The Particles That Substances Are Made of
SECTION A: MULTIPLE CHOICE
(Total: 10 marks)
1. The kinetic molecular theory states that particles in a gas are:
A. closely packed and vibrate in fixed positionsB. in constant random motion and collide with each other and the container walls
C. moving slowly and rarely collide
D. stationary and have no kinetic energy
2. The SI unit for temperature used in gas law calculations is:
A. degrees Celsius (°C)B. Kelvin (K)
C. joules (J)
D. pascals (Pa)
3. A gas occupies a volume of 2.5 m³ at a pressure of 100 kPa. If the pressure increases to 200 kPa at constant temperature, what is the new volume?
A. 1.25 m³B. 5.0 m³
C. 2.5 m³
D. 0.50 m³
4. Which state of matter has particles with the strongest intermolecular forces?
A. GasB. Liquid
C. Solid
D. Plasma
5. A sealed syringe containing air is heated from 27 °C to 127 °C at constant pressure. The volume of the air:
A. decreases by a factor of 4.7B. increases by a factor of 1.33
C. remains constant
D. decreases by half
6. According to the kinetic molecular theory, when the temperature of a gas increases, the average kinetic energy of the particles:
A. decreasesB. remains constant
C. increases
D. becomes zero
7. A gas has a volume of 500 cm³ at 25 °C. If the gas is cooled to 0 °C at constant pressure, what is the new volume? (Assume ideal gas behaviour)
A. 458 cm³B. 546 cm³
C. 500 cm³
D. 273 cm³
8. The process by which particles move from an area of high concentration to an area of low concentration is called:
A. condensationB. diffusion
C. sublimation
D. deposition
9. At standard temperature and pressure (STP), one mole of any ideal gas occupies approximately:
A. 1 dm³B. 22.4 dm³
C. 100 dm³
D. 273 dm³
10. A container holds 3 moles of nitrogen gas at STP. What volume does the gas occupy?
A. 22.4 dm³B. 44.8 dm³
C. 67.2 dm³
D. 7.5 dm³
SECTION B: STRUCTURED QUESTIONS
(Total: 30 marks)
Question 11
(a) Define the term kinetic molecular theory.
(b) State two assumptions of the kinetic molecular theory regarding the behaviour of gas particles.
(c) Explain why increasing the temperature of a gas in a closed rigid container causes the pressure to increase.
(d) A sample of oxygen gas occupies a volume of 4.0 dm³ at a temperature of 300 K and a pressure of 150 kPa. Calculate the volume of the gas when the temperature is increased to 450 K and the pressure is decreased to 100 kPa.
(e) Thabo observes that when he sprays air freshener in one corner of a room, the smell spreads throughout the entire room within a few minutes. Use the kinetic molecular theory to explain this observation.
Question 12
(a) Distinguish between the terms intermolecular forces and intramolecular forces.
(b) Arrange the three states of matter (solid, liquid, gas) in order of increasing particle kinetic energy.
(c) A rigid sealed container holds 0.5 moles of helium gas at 27 °C and a pressure of 200 kPa. Calculate the volume of the container. (Use the ideal gas constant R = 8.31 J·mol⁻¹·K⁻¹)
(d) Nombuso has two balloons of equal size filled with different gases at the same temperature and pressure. Balloon A contains hydrogen gas (molar mass = 2 g·mol⁻¹) and Balloon B contains carbon dioxide gas (molar mass = 44 g·mol⁻¹). She releases both balloons simultaneously. Predict which gas particles have a higher average speed and explain your answer using principles of the kinetic molecular theory.
SECTION C: DATA ANALYSIS AND GRAPHING
(Total: 15 marks)
Question 13
Lerato investigates the relationship between the volume and pressure of a fixed mass of nitrogen gas at constant temperature. She records the following data:
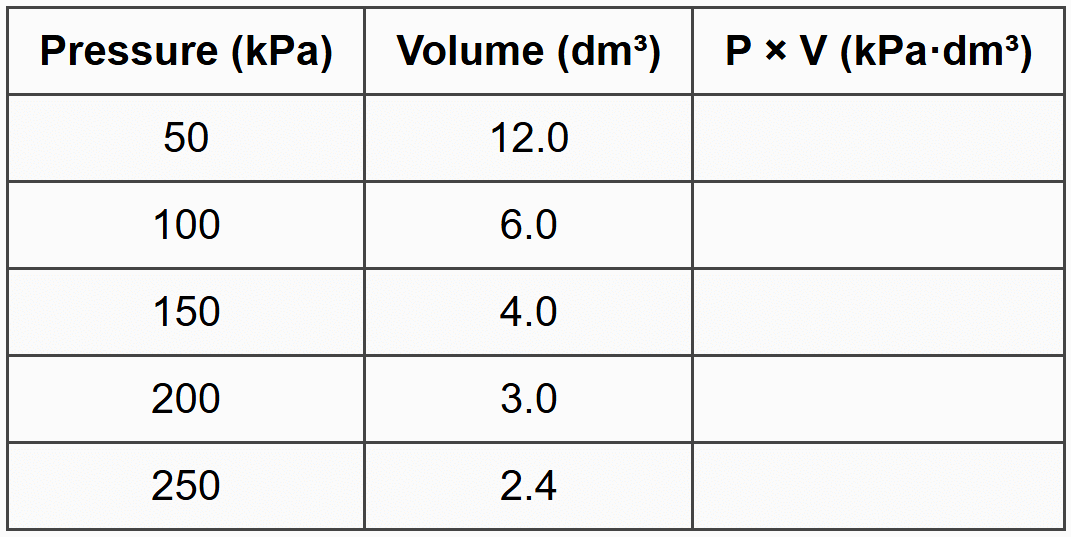
(a) Complete the third column of the table by calculating the product of pressure and volume for each measurement.
(b) State the gas law that is being investigated in this experiment.
(c) What conclusion can you draw from the calculated values in the third column? Explain what this tells you about the relationship between pressure and volume.
(d) Predict the volume of the gas if the pressure is increased to 300 kPa, assuming the temperature remains constant. Show all working.
SECTION D: SCIENTIFIC INVESTIGATION
(Total: 15 marks)
Question 14
Sipho designs an investigation to determine how temperature affects the volume of a gas at constant pressure. He takes a sealed syringe containing 50 cm³ of air and heats it in a water bath. He records the temperature of the water and the new volume reading on the syringe at different temperatures. He keeps the pressure constant by allowing the plunger to move freely. His results are shown below:
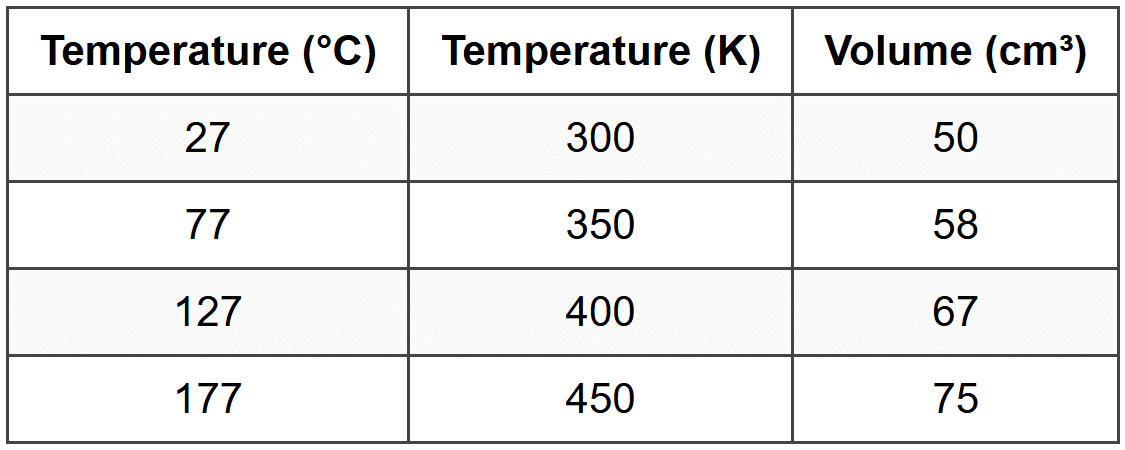
(a) Identify the independent variable in this investigation.
(b) Identify the dependent variable in this investigation.
(c) Name one controlled variable in this investigation and explain why it must be kept constant.
(d) Write a suitable hypothesis for this investigation using the format: If [condition], then [expected result], because [scientific reason].
(e) Use the data in the table to evaluate whether the hypothesis is supported. Calculate the ratio of volume to temperature (in Kelvin) for each measurement and draw a conclusion about the relationship between temperature and volume. State whether Sipho's results support Charles's Law.
GRAND TOTAL: 70
ANSWER KEY
Dear student, well done on completing the worksheet! Before you check your answers, make sure you have attempted every question to the best of your ability. Use this answer key to learn from your mistakes and understand the scientific reasoning behind each answer. Remember, making mistakes is part of the learning process!
SECTION A - Question 1
Answer: B - in constant random motion and collide with each other and the container walls
This is correct because the kinetic molecular theory describes gas particles as being in continuous, random motion. They move freely and collide elastically with each other and with the walls of their container, which is what causes gas pressure.
SECTION A - Question 2
Answer: B - Kelvin (K)
The Kelvin scale is the SI unit for temperature and must be used in all gas law calculations. Remember to convert degrees Celsius to Kelvin by adding 273: K = °C + 273.
SECTION A - Question 3
Answer: A - 1.25 m³
This question tests Boyle's Law (P₁V₁ = P₂V₂). When pressure doubles from 100 kPa to 200 kPa at constant temperature, volume halves from 2.5 m³ to 1.25 m³. Pressure and volume are inversely proportional.
SECTION A - Question 4
Answer: C - Solid
Solids have the strongest intermolecular forces, which hold particles in fixed positions. This is why solids have a definite shape and volume. Gases have the weakest intermolecular forces, and liquids are in between.
SECTION A - Question 5
Answer: B - increases by a factor of 1.33
Using Charles's Law, we convert to Kelvin: 27 °C = 300 K and 127 °C = 400 K. The ratio is 400/300 = 1.33, so volume increases by a factor of 1.33 when temperature increases from 300 K to 400 K at constant pressure.
SECTION A - Question 6
Answer: C - increases
Temperature is a measure of the average kinetic energy of particles. When temperature increases, the particles move faster, meaning their average kinetic energy increases. This is a fundamental principle of the kinetic molecular theory.
SECTION A - Question 7
Answer: A - 458 cm³
Convert to Kelvin: 25 °C = 298 K and 0 °C = 273 K. Using Charles's Law: V₂ = V₁ × (T₂/T₁) = 500 × (273/298) = 458 cm³. Volume decreases when temperature decreases at constant pressure.
SECTION A - Question 8
Answer: B - diffusion
Diffusion is the net movement of particles from an area of higher concentration to an area of lower concentration. This process occurs because particles are in constant random motion, as explained by the kinetic molecular theory.
SECTION A - Question 9
Answer: B - 22.4 dm³
At STP (standard temperature of 273 K and standard pressure of 101.3 kPa), one mole of any ideal gas occupies a volume of 22.4 dm³. This is a very important constant to remember for gas calculations.
SECTION A - Question 10
Answer: C - 67.2 dm³
Since one mole occupies 22.4 dm³ at STP, three moles occupy 3 × 22.4 = 67.2 dm³. This is a direct proportional relationship between the number of moles and volume at constant temperature and pressure.
SECTION B - Question 11(a)
The kinetic molecular theory is a model that explains the behaviour of matter in terms of particles (atoms or molecules) that are in constant motion. It describes the properties of gases, liquids, and solids based on the movement and arrangement of their particles and the forces between them.
SECTION B - Question 11(b)
Two assumptions of the kinetic molecular theory are:
1. Gas particles are in constant, random motion and move in straight lines until they collide with each other or the walls of the container.
2. The volume of the individual gas particles is negligible compared to the total volume of the gas (or: collisions between gas particles are perfectly elastic, meaning no kinetic energy is lost during collisions).
SECTION B - Question 11(c)
When the temperature of a gas in a closed rigid container increases, the average kinetic energy of the gas particles increases. This means the particles move faster and collide with the container walls more frequently and with greater force. Since the volume is constant (rigid container), the increased collision rate and force result in an increase in pressure.
SECTION B - Question 11(d)
Formula: Combined Gas Law: \( \frac{P_1 V_1}{T_1} = \frac{P_2 V_2}{T_2} \)
Substitution:
\( \frac{(150 \text{ kPa})(4.0 \text{ dm}^3)}{300 \text{ K}} = \frac{(100 \text{ kPa}) V_2}{450 \text{ K}} \)
Working:
\( \frac{600 \text{ kPa·dm}^3}{300 \text{ K}} = \frac{100 \text{ kPa} \times V_2}{450 \text{ K}} \)
\( 2 \text{ kPa·dm}^3·\text{K}^{-1} = \frac{100 \text{ kPa} \times V_2}{450 \text{ K}} \)
\( V_2 = \frac{2 \text{ kPa·dm}^3·\text{K}^{-1} \times 450 \text{ K}}{100 \text{ kPa}} \)
\( V_2 = \frac{900 \text{ kPa·dm}^3}{100 \text{ kPa}} \)
\( V_2 = 9.0 \text{ dm}^3 \)
Final Answer: 9.0 dm³ (The unit dm³ is essential for full marks.)
SECTION B - Question 11(e)
According to the kinetic molecular theory, gas particles (in this case, the air freshener molecules) are in constant, random motion. When Thabo sprays the air freshener, the particles move randomly in all directions and spread out from the area of high concentration (where it was sprayed) to areas of low concentration throughout the room. This process is called diffusion, and it continues until the air freshener molecules are evenly distributed. The random motion of particles explains why the smell eventually reaches every part of the room.
SECTION B - Question 12(a)
Intermolecular forces are forces of attraction that exist between different molecules. They are relatively weak and determine properties like boiling point and melting point. Intramolecular forces are forces of attraction that exist within a molecule, holding the atoms together (these are chemical bonds such as covalent or ionic bonds). Intramolecular forces are much stronger than intermolecular forces.
SECTION B - Question 12(b)
In order of increasing particle kinetic energy:
Solid < liquid=""><>
Solid particles vibrate in fixed positions and have the lowest kinetic energy. Liquid particles move more freely and have intermediate kinetic energy. Gas particles move rapidly and randomly and have the highest kinetic energy.
SECTION B - Question 12(c)
Formula: Ideal Gas Law: \( PV = nRT \)
Rearranged: \( V = \frac{nRT}{P} \)
Conversion: T = 27 °C = 27 + 273 = 300 K
P = 200 kPa = 200 000 Pa (must convert to Pa because R is in J·mol⁻¹·K⁻¹ and 1 Pa = 1 J·m⁻³)
Substitution:
\( V = \frac{(0.5 \text{ mol})(8.31 \text{ J·mol}^{-1}·\text{K}^{-1})(300 \text{ K})}{200\,000 \text{ Pa}} \)
Working:
\( V = \frac{0.5 \times 8.31 \times 300}{200\,000} \text{ m}^3 \)
\( V = \frac{1246.5}{200\,000} \text{ m}^3 \)
\( V = 0.00623 \text{ m}^3 \)
\( V = 6.23 \text{ dm}^3 \) (since 1 m³ = 1000 dm³)
Final Answer: 6.23 dm³ or 0.00623 m³ (Either unit is acceptable, but the unit must be included for full marks.)
SECTION B - Question 12(d)
The hydrogen gas particles in Balloon A have a higher average speed. According to the kinetic molecular theory, at the same temperature, all gas particles have the same average kinetic energy. Kinetic energy is given by \( KE = \frac{1}{2}mv^2 \), where m is mass and v is speed. Since hydrogen molecules have a much smaller molar mass (2 g·mol⁻¹) compared to carbon dioxide molecules (44 g·mol⁻¹), and the kinetic energy is the same, the hydrogen molecules must move at a higher speed to compensate for their smaller mass. Lighter gas particles move faster than heavier gas particles at the same temperature.
SECTION C - Question 13(a)
Completed third column:
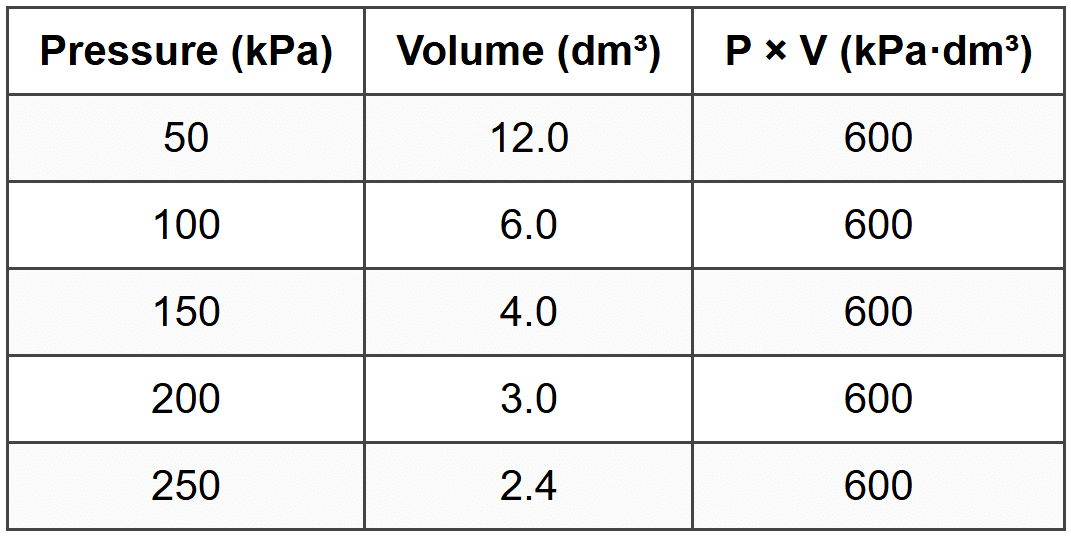
(Award 1 mark for all five values correct.)
SECTION C - Question 13(b)
Boyle's Law is being investigated.
(This law states that the pressure and volume of a fixed mass of gas are inversely proportional at constant temperature, or P₁V₁ = P₂V₂.)
SECTION C - Question 13(c)
The calculated values in the third column are all constant (600 kPa·dm³). This shows that the product of pressure and volume remains constant. This demonstrates that pressure and volume are inversely proportional: when pressure increases, volume decreases proportionally, and vice versa. This confirms Boyle's Law, which states that for a fixed mass of gas at constant temperature, PV = constant.
(Full marks: student states that P × V is constant AND explains that this shows an inverse relationship. Partial marks: only stating P × V is constant without explaining the relationship.)
SECTION C - Question 13(d)
Formula: Boyle's Law: \( P_1 V_1 = P_2 V_2 \) or PV = constant
From the table, the constant = 600 kPa·dm³
Substitution:
\( (300 \text{ kPa}) V_2 = 600 \text{ kPa·dm}^3 \)
Working:
\( V_2 = \frac{600 \text{ kPa·dm}^3}{300 \text{ kPa}} \)
\( V_2 = 2.0 \text{ dm}^3 \)
Final Answer: 2.0 dm³ (Unit is required for full marks.)
SECTION D - Question 14(a)
The independent variable is temperature (in °C or K).
This is the variable that Sipho deliberately changes or controls during the investigation by heating the water bath to different temperatures.
SECTION D - Question 14(b)
The dependent variable is volume (in cm³).
This is the variable that responds to the change in temperature. Sipho measures how the volume of the gas changes as he changes the temperature.
SECTION D - Question 14(c)
One controlled variable is pressure (or: the mass/amount of gas, or: the type of gas).
Pressure must be kept constant because the investigation is testing the relationship between temperature and volume only (Charles's Law). If pressure also changed, Sipho would not know whether changes in volume were due to temperature or pressure, making the results unreliable and the investigation invalid.
SECTION D - Question 14(d)
Model Hypothesis:
If the temperature of the gas increases, then the volume of the gas will increase proportionally, because according to the kinetic molecular theory, increasing temperature increases the average kinetic energy of the gas particles, causing them to move faster and occupy a larger volume at constant pressure.
(Award full marks if the hypothesis follows the correct format and includes a valid scientific reason related to particle motion or kinetic molecular theory.)
SECTION D - Question 14(e)
Ratio Calculations:
At 300 K: V/T = 50/300 = 0.167 cm³·K⁻¹
At 350 K: V/T = 58/350 = 0.166 cm³·K⁻¹
At 400 K: V/T = 67/400 = 0.168 cm³·K⁻¹
At 450 K: V/T = 75/450 = 0.167 cm³·K⁻¹
Conclusion:
The ratio of volume to temperature (V/T) remains approximately constant (0.167 cm³·K⁻¹) for all measurements. This shows that volume is directly proportional to temperature when pressure and the amount of gas are kept constant. Sipho's results support Charles's Law, which states that V/T = constant for a fixed mass of gas at constant pressure. The hypothesis is supported because the data clearly shows that as temperature increases, volume increases proportionally.
(Full marks: student calculates at least three V/T ratios, states they are constant, identifies Charles's Law, and explicitly states the hypothesis is supported with reference to the proportional relationship. Partial marks: identifies the proportional relationship without calculating ratios, or calculates ratios without drawing a full conclusion.)
Mark Allocation Summary Table
Congratulations on completing this worksheet! Review any questions you found challenging and make sure you understand the kinetic molecular theory and gas laws thoroughly. Keep practicing and you will master these concepts!





