Grade 10 Exam > Grade 10 Notes > Physical Sciences > Physical and Chemical Change
Physical and Chemical Change
# Grade 10 Physical Sciences Worksheet ## SECTION A: MULTIPLE CHOICE (Total: 10 marks) **1.** Which of the following is an example of a chemical change?
A. Ice melting into water
B. Iron rusting in moist air
C. Sugar dissolving in tea
D. Wax melting in a candle **2.** During a physical change:
A. New substances with different properties are formed
B. The composition of the substance remains the same
C. Chemical bonds are broken and new bonds are formed
D. The change is always irreversible **3.** Thabo heats 50 g of water from 20 °C to 80 °C. The specific heat capacity of water is 4.18 J·g⁻¹·°C⁻¹. How much energy is required?
A. 12 540 J
B. 4 180 J
C. 20 900 J
D. 25 080 J **4.** Which of the following observations indicates that a chemical change has occurred?
A. A substance changes state from solid to liquid
B. A gas is produced when two solutions are mixed
C. Salt crystals become smaller when stirred in water
D. A metal bar expands when heated **5.** The process of photosynthesis in plants is an example of:
A. A physical change because the plant grows larger
B. A chemical change because new substances are formed
C. A physical change because water is absorbed
D. Neither a physical nor a chemical change **6.** When magnesium ribbon burns in air, 2.4 g of magnesium combines with 1.6 g of oxygen to form magnesium oxide. What is the total mass of magnesium oxide produced?
A. 0.8 g
B. 2.4 g
C. 4.0 g
D. 3.2 g **7.** Which statement best describes a reversible change?
A. The original substance can be recovered by physical means
B. New chemical bonds are always formed
C. Energy is always released during the change
D. The change can only occur at high temperatures **8.** Lerato observes that when she crushes a rock into powder, the total mass remains the same. This observation supports:
A. The law of conservation of energy
B. The law of conservation of mass
C. The law of definite proportions
D. The law of multiple proportions **9.** Which of the following changes is exothermic?
A. Ice melting
B. Water evaporating
C. Water freezing
D. Salt dissolving in water **10.** A student heats copper carbonate and observes a color change from green to black, and a gas is released. How many indicators of a chemical change are observed?
A. One
B. Two
C. Three
D. Four ## SECTION B: STRUCTURED QUESTIONS (Total: 30 marks) ### Question 11 Sipho investigates the burning of a candle. He observes that the solid wax melts near the wick, the liquid wax is drawn up the wick, and then the wax vapor burns in the flame producing carbon dioxide and water vapor. **(a)** Identify one physical change that occurs during the burning of the candle. **(b)** Identify one chemical change that occurs during the burning of the candle. **(c)** Explain why the melting of wax is classified as a physical change. **(d)** The candle has a mass of 150 g before burning. After burning for 2 hours, the mass is 120 g. Calculate the mass of wax that was converted to gaseous products. **(e)** Sipho claims that matter was destroyed because the candle became lighter. Evaluate this claim using the law of conservation of mass. ### Question 12 Zanele conducts an experiment where she adds 5.0 g of zinc metal to 50 cm³ of dilute hydrochloric acid in a beaker. She observes bubbles forming, the beaker becomes warm, and after the reaction stops, no solid zinc remains. The solution in the beaker is now clear and colorless. **(a)** List three observations that indicate a chemical change occurred. **(b)** Name the type of energy change that occurred during this reaction. **(c)** Calculate the total mass of the products if the mass of the hydrochloric acid solution was 52.5 g. Assume no gases escape. **(d)** Zanele repeats the experiment but uses zinc powder instead of a zinc strip. Predict and explain how this change would affect the rate of the reaction. ## SECTION C: DATA ANALYSIS AND GRAPHING (Total: 15 marks) ### Question 13 Nomsa investigates the temperature change when different masses of ammonium nitrate dissolve in 100 cm³ of water at room temperature (25 °C). She records her results in the table below: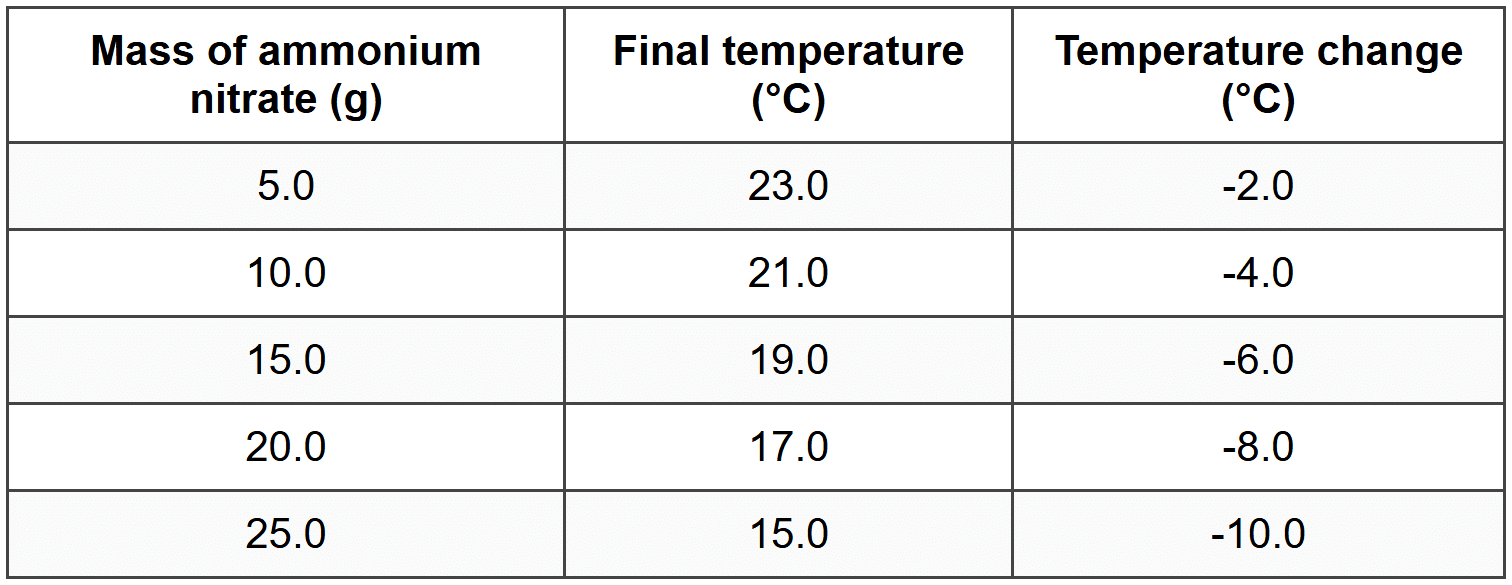 **(a)** Is the dissolving of ammonium nitrate in water a physical or chemical change? Justify your answer. **(b)** What type of energy change is observed in this investigation? **(c)** Describe the relationship between the mass of ammonium nitrate dissolved and the temperature change. **(d)** Calculate the total energy absorbed by the water when 25.0 g of ammonium nitrate dissolves, if the mass of water is 100 g and the specific heat capacity of water is 4.18 J·g⁻¹·°C⁻¹. **(e)** Nomsa's friend suggests that this process could be used to make an instant cold pack for sports injuries. Evaluate whether this is a practical application based on the data provided. ## SECTION D: SCIENTIFIC INVESTIGATION (Total: 15 marks) ### Question 14 Bongani investigates whether the size of marble chips (calcium carbonate) affects the rate at which carbon dioxide gas is produced when reacted with dilute hydrochloric acid. He uses three different sizes of marble chips: large chips, medium chips, and small chips. For each experiment, he uses 10 g of marble chips and 100 cm³ of 1.0 mol·dm⁻³ hydrochloric acid at 25 °C. He measures the volume of carbon dioxide gas produced every 30 seconds. His results show that small chips produce 240 cm³ of gas in 2 minutes, medium chips produce 240 cm³ of gas in 4 minutes, and large chips produce 240 cm³ of gas in 7 minutes. **(a)** Identify the independent variable in this investigation. **(b)** Identify the dependent variable in this investigation. **(c)** Name one variable that Bongani controlled in this investigation and explain why it must be kept constant. **(d)** Write a hypothesis for this investigation using the format: If [condition], then [expected result], because [scientific reason]. **(e)** Based on the results provided, state whether the hypothesis is supported. Justify your conclusion with reference to the data. **(f)** Explain, using collision theory, why small marble chips produce gas faster than large marble chips. **GRAND TOTAL: 70** --- ## ANSWER KEY Dear student, Well done on completing this worksheet! Remember to attempt all questions before checking the answers. Use this answer key to identify areas where you did well and topics you may need to revise. Pay special attention to the worked solutions for calculations - they show you the exact format expected in tests and exams. Keep up the excellent work! --- ### SECTION A - Question 1 **Answer: B. Iron rusting in moist air** Iron rusting is a chemical change because iron reacts with oxygen and water to form a new substance called iron oxide (rust), which has different properties from the original iron metal. --- ### SECTION A - Question 2 **Answer: B. The composition of the substance remains the same** During a physical change, only the physical properties such as shape, size, or state may change, but the chemical composition and identity of the substance stay exactly the same. No new substances are formed. --- ### SECTION A - Question 3 **Answer: A. 12 540 J** This question requires you to calculate energy using the formula \( Q = m \times c \times \Delta T \). Step 1: Formula
**(a)** Is the dissolving of ammonium nitrate in water a physical or chemical change? Justify your answer. **(b)** What type of energy change is observed in this investigation? **(c)** Describe the relationship between the mass of ammonium nitrate dissolved and the temperature change. **(d)** Calculate the total energy absorbed by the water when 25.0 g of ammonium nitrate dissolves, if the mass of water is 100 g and the specific heat capacity of water is 4.18 J·g⁻¹·°C⁻¹. **(e)** Nomsa's friend suggests that this process could be used to make an instant cold pack for sports injuries. Evaluate whether this is a practical application based on the data provided. ## SECTION D: SCIENTIFIC INVESTIGATION (Total: 15 marks) ### Question 14 Bongani investigates whether the size of marble chips (calcium carbonate) affects the rate at which carbon dioxide gas is produced when reacted with dilute hydrochloric acid. He uses three different sizes of marble chips: large chips, medium chips, and small chips. For each experiment, he uses 10 g of marble chips and 100 cm³ of 1.0 mol·dm⁻³ hydrochloric acid at 25 °C. He measures the volume of carbon dioxide gas produced every 30 seconds. His results show that small chips produce 240 cm³ of gas in 2 minutes, medium chips produce 240 cm³ of gas in 4 minutes, and large chips produce 240 cm³ of gas in 7 minutes. **(a)** Identify the independent variable in this investigation. **(b)** Identify the dependent variable in this investigation. **(c)** Name one variable that Bongani controlled in this investigation and explain why it must be kept constant. **(d)** Write a hypothesis for this investigation using the format: If [condition], then [expected result], because [scientific reason]. **(e)** Based on the results provided, state whether the hypothesis is supported. Justify your conclusion with reference to the data. **(f)** Explain, using collision theory, why small marble chips produce gas faster than large marble chips. **GRAND TOTAL: 70** --- ## ANSWER KEY Dear student, Well done on completing this worksheet! Remember to attempt all questions before checking the answers. Use this answer key to identify areas where you did well and topics you may need to revise. Pay special attention to the worked solutions for calculations - they show you the exact format expected in tests and exams. Keep up the excellent work! --- ### SECTION A - Question 1 **Answer: B. Iron rusting in moist air** Iron rusting is a chemical change because iron reacts with oxygen and water to form a new substance called iron oxide (rust), which has different properties from the original iron metal. --- ### SECTION A - Question 2 **Answer: B. The composition of the substance remains the same** During a physical change, only the physical properties such as shape, size, or state may change, but the chemical composition and identity of the substance stay exactly the same. No new substances are formed. --- ### SECTION A - Question 3 **Answer: A. 12 540 J** This question requires you to calculate energy using the formula \( Q = m \times c \times \Delta T \). Step 1: Formula
\( Q = m \times c \times \Delta T \) Step 2: Substitute values
\( Q = 50 \, \text{g} \times 4.18 \, \text{J·g}^{-1}\text{·°C}^{-1} \times (80 - 20) \, \text{°C} \) Step 3: Calculate
\( Q = 50 \times 4.18 \times 60 \)
\( Q = 12\,540 \, \text{J} \) Step 4: Final answer
\( Q = 12\,540 \, \text{J} \) --- ### SECTION A - Question 4 **Answer: B. A gas is produced when two solutions are mixed** The production of a gas is a key indicator that a chemical reaction (chemical change) has occurred, because it shows that new substances are being formed. --- ### SECTION A - Question 5 **Answer: B. A chemical change because new substances are formed** Photosynthesis is a chemical change because carbon dioxide and water are chemically transformed into glucose and oxygen, which are entirely new substances with different chemical properties. --- ### SECTION A - Question 6 **Answer: C. 4.0 g** According to the law of conservation of mass, matter cannot be created or destroyed in a chemical reaction. Step 1: Apply the law of conservation of mass
Total mass of products = Total mass of reactants Step 2: Substitute values
Mass of magnesium oxide = 2.4 g + 1.6 g Step 3: Calculate
Mass of magnesium oxide = 4.0 g Step 4: Final answer
Mass of magnesium oxide = 4.0 g --- ### SECTION A - Question 7 **Answer: A. The original substance can be recovered by physical means** A reversible change is typically a physical change where the original substance can be recovered by simple physical methods such as cooling, heating, or evaporation, without any chemical reaction. --- ### SECTION A - Question 8 **Answer: B. The law of conservation of mass** Crushing a rock changes its physical form (size and shape) but does not change the total mass. This observation supports the law of conservation of mass, which states that mass is neither created nor destroyed during a physical change. --- ### SECTION A - Question 9 **Answer: C. Water freezing** Water freezing is an exothermic change because energy is released to the surroundings when liquid water changes to solid ice. The other options are endothermic processes that absorb energy from the surroundings. --- ### SECTION A - Question 10 **Answer: B. Two** The two indicators of a chemical change observed are: (1) a color change from green to black, and (2) the release of a gas. These indicate that new substances with different properties have been formed. --- ### SECTION B - Question 11(a) **Answer:** The melting of solid wax into liquid wax is a physical change. (Or: The evaporation of liquid wax into wax vapor is a physical change.) Both melting and evaporation are physical changes because the chemical composition of the wax remains the same - only the state changes from solid to liquid or liquid to gas. --- ### SECTION B - Question 11(b) **Answer:** The burning of wax vapor in the flame to produce carbon dioxide and water vapor is a chemical change. This is a chemical change because the wax reacts with oxygen to form new substances (carbon dioxide and water) that have completely different properties from the original wax. --- ### SECTION B - Question 11(c) **Answer:** The melting of wax is classified as a physical change because the wax changes state from solid to liquid, but its chemical composition remains the same. No new substances are formed, and the process is reversible - the liquid wax can solidify again when cooled. --- ### SECTION B - Question 11(d) Step 1: Formula or concept
Mass of wax converted = Initial mass - Final mass Step 2: Substitute values
Mass of wax converted = 150 g - 120 g Step 3: Calculate
Mass of wax converted = 30 g Step 4: Final answer
Mass of wax converted = 30 g *Note: The unit (g) must be included for full marks.* --- ### SECTION B - Question 11(e) **Answer:** Sipho's claim is incorrect. According to the law of conservation of mass, matter cannot be created or destroyed during a chemical reaction. The 30 g of wax that "disappeared" was not destroyed - it was converted into gaseous products (carbon dioxide and water vapor) that escaped into the air. If the gases were captured and measured, the total mass of all products would equal the mass of the reactants. *This answer earns full marks by: (1) stating the claim is incorrect, (2) referring to the law of conservation of mass, and (3) explaining where the "missing" mass went.* --- ### SECTION B - Question 12(a) **Answer:** Three observations that indicate a chemical change:
Total mass of products = Total mass of reactants Step 2: Substitute values
Total mass of products = mass of zinc + mass of hydrochloric acid solution
Total mass of products = 5.0 g + 52.5 g Step 3: Calculate
Total mass of products = 57.5 g Step 4: Final answer
Total mass of products = 57.5 g *Note: This calculation assumes no gases escape. If gases escape, the measured mass would be less, but the actual total mass (including escaped gas) remains 57.5 g.* --- ### SECTION B - Question 12(d) **Answer:** Using zinc powder instead of a zinc strip would increase the rate of the reaction (the reaction would occur faster). This is because zinc powder has a much larger surface area than a zinc strip. A larger surface area means more zinc particles are exposed to the hydrochloric acid at the same time, so more collisions occur between reactant particles per second, increasing the reaction rate. *Full marks awarded for: (1) stating the rate increases, and (2) explaining the role of increased surface area in increasing collision frequency.* --- ### SECTION C - Question 13(a) **Answer:** The dissolving of ammonium nitrate in water is a physical change. Although energy is absorbed (the temperature drops), no new chemical substances are formed - the ammonium nitrate can be recovered by evaporating the water. The process involves breaking ionic bonds in the solid and forming interactions with water molecules, but the chemical identity of ammonium nitrate remains unchanged. *Full marks for stating it is a physical change and providing a clear justification.* --- ### SECTION C - Question 13(b) **Answer:** The energy change is endothermic. The temperature of the water decreases (negative temperature change), which means energy is absorbed from the surroundings during the dissolving process. --- ### SECTION C - Question 13(c) **Answer:** There is a direct or linear relationship between the mass of ammonium nitrate dissolved and the temperature change. As the mass of ammonium nitrate increases, the temperature change becomes more negative (the temperature drops more). Specifically, for every 5.0 g increase in mass, the temperature drops by an additional 2.0 °C. *Full marks for identifying the relationship and describing the pattern in the data.* --- ### SECTION C - Question 13(d) Step 1: Formula
\( Q = m \times c \times \Delta T \) Step 2: Substitute values with units
\( Q = 100 \, \text{g} \times 4.18 \, \text{J·g}^{-1}\text{·°C}^{-1} \times (-10.0) \, \text{°C} \) Step 3: Calculate
\( Q = 100 \times 4.18 \times (-10.0) \)
\( Q = -4\,180 \, \text{J} \) Step 4: Final answer
\( Q = -4\,180 \, \text{J} \) or \( 4\,180 \, \text{J} \) absorbed *The negative sign indicates energy is absorbed by the dissolving process from the water. Stating "4 180 J absorbed" is also acceptable. The unit J must be included for full marks.* --- ### SECTION C - Question 13(e) **Answer:** This is a practical application. The data shows that dissolving 25.0 g of ammonium nitrate in 100 g of water causes a temperature drop of 10 °C, which is significant enough to provide a cooling effect for treating sports injuries. Instant cold packs work on this principle - when the inner pouch is broken, ammonium nitrate dissolves in water, absorbing energy and creating a cold sensation. The process is quick, portable, and does not require refrigeration, making it very practical for first aid use. *Full marks for: (1) stating it is practical, (2) referring to the data (temperature drop of 10 °C), and (3) explaining how this relates to the real-world application.* --- ### SECTION D - Question 14(a) **Answer:** The independent variable is the size of the marble chips (or particle size of calcium carbonate). The independent variable is the one that Bongani deliberately changed to observe its effect on the rate of reaction. --- ### SECTION D - Question 14(b) **Answer:** The dependent variable is the rate at which carbon dioxide gas is produced (or the volume of gas produced over time, or the time taken to produce a certain volume of gas). The dependent variable is what Bongani measured to see the effect of changing the marble chip size. --- ### SECTION D - Question 14(c) **Answer (example):** One controlled variable is the concentration of hydrochloric acid (1.0 mol·dm⁻³). This must be kept constant because changing the concentration would change the number of acid particles available to react, which would affect the rate of the reaction independently of the marble chip size, making it impossible to fairly compare the effect of chip size alone. *Other acceptable controlled variables include: mass of marble chips (10 g), volume of acid (100 cm³), or temperature (25 °C). Full marks for naming the variable and explaining why it must be controlled.* --- ### SECTION D - Question 14(d) **Answer (model hypothesis):** If the size of marble chips decreases, then the rate of carbon dioxide production will increase, because smaller chips have a larger total surface area exposed to the acid, which increases the frequency of collisions between reactant particles. *Full marks for a correctly formatted hypothesis that includes: condition (chip size decreases), expected result (rate increases), and scientific reason (surface area and collision frequency).* --- ### SECTION D - Question 14(e) **Answer:** The hypothesis is supported by the data. The results show that small chips produced 240 cm³ of gas in 2 minutes, medium chips took 4 minutes, and large chips took 7 minutes to produce the same volume of gas. This clearly demonstrates that smaller marble chips react faster (higher rate) than larger chips, which matches the prediction in the hypothesis. All three sizes produced the same total volume of gas, but the time taken decreased as chip size decreased. *Full marks for: (1) stating the hypothesis is supported, (2) quoting specific data values, and (3) linking the data to the hypothesis prediction.* --- ### SECTION D - Question 14(f) **Answer:** According to collision theory, a chemical reaction occurs when reactant particles collide with sufficient energy. Small marble chips have a much larger total surface area compared to large chips of the same mass. This means more calcium carbonate particles are exposed to the hydrochloric acid at any given time, which increases the number of collisions between acid particles and carbonate particles per second. More collisions per second result in a faster rate of reaction, so gas is produced more quickly with small chips than with large chips. *Full marks for: (1) mentioning collision theory or collisions, (2) explaining the role of surface area, and (3) linking increased collisions to increased reaction rate.* --- ## Mark Allocation Summary --- **End of Answer Key** Keep practicing, stay curious, and remember that understanding the difference between physical and chemical changes is fundamental to all of chemistry. You're doing great!
--- **End of Answer Key** Keep practicing, stay curious, and remember that understanding the difference between physical and chemical changes is fundamental to all of chemistry. You're doing great!
A. Ice melting into water
B. Iron rusting in moist air
C. Sugar dissolving in tea
D. Wax melting in a candle **2.** During a physical change:
A. New substances with different properties are formed
B. The composition of the substance remains the same
C. Chemical bonds are broken and new bonds are formed
D. The change is always irreversible **3.** Thabo heats 50 g of water from 20 °C to 80 °C. The specific heat capacity of water is 4.18 J·g⁻¹·°C⁻¹. How much energy is required?
A. 12 540 J
B. 4 180 J
C. 20 900 J
D. 25 080 J **4.** Which of the following observations indicates that a chemical change has occurred?
A. A substance changes state from solid to liquid
B. A gas is produced when two solutions are mixed
C. Salt crystals become smaller when stirred in water
D. A metal bar expands when heated **5.** The process of photosynthesis in plants is an example of:
A. A physical change because the plant grows larger
B. A chemical change because new substances are formed
C. A physical change because water is absorbed
D. Neither a physical nor a chemical change **6.** When magnesium ribbon burns in air, 2.4 g of magnesium combines with 1.6 g of oxygen to form magnesium oxide. What is the total mass of magnesium oxide produced?
A. 0.8 g
B. 2.4 g
C. 4.0 g
D. 3.2 g **7.** Which statement best describes a reversible change?
A. The original substance can be recovered by physical means
B. New chemical bonds are always formed
C. Energy is always released during the change
D. The change can only occur at high temperatures **8.** Lerato observes that when she crushes a rock into powder, the total mass remains the same. This observation supports:
A. The law of conservation of energy
B. The law of conservation of mass
C. The law of definite proportions
D. The law of multiple proportions **9.** Which of the following changes is exothermic?
A. Ice melting
B. Water evaporating
C. Water freezing
D. Salt dissolving in water **10.** A student heats copper carbonate and observes a color change from green to black, and a gas is released. How many indicators of a chemical change are observed?
A. One
B. Two
C. Three
D. Four ## SECTION B: STRUCTURED QUESTIONS (Total: 30 marks) ### Question 11 Sipho investigates the burning of a candle. He observes that the solid wax melts near the wick, the liquid wax is drawn up the wick, and then the wax vapor burns in the flame producing carbon dioxide and water vapor. **(a)** Identify one physical change that occurs during the burning of the candle. **(b)** Identify one chemical change that occurs during the burning of the candle. **(c)** Explain why the melting of wax is classified as a physical change. **(d)** The candle has a mass of 150 g before burning. After burning for 2 hours, the mass is 120 g. Calculate the mass of wax that was converted to gaseous products. **(e)** Sipho claims that matter was destroyed because the candle became lighter. Evaluate this claim using the law of conservation of mass. ### Question 12 Zanele conducts an experiment where she adds 5.0 g of zinc metal to 50 cm³ of dilute hydrochloric acid in a beaker. She observes bubbles forming, the beaker becomes warm, and after the reaction stops, no solid zinc remains. The solution in the beaker is now clear and colorless. **(a)** List three observations that indicate a chemical change occurred. **(b)** Name the type of energy change that occurred during this reaction. **(c)** Calculate the total mass of the products if the mass of the hydrochloric acid solution was 52.5 g. Assume no gases escape. **(d)** Zanele repeats the experiment but uses zinc powder instead of a zinc strip. Predict and explain how this change would affect the rate of the reaction. ## SECTION C: DATA ANALYSIS AND GRAPHING (Total: 15 marks) ### Question 13 Nomsa investigates the temperature change when different masses of ammonium nitrate dissolve in 100 cm³ of water at room temperature (25 °C). She records her results in the table below:
 **(a)** Is the dissolving of ammonium nitrate in water a physical or chemical change? Justify your answer. **(b)** What type of energy change is observed in this investigation? **(c)** Describe the relationship between the mass of ammonium nitrate dissolved and the temperature change. **(d)** Calculate the total energy absorbed by the water when 25.0 g of ammonium nitrate dissolves, if the mass of water is 100 g and the specific heat capacity of water is 4.18 J·g⁻¹·°C⁻¹. **(e)** Nomsa's friend suggests that this process could be used to make an instant cold pack for sports injuries. Evaluate whether this is a practical application based on the data provided. ## SECTION D: SCIENTIFIC INVESTIGATION (Total: 15 marks) ### Question 14 Bongani investigates whether the size of marble chips (calcium carbonate) affects the rate at which carbon dioxide gas is produced when reacted with dilute hydrochloric acid. He uses three different sizes of marble chips: large chips, medium chips, and small chips. For each experiment, he uses 10 g of marble chips and 100 cm³ of 1.0 mol·dm⁻³ hydrochloric acid at 25 °C. He measures the volume of carbon dioxide gas produced every 30 seconds. His results show that small chips produce 240 cm³ of gas in 2 minutes, medium chips produce 240 cm³ of gas in 4 minutes, and large chips produce 240 cm³ of gas in 7 minutes. **(a)** Identify the independent variable in this investigation. **(b)** Identify the dependent variable in this investigation. **(c)** Name one variable that Bongani controlled in this investigation and explain why it must be kept constant. **(d)** Write a hypothesis for this investigation using the format: If [condition], then [expected result], because [scientific reason]. **(e)** Based on the results provided, state whether the hypothesis is supported. Justify your conclusion with reference to the data. **(f)** Explain, using collision theory, why small marble chips produce gas faster than large marble chips. **GRAND TOTAL: 70** --- ## ANSWER KEY Dear student, Well done on completing this worksheet! Remember to attempt all questions before checking the answers. Use this answer key to identify areas where you did well and topics you may need to revise. Pay special attention to the worked solutions for calculations - they show you the exact format expected in tests and exams. Keep up the excellent work! --- ### SECTION A - Question 1 **Answer: B. Iron rusting in moist air** Iron rusting is a chemical change because iron reacts with oxygen and water to form a new substance called iron oxide (rust), which has different properties from the original iron metal. --- ### SECTION A - Question 2 **Answer: B. The composition of the substance remains the same** During a physical change, only the physical properties such as shape, size, or state may change, but the chemical composition and identity of the substance stay exactly the same. No new substances are formed. --- ### SECTION A - Question 3 **Answer: A. 12 540 J** This question requires you to calculate energy using the formula \( Q = m \times c \times \Delta T \). Step 1: Formula
**(a)** Is the dissolving of ammonium nitrate in water a physical or chemical change? Justify your answer. **(b)** What type of energy change is observed in this investigation? **(c)** Describe the relationship between the mass of ammonium nitrate dissolved and the temperature change. **(d)** Calculate the total energy absorbed by the water when 25.0 g of ammonium nitrate dissolves, if the mass of water is 100 g and the specific heat capacity of water is 4.18 J·g⁻¹·°C⁻¹. **(e)** Nomsa's friend suggests that this process could be used to make an instant cold pack for sports injuries. Evaluate whether this is a practical application based on the data provided. ## SECTION D: SCIENTIFIC INVESTIGATION (Total: 15 marks) ### Question 14 Bongani investigates whether the size of marble chips (calcium carbonate) affects the rate at which carbon dioxide gas is produced when reacted with dilute hydrochloric acid. He uses three different sizes of marble chips: large chips, medium chips, and small chips. For each experiment, he uses 10 g of marble chips and 100 cm³ of 1.0 mol·dm⁻³ hydrochloric acid at 25 °C. He measures the volume of carbon dioxide gas produced every 30 seconds. His results show that small chips produce 240 cm³ of gas in 2 minutes, medium chips produce 240 cm³ of gas in 4 minutes, and large chips produce 240 cm³ of gas in 7 minutes. **(a)** Identify the independent variable in this investigation. **(b)** Identify the dependent variable in this investigation. **(c)** Name one variable that Bongani controlled in this investigation and explain why it must be kept constant. **(d)** Write a hypothesis for this investigation using the format: If [condition], then [expected result], because [scientific reason]. **(e)** Based on the results provided, state whether the hypothesis is supported. Justify your conclusion with reference to the data. **(f)** Explain, using collision theory, why small marble chips produce gas faster than large marble chips. **GRAND TOTAL: 70** --- ## ANSWER KEY Dear student, Well done on completing this worksheet! Remember to attempt all questions before checking the answers. Use this answer key to identify areas where you did well and topics you may need to revise. Pay special attention to the worked solutions for calculations - they show you the exact format expected in tests and exams. Keep up the excellent work! --- ### SECTION A - Question 1 **Answer: B. Iron rusting in moist air** Iron rusting is a chemical change because iron reacts with oxygen and water to form a new substance called iron oxide (rust), which has different properties from the original iron metal. --- ### SECTION A - Question 2 **Answer: B. The composition of the substance remains the same** During a physical change, only the physical properties such as shape, size, or state may change, but the chemical composition and identity of the substance stay exactly the same. No new substances are formed. --- ### SECTION A - Question 3 **Answer: A. 12 540 J** This question requires you to calculate energy using the formula \( Q = m \times c \times \Delta T \). Step 1: Formula\( Q = m \times c \times \Delta T \) Step 2: Substitute values
\( Q = 50 \, \text{g} \times 4.18 \, \text{J·g}^{-1}\text{·°C}^{-1} \times (80 - 20) \, \text{°C} \) Step 3: Calculate
\( Q = 50 \times 4.18 \times 60 \)
\( Q = 12\,540 \, \text{J} \) Step 4: Final answer
\( Q = 12\,540 \, \text{J} \) --- ### SECTION A - Question 4 **Answer: B. A gas is produced when two solutions are mixed** The production of a gas is a key indicator that a chemical reaction (chemical change) has occurred, because it shows that new substances are being formed. --- ### SECTION A - Question 5 **Answer: B. A chemical change because new substances are formed** Photosynthesis is a chemical change because carbon dioxide and water are chemically transformed into glucose and oxygen, which are entirely new substances with different chemical properties. --- ### SECTION A - Question 6 **Answer: C. 4.0 g** According to the law of conservation of mass, matter cannot be created or destroyed in a chemical reaction. Step 1: Apply the law of conservation of mass
Total mass of products = Total mass of reactants Step 2: Substitute values
Mass of magnesium oxide = 2.4 g + 1.6 g Step 3: Calculate
Mass of magnesium oxide = 4.0 g Step 4: Final answer
Mass of magnesium oxide = 4.0 g --- ### SECTION A - Question 7 **Answer: A. The original substance can be recovered by physical means** A reversible change is typically a physical change where the original substance can be recovered by simple physical methods such as cooling, heating, or evaporation, without any chemical reaction. --- ### SECTION A - Question 8 **Answer: B. The law of conservation of mass** Crushing a rock changes its physical form (size and shape) but does not change the total mass. This observation supports the law of conservation of mass, which states that mass is neither created nor destroyed during a physical change. --- ### SECTION A - Question 9 **Answer: C. Water freezing** Water freezing is an exothermic change because energy is released to the surroundings when liquid water changes to solid ice. The other options are endothermic processes that absorb energy from the surroundings. --- ### SECTION A - Question 10 **Answer: B. Two** The two indicators of a chemical change observed are: (1) a color change from green to black, and (2) the release of a gas. These indicate that new substances with different properties have been formed. --- ### SECTION B - Question 11(a) **Answer:** The melting of solid wax into liquid wax is a physical change. (Or: The evaporation of liquid wax into wax vapor is a physical change.) Both melting and evaporation are physical changes because the chemical composition of the wax remains the same - only the state changes from solid to liquid or liquid to gas. --- ### SECTION B - Question 11(b) **Answer:** The burning of wax vapor in the flame to produce carbon dioxide and water vapor is a chemical change. This is a chemical change because the wax reacts with oxygen to form new substances (carbon dioxide and water) that have completely different properties from the original wax. --- ### SECTION B - Question 11(c) **Answer:** The melting of wax is classified as a physical change because the wax changes state from solid to liquid, but its chemical composition remains the same. No new substances are formed, and the process is reversible - the liquid wax can solidify again when cooled. --- ### SECTION B - Question 11(d) Step 1: Formula or concept
Mass of wax converted = Initial mass - Final mass Step 2: Substitute values
Mass of wax converted = 150 g - 120 g Step 3: Calculate
Mass of wax converted = 30 g Step 4: Final answer
Mass of wax converted = 30 g *Note: The unit (g) must be included for full marks.* --- ### SECTION B - Question 11(e) **Answer:** Sipho's claim is incorrect. According to the law of conservation of mass, matter cannot be created or destroyed during a chemical reaction. The 30 g of wax that "disappeared" was not destroyed - it was converted into gaseous products (carbon dioxide and water vapor) that escaped into the air. If the gases were captured and measured, the total mass of all products would equal the mass of the reactants. *This answer earns full marks by: (1) stating the claim is incorrect, (2) referring to the law of conservation of mass, and (3) explaining where the "missing" mass went.* --- ### SECTION B - Question 12(a) **Answer:** Three observations that indicate a chemical change:
- Bubbles (gas) were produced.
- The beaker became warm (energy was released).
- The solid zinc disappeared completely (a new substance was formed).
Total mass of products = Total mass of reactants Step 2: Substitute values
Total mass of products = mass of zinc + mass of hydrochloric acid solution
Total mass of products = 5.0 g + 52.5 g Step 3: Calculate
Total mass of products = 57.5 g Step 4: Final answer
Total mass of products = 57.5 g *Note: This calculation assumes no gases escape. If gases escape, the measured mass would be less, but the actual total mass (including escaped gas) remains 57.5 g.* --- ### SECTION B - Question 12(d) **Answer:** Using zinc powder instead of a zinc strip would increase the rate of the reaction (the reaction would occur faster). This is because zinc powder has a much larger surface area than a zinc strip. A larger surface area means more zinc particles are exposed to the hydrochloric acid at the same time, so more collisions occur between reactant particles per second, increasing the reaction rate. *Full marks awarded for: (1) stating the rate increases, and (2) explaining the role of increased surface area in increasing collision frequency.* --- ### SECTION C - Question 13(a) **Answer:** The dissolving of ammonium nitrate in water is a physical change. Although energy is absorbed (the temperature drops), no new chemical substances are formed - the ammonium nitrate can be recovered by evaporating the water. The process involves breaking ionic bonds in the solid and forming interactions with water molecules, but the chemical identity of ammonium nitrate remains unchanged. *Full marks for stating it is a physical change and providing a clear justification.* --- ### SECTION C - Question 13(b) **Answer:** The energy change is endothermic. The temperature of the water decreases (negative temperature change), which means energy is absorbed from the surroundings during the dissolving process. --- ### SECTION C - Question 13(c) **Answer:** There is a direct or linear relationship between the mass of ammonium nitrate dissolved and the temperature change. As the mass of ammonium nitrate increases, the temperature change becomes more negative (the temperature drops more). Specifically, for every 5.0 g increase in mass, the temperature drops by an additional 2.0 °C. *Full marks for identifying the relationship and describing the pattern in the data.* --- ### SECTION C - Question 13(d) Step 1: Formula
\( Q = m \times c \times \Delta T \) Step 2: Substitute values with units
\( Q = 100 \, \text{g} \times 4.18 \, \text{J·g}^{-1}\text{·°C}^{-1} \times (-10.0) \, \text{°C} \) Step 3: Calculate
\( Q = 100 \times 4.18 \times (-10.0) \)
\( Q = -4\,180 \, \text{J} \) Step 4: Final answer
\( Q = -4\,180 \, \text{J} \) or \( 4\,180 \, \text{J} \) absorbed *The negative sign indicates energy is absorbed by the dissolving process from the water. Stating "4 180 J absorbed" is also acceptable. The unit J must be included for full marks.* --- ### SECTION C - Question 13(e) **Answer:** This is a practical application. The data shows that dissolving 25.0 g of ammonium nitrate in 100 g of water causes a temperature drop of 10 °C, which is significant enough to provide a cooling effect for treating sports injuries. Instant cold packs work on this principle - when the inner pouch is broken, ammonium nitrate dissolves in water, absorbing energy and creating a cold sensation. The process is quick, portable, and does not require refrigeration, making it very practical for first aid use. *Full marks for: (1) stating it is practical, (2) referring to the data (temperature drop of 10 °C), and (3) explaining how this relates to the real-world application.* --- ### SECTION D - Question 14(a) **Answer:** The independent variable is the size of the marble chips (or particle size of calcium carbonate). The independent variable is the one that Bongani deliberately changed to observe its effect on the rate of reaction. --- ### SECTION D - Question 14(b) **Answer:** The dependent variable is the rate at which carbon dioxide gas is produced (or the volume of gas produced over time, or the time taken to produce a certain volume of gas). The dependent variable is what Bongani measured to see the effect of changing the marble chip size. --- ### SECTION D - Question 14(c) **Answer (example):** One controlled variable is the concentration of hydrochloric acid (1.0 mol·dm⁻³). This must be kept constant because changing the concentration would change the number of acid particles available to react, which would affect the rate of the reaction independently of the marble chip size, making it impossible to fairly compare the effect of chip size alone. *Other acceptable controlled variables include: mass of marble chips (10 g), volume of acid (100 cm³), or temperature (25 °C). Full marks for naming the variable and explaining why it must be controlled.* --- ### SECTION D - Question 14(d) **Answer (model hypothesis):** If the size of marble chips decreases, then the rate of carbon dioxide production will increase, because smaller chips have a larger total surface area exposed to the acid, which increases the frequency of collisions between reactant particles. *Full marks for a correctly formatted hypothesis that includes: condition (chip size decreases), expected result (rate increases), and scientific reason (surface area and collision frequency).* --- ### SECTION D - Question 14(e) **Answer:** The hypothesis is supported by the data. The results show that small chips produced 240 cm³ of gas in 2 minutes, medium chips took 4 minutes, and large chips took 7 minutes to produce the same volume of gas. This clearly demonstrates that smaller marble chips react faster (higher rate) than larger chips, which matches the prediction in the hypothesis. All three sizes produced the same total volume of gas, but the time taken decreased as chip size decreased. *Full marks for: (1) stating the hypothesis is supported, (2) quoting specific data values, and (3) linking the data to the hypothesis prediction.* --- ### SECTION D - Question 14(f) **Answer:** According to collision theory, a chemical reaction occurs when reactant particles collide with sufficient energy. Small marble chips have a much larger total surface area compared to large chips of the same mass. This means more calcium carbonate particles are exposed to the hydrochloric acid at any given time, which increases the number of collisions between acid particles and carbonate particles per second. More collisions per second result in a faster rate of reaction, so gas is produced more quickly with small chips than with large chips. *Full marks for: (1) mentioning collision theory or collisions, (2) explaining the role of surface area, and (3) linking increased collisions to increased reaction rate.* --- ## Mark Allocation Summary
 --- **End of Answer Key** Keep practicing, stay curious, and remember that understanding the difference between physical and chemical changes is fundamental to all of chemistry. You're doing great!
--- **End of Answer Key** Keep practicing, stay curious, and remember that understanding the difference between physical and chemical changes is fundamental to all of chemistry. You're doing great!The document Physical and Chemical Change is a part of the Grade 10 Course Physical Sciences for Grade 10.
All you need of Grade 10 at this link: Grade 10
Related Searches
Physical and Chemical Change, Important questions, Free, Semester Notes, Physical and Chemical Change, Physical and Chemical Change, Objective type Questions, shortcuts and tricks, Exam, MCQs, Sample Paper, video lectures, practice quizzes, past year papers, mock tests for examination, Extra Questions, Summary, Previous Year Questions with Solutions, ppt, study material, Viva Questions, pdf ;





