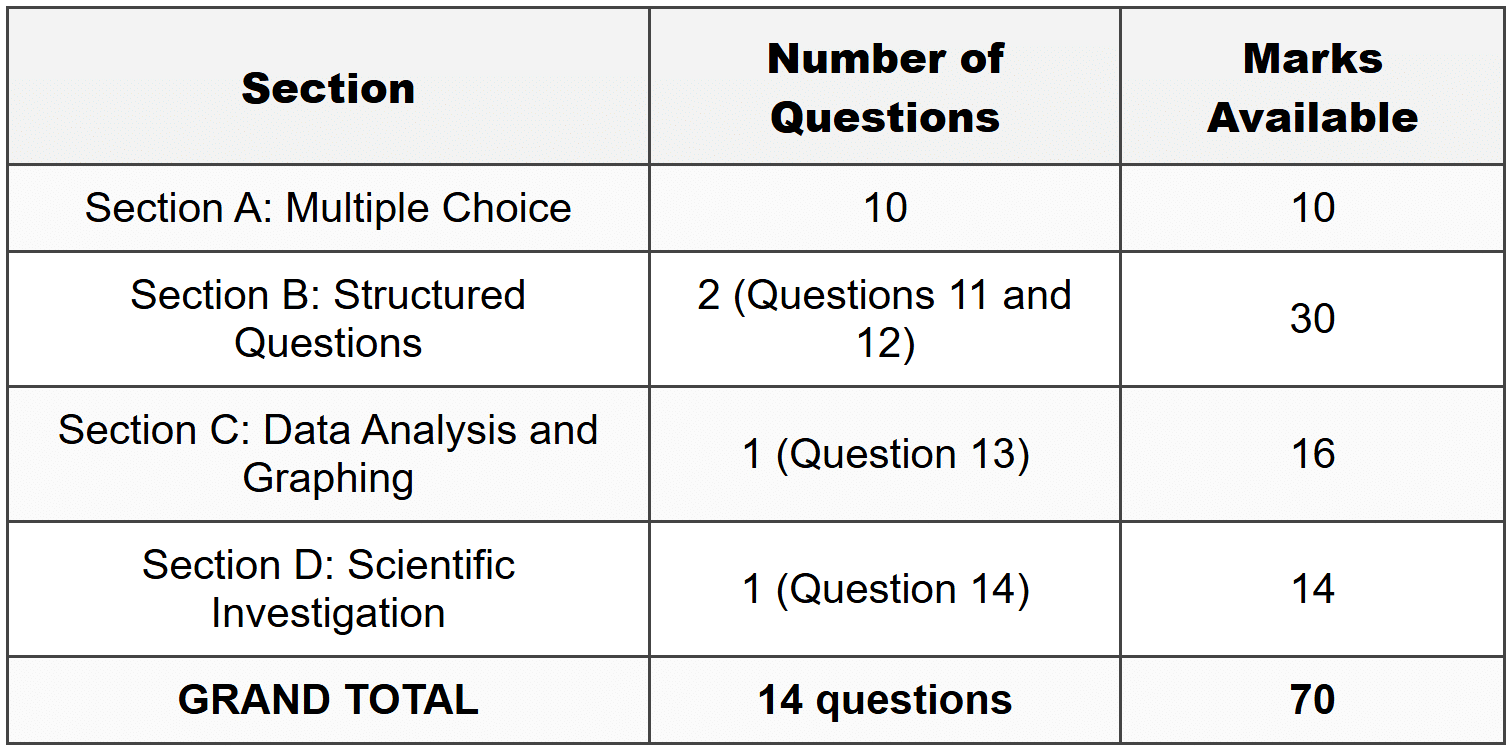
SECTION A: MULTIPLE CHOICE
(Total: 10 marks) 1. Which of the following best describes electromagnetic radiation?A. Energy that requires a medium to travel through
B. Energy that travels as transverse waves at the speed of light in a vacuum
C. Energy that can only travel through solids
D. Energy that is always visible to the human eye 2. The SI unit for frequency is:
A. metre (m)
B. second (s)
C. hertz (Hz)
D. joule (J) 3. Which type of electromagnetic radiation has the longest wavelength?
A. Gamma rays
B. Visible light
C. Ultraviolet radiation
D. Radio waves 4. The speed of light in a vacuum is approximately:
A. 3 × 106 m·s⁻¹
B. 3 × 108 m·s⁻¹
C. 3 × 1010 m·s⁻¹
D. 3 × 1012 m·s⁻¹ 5. Calculate the frequency of an electromagnetic wave with a wavelength of 5.0 × 10-7 m travelling through a vacuum.
A. 1.5 × 10-15 Hz
B. 1.67 × 10-1 Hz
C. 6.0 × 1014 Hz
D. 1.5 × 1015 Hz 6. Which statement about the electromagnetic spectrum is correct?
A. All electromagnetic waves travel at different speeds in a vacuum
B. Higher frequency electromagnetic waves have longer wavelengths
C. All electromagnetic waves are transverse waves
D. Only visible light can transfer energy 7. An electromagnetic wave has a frequency of 2.0 × 109 Hz. What is its wavelength in a vacuum?
A. 0.15 m
B. 0.67 m
C. 6.0 m
D. 6.0 × 1017 m 8. Which region of the electromagnetic spectrum is used in remote controls for televisions?
A. Microwaves
B. Infrared
C. Ultraviolet
D. X-rays 9. If the wavelength of an electromagnetic wave decreases, what happens to its frequency?
A. It decreases
B. It increases
C. It remains constant
D. It becomes zero 10. Calculate the wavelength of a radio wave with a frequency of 95.0 MHz (95.0 × 106 Hz).
A. 3.16 × 10-15 m
B. 3.16 m
C. 285 m
D. 2.85 × 1015 m
SECTION B: STRUCTURED QUESTIONS
(Total: 30 marks)Question 11
(a) Define the term wavelength as it applies to electromagnetic radiation. (b) State the relationship between frequency and wavelength for electromagnetic waves travelling through a vacuum. (c) A microwave oven produces electromagnetic waves with a frequency of 2.45 × 109 Hz. Calculate the wavelength of these microwaves. Show all working. (d) Thabo observes that when he increases the power setting on his microwave oven, the food heats faster, but the frequency of the microwaves remains the same. Explain what property of the electromagnetic waves changes when the power is increased, and why this causes faster heating.Question 12
(a) List the seven main types of electromagnetic radiation in order from longest wavelength to shortest wavelength. (b) Explain why ultraviolet radiation from the sun is considered more dangerous to human skin than visible light. (c) A hospital uses X-rays with a wavelength of 1.0 × 10-10 m for medical imaging. Calculate the frequency of these X-rays. Show all working. (d) Nomvula reads that gamma rays have the highest frequency in the electromagnetic spectrum. She then reads that gamma rays are used to sterilize medical equipment and to treat certain cancers. Using your understanding of the relationship between frequency and energy, evaluate why gamma rays would be effective for these purposes but also why they must be used with extreme caution.SECTION C: DATA ANALYSIS AND GRAPHING
(Total: 16 marks)Question 13
Sipho conducted an investigation into different types of electromagnetic radiation. He collected data on the frequency and wavelength of various electromagnetic waves. His results are shown in the table below: (a) Identify which type of electromagnetic radiation in the table has the highest frequency. (b) Using the data for Radio wave A, verify whether the relationship \( c = f \lambda \) holds true. Show all your working. (c) Calculate the ratio of the frequency of X-ray E to the frequency of Visible light C. Show all working. (d) By analyzing the data in the table, describe the relationship between frequency and wavelength for electromagnetic radiation. Explain whether this relationship is consistent across all the types of radiation shown, and suggest what this tells us about the nature of electromagnetic waves.
(a) Identify which type of electromagnetic radiation in the table has the highest frequency. (b) Using the data for Radio wave A, verify whether the relationship \( c = f \lambda \) holds true. Show all your working. (c) Calculate the ratio of the frequency of X-ray E to the frequency of Visible light C. Show all working. (d) By analyzing the data in the table, describe the relationship between frequency and wavelength for electromagnetic radiation. Explain whether this relationship is consistent across all the types of radiation shown, and suggest what this tells us about the nature of electromagnetic waves.SECTION D: SCIENTIFIC INVESTIGATION
(Total: 14 marks)Question 14
Lebohang wants to investigate how the wavelength of electromagnetic waves affects their ability to penetrate different materials. She sets up an experiment using electromagnetic radiation sources of different wavelengths: radio waves (wavelength = 1 m), visible light (wavelength = 5.0 × 10-7 m), and X-rays (wavelength = 1.0 × 10-10 m). She directs each type of radiation at a 5 cm thick wooden board and measures whether the radiation is detected on the other side using appropriate detectors. She keeps the intensity of each radiation source constant and uses the same wooden board for all tests. Her results show that radio waves pass through easily, visible light does not pass through at all, and X-rays pass through but with reduced intensity. (a) Identify the independent variable in Lebohang's investigation. (b) Identify the dependent variable in Lebohang's investigation. (c) Name one controlled variable in this investigation and explain why it must be kept constant. (d) Write a hypothesis for this investigation using the format: If [condition], then [expected result], because [scientific reason]. (e) Based on the results described, state a conclusion for this investigation. In your conclusion, explain whether the hypothesis you wrote in part (d) was supported by the evidence, and provide a scientific explanation for why different wavelengths of electromagnetic radiation interact differently with the wooden board. GRAND TOTAL: 70ANSWER KEY
Dear Student,
Well done for completing this worksheet! Before you check the answers below, make sure you have attempted every question to the best of your ability. Use this answer key to understand where you went wrong and to learn from your mistakes. Remember, making mistakes is part of learning! Good luck with your studies.
SECTION A - Question 1
Answer: B. Energy that travels as transverse waves at the speed of light in a vacuumElectromagnetic radiation is a form of energy that travels as transverse waves through space (it does not need a medium). All electromagnetic waves travel at the speed of light in a vacuum, which is approximately 3 × 108 m·s⁻¹.
SECTION A - Question 2
Answer: C. hertz (Hz)The SI unit for frequency is the hertz (Hz), which is equivalent to one cycle per second or s⁻¹.
SECTION A - Question 3
Answer: D. Radio wavesIn the electromagnetic spectrum, radio waves have the longest wavelength and the lowest frequency, while gamma rays have the shortest wavelength and the highest frequency.
SECTION A - Question 4
Answer: B. 3 × 108 m·s⁻¹The speed of light in a vacuum is a fundamental constant, denoted by c, and its value is approximately 3 × 108 m·s⁻¹.
SECTION A - Question 5
Answer: C. 6.0 × 1014 HzUsing the formula \( c = f \lambda \), we rearrange to \( f = \frac{c}{\lambda} \).
\( f = \frac{3 \times 10^8}{5.0 \times 10^{-7}} = 6.0 \times 10^{14} \) Hz.
This calculation shows that a wave with a wavelength of 5.0 × 10-7 m (which is in the visible light range) has a frequency of 6.0 × 1014 Hz.
SECTION A - Question 6
Answer: C. All electromagnetic waves are transverse wavesAll electromagnetic waves are transverse waves, meaning the oscillations of the electric and magnetic fields are perpendicular to the direction of wave propagation. They all travel at the same speed in a vacuum and can all transfer energy.
SECTION A - Question 7
Answer: A. 0.15 mUsing \( c = f \lambda \), we rearrange to \( \lambda = \frac{c}{f} \).
\( \lambda = \frac{3 \times 10^8}{2.0 \times 10^9} = 0.15 \) m.
This wave is in the microwave region of the electromagnetic spectrum.
SECTION A - Question 8
Answer: B. InfraredRemote controls for televisions and other electronic devices typically use infrared radiation. These waves have wavelengths slightly longer than visible light and can carry signals over short distances.
SECTION A - Question 9
Answer: B. It increasesAccording to the relationship \( c = f \lambda \), if the wavelength decreases, the frequency must increase to keep the speed of light constant in a vacuum. Frequency and wavelength are inversely proportional.
SECTION A - Question 10
Answer: B. 3.16 mUsing \( \lambda = \frac{c}{f} \):
\( \lambda = \frac{3 \times 10^8}{95.0 \times 10^6} = \frac{3 \times 10^8}{9.5 \times 10^7} = 3.16 \) m.
Radio waves used in FM broadcasting have wavelengths in the range of a few metres.
SECTION B - Question 11(a)
Wavelength is defined as the distance between two consecutive corresponding points on a wave, such as from one crest to the next crest, or from one trough to the next trough. It is measured in metres (m). For electromagnetic radiation, wavelength determines the type of radiation in the electromagnetic spectrum.SECTION B - Question 11(b)
The relationship between frequency and wavelength for electromagnetic waves is given by the equation \( c = f \lambda \), where c is the speed of light in a vacuum (3 × 108 m·s⁻¹), f is the frequency in hertz (Hz), and \( \lambda \) is the wavelength in metres (m). This equation shows that frequency and wavelength are inversely proportional: as frequency increases, wavelength decreases, and vice versa.SECTION B - Question 11(c)
Step 1: Formula: \( c = f \lambda \) therefore \( \lambda = \frac{c}{f} \)Step 2: Substitute: \( \lambda = \frac{3 \times 10^8 \text{ m·s}^{-1}}{2.45 \times 10^9 \text{ Hz}} \)
Step 3: Calculate:
\( \lambda = \frac{3 \times 10^8}{2.45 \times 10^9} \)
\( \lambda = 1.224 \times 10^{-1} \) m
\( \lambda = 0.122 \) m or 12.2 cm
Step 4: Final answer: λ = 0.122 m or 12.2 cm
Note: The unit (m or cm) is required for full marks.
SECTION B - Question 11(d)
When Thabo increases the power setting, the amplitude (or intensity) of the electromagnetic waves increases, not the frequency or wavelength. The amplitude of an electromagnetic wave is related to the amount of energy it carries. A higher amplitude means more energy is transferred to the food per unit time, which causes the water molecules in the food to vibrate more vigorously and heat up faster. The frequency remains constant because it is determined by the design of the microwave oven's magnetron.SECTION B - Question 12(a)
The seven main types of electromagnetic radiation in order from longest wavelength to shortest wavelength are:Radio waves, Microwaves, Infrared, Visible light, Ultraviolet, X-rays, Gamma rays
SECTION B - Question 12(b)
Ultraviolet (UV) radiation is more dangerous to human skin than visible light because UV radiation has a higher frequency and therefore carries more energy per photon. This higher energy allows UV radiation to penetrate the skin and damage the DNA in skin cells, which can lead to mutations, sunburn, premature aging, and an increased risk of skin cancer. Visible light has lower frequency and energy, and therefore does not cause the same level of cellular damage.SECTION B - Question 12(c)
Step 1: Formula: \( c = f \lambda \) therefore \( f = \frac{c}{\lambda} \)Step 2: Substitute: \( f = \frac{3 \times 10^8 \text{ m·s}^{-1}}{1.0 \times 10^{-10} \text{ m}} \)
Step 3: Calculate:
\( f = 3 \times 10^8 \times 10^{10} \)
\( f = 3 \times 10^{18} \) Hz
Step 4: Final answer: f = 3.0 × 1018 Hz
Note: The unit Hz is required for full marks.
SECTION B - Question 12(d)
Gamma rays have the highest frequency in the electromagnetic spectrum, which means they also have the highest energy per photon. This high energy gives gamma rays the ability to penetrate deeply into materials, including living tissue, and to break molecular bonds, including the bonds in DNA. This property makes gamma rays effective for sterilizing medical equipment (by killing bacteria and viruses) and for treating cancers (by destroying cancer cells). However, because gamma rays can damage or destroy any living cells, not just harmful ones, they must be used with extreme caution. Overexposure to gamma rays can cause radiation sickness, tissue damage, and increase the risk of cancer. Therefore, their use in medicine requires careful control, shielding, and precise targeting to minimize harm to healthy tissue.SECTION C - Question 13(a)
X-ray E has the highest frequency at 3.0 × 1018 Hz.This can be identified directly from the table by comparing the frequency values.
SECTION C - Question 13(b)
Step 1: Formula: \( c = f \lambda \)Step 2: Substitute values for Radio wave A:
Frequency \( f = 3.0 \times 10^8 \) Hz
Wavelength \( \lambda = 1.0 \) m
Speed of light \( c = 3 \times 10^8 \) m·s⁻¹
Step 3: Calculate:
\( f \lambda = (3.0 \times 10^8 \text{ Hz}) \times (1.0 \text{ m}) \)
\( f \lambda = 3.0 \times 10^8 \text{ m·s}^{-1} \)
Step 4: This equals the speed of light \( c = 3 \times 10^8 \) m·s⁻¹.
Conclusion: Yes, the relationship \( c = f \lambda \) holds true for Radio wave A because the product of frequency and wavelength equals the speed of light.
SECTION C - Question 13(c)
Step 1: Formula for ratio: \( \text{Ratio} = \frac{f_{\text{X-ray}}}{f_{\text{Visible light}}} \)Step 2: Substitute values:
\( \text{Ratio} = \frac{3.0 \times 10^{18} \text{ Hz}}{6.0 \times 10^{14} \text{ Hz}} \)
Step 3: Calculate:
\( \text{Ratio} = \frac{3.0}{6.0} \times 10^{18-14} \)
\( \text{Ratio} = 0.5 \times 10^{4} \)
\( \text{Ratio} = 5.0 \times 10^{3} \) or 5000
Step 4: Final answer: The ratio is 5.0 × 103 or 5000:1
This means X-ray E has a frequency 5000 times greater than Visible light C.
SECTION C - Question 13(d)
The data in the table shows an inverse relationship between frequency and wavelength for electromagnetic radiation. As frequency increases, wavelength decreases. For example, Radio wave A has the lowest frequency (3.0 × 108 Hz) and the longest wavelength (1.0 m), while X-ray E has the highest frequency (3.0 × 1018 Hz) and the shortest wavelength (1.0 × 10-10 m). This relationship is consistent across all types of radiation shown in the table. This tells us that all electromagnetic waves obey the same fundamental relationship \( c = f \lambda \), meaning they all travel at the same speed in a vacuum (the speed of light), regardless of their frequency or wavelength. The inverse relationship arises because the speed is constant.Full marks: A clear statement of inverse relationship, reference to specific data, mention of consistency, and explanation of the constant speed of light.
Partial marks: Identification of inverse relationship without full explanation or data reference.
SECTION D - Question 14(a)
The independent variable is the wavelength of the electromagnetic radiation (or the type of electromagnetic radiation).This is the variable that Lebohang deliberately changed in her experiment by using radio waves, visible light, and X-rays, each with different wavelengths.
SECTION D - Question 14(b)
The dependent variable is the ability of the radiation to penetrate the wooden board (or whether the radiation is detected on the other side or the amount/intensity of radiation that passes through).This is the variable that Lebohang measured or observed in response to changing the wavelength.
SECTION D - Question 14(c)
One controlled variable is the thickness of the wooden board (5 cm).This must be kept constant because if the thickness varied, it would affect how much radiation could penetrate through the material, making it impossible to fairly compare the effect of wavelength alone on penetration ability. Other acceptable controlled variables include: the type of material (wood), the intensity of the radiation source, or the distance between the source and the board.
SECTION D - Question 14(d)
Model hypothesis:If the wavelength of electromagnetic radiation decreases, then the ability of the radiation to penetrate the wooden board will increase, because shorter wavelength (higher frequency) electromagnetic waves carry more energy and can interact more effectively with the atomic structure of materials, allowing some to pass through.
Or:
If electromagnetic radiation with shorter wavelengths is used, then more of the radiation will be detected on the other side of the wooden board, because higher energy radiation can penetrate materials more effectively than lower energy radiation.
Note: The hypothesis must be in the correct format and include a scientific reason based on the relationship between wavelength/frequency and energy.
SECTION D - Question 14(e)
Model conclusion:The results show that the ability of electromagnetic radiation to penetrate the wooden board varies with wavelength, but not in a simple linear manner. Radio waves (longest wavelength, lowest frequency and energy) passed through easily, X-rays (shortest wavelength, highest frequency and energy) also passed through but with reduced intensity, while visible light (intermediate wavelength) did not pass through at all. The hypothesis is partially supported. While X-rays did penetrate better than visible light, suggesting that higher energy radiation can penetrate materials, the fact that radio waves (with the lowest energy) penetrated most easily contradicts the hypothesis. This can be explained scientifically: the interaction of electromagnetic radiation with matter depends not only on energy, but also on the size of the wavelength relative to the structure of the material. Radio waves have wavelengths much larger than the gaps and structures in wood, so they pass through without much interaction. Visible light has wavelengths comparable to the cellular structures in wood, causing scattering and absorption, blocking transmission. X-rays have wavelengths much smaller than atomic structures, allowing them to pass between atoms, though some energy is still absorbed. Therefore, penetration depends on both the energy of the radiation and how its wavelength compares to the physical structure of the material.
Full marks: States whether hypothesis is supported (with justification), references the specific results, provides scientific explanation involving both energy and wavelength-structure interaction.
Partial marks: States support for hypothesis without full scientific reasoning, or discusses results without clear conclusion.
MARK ALLOCATION SUMMARY

The document Electromagnetic Radiation is a part of the Grade 10 Course Physical Sciences for Grade 10.
All you need of Grade 10 at this link: Grade 10
Related Searches
mock tests for examination, Important questions, shortcuts and tricks, practice quizzes, Free, past year papers, Electromagnetic Radiation, ppt, Extra Questions, Sample Paper, Exam, video lectures, study material, Previous Year Questions with Solutions, Electromagnetic Radiation, Electromagnetic Radiation, Viva Questions, MCQs, Objective type Questions, pdf , Summary, Semester Notes;






