Representing Chemical Change
SECTION A: MULTIPLE CHOICE
(Total: 10 marks)
1. Which one of the following correctly represents a balanced chemical equation for the combustion of methane gas?
A. CH4 + O2 → CO2 + H2O
B. CH4 + 2O2 → CO2 + 2H2O
C. 2CH4 + O2 → 2CO2 + H2O
D. CH4 + O2 → CO2 + 2H2O
2. The law of conservation of mass states that:
A. Mass is always lost during a chemical reaction
B. The total mass of reactants equals the total mass of products in a closed system
C. Mass can be created but not destroyed
D. The mass of products is always greater than the mass of reactants
3. In the equation 2H2 + O2 → 2H2O, the coefficient 2 in front of H2 indicates:
A. Two atoms of hydrogen
B. Two molecules of hydrogen
C. Two grams of hydrogen
D. Two moles of hydrogen atoms
4. How many oxygen atoms are represented in the formula 3Ca(NO3)2?
A. 6
B. 9
C. 12
D. 18
5. When magnesium ribbon burns in oxygen, 4.8 g of magnesium reacts completely with 3.2 g of oxygen to form magnesium oxide. What mass of magnesium oxide is produced?
A. 1.6 g
B. 4.8 g
C. 8.0 g
D. 12.0 g
6. Which statement best describes a chemical equation?
A. A representation showing only the reactants in a chemical reaction
B. A symbolic representation of a chemical reaction showing reactants and products
C. A list of all elements present in a compound
D. A diagram showing the physical state of matter only
7. Thabo balances the equation for the reaction between aluminium and oxygen: __Al + __O2 → __Al2O3. What are the correct coefficients in order?
A. 2, 3, 2
B. 4, 3, 2
C. 3, 2, 3
D. 2, 2, 1
8. In a closed container, 10.0 g of sodium reacts with 8.0 g of chlorine gas to form 18.0 g of sodium chloride. Which law does this observation support?
A. Law of definite proportions
B. Law of multiple proportions
C. Law of conservation of mass
D. Law of constant composition
>9. The state symbol (aq) in a chemical equation represents:
A. A gaseous substance
B. A liquid substance
C. A solid substance
D. A substance dissolved in water
10. Consider the equation: N2(g) + 3H2(g) → 2NH3(g). If 28 g of nitrogen reacts completely with hydrogen, what mass of ammonia is produced? (Relative atomic masses: N = 14; H = 1)
A. 17 g
B. 28 g
C. 34 g
D. 51 g
SECTION B: STRUCTURED QUESTIONS
(Total: 30 marks)
Question 11
(a) Define the term balanced chemical equation.
(b) Explain why it is necessary to balance a chemical equation.
(c) Balance the following chemical equation:
Fe + O2 → Fe2O3
(d) Nombuso conducts an experiment where she heats 6.4 g of copper in air to form copper(II) oxide. The equation for the reaction is:
2Cu(s) + O2(g) → 2CuO(s)
Calculate the mass of copper(II) oxide formed. (Relative atomic masses: Cu = 64; O = 16)
(e) In the reaction above, Nombuso observes that the mass of the product is greater than the mass of copper she started with. Explain this observation using the law of conservation of mass.
Question 12
(a) Write down the meaning of each of the following state symbols used in chemical equations:
(i) (s)
(ii) (l)
(iii) (g)
(iv) (aq)
(b) Write a balanced chemical equation for the reaction between hydrochloric acid and sodium hydroxide to form sodium chloride and water. Include state symbols.
(c) Sipho carries out a neutralisation reaction by mixing 40.0 g of sodium hydroxide solution with excess hydrochloric acid. He collects 58.5 g of sodium chloride. The balanced equation for the reaction is:
NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(l)
If 4.0 g of sodium hydroxide was dissolved in the solution, calculate the mass of water produced in this reaction.
(d) Evaluate whether the law of conservation of mass is obeyed in Sipho's experiment. Show all calculations to support your answer.
SECTION C: DATA ANALYSIS AND GRAPHING
(Total: 15 marks)
Question 13
Lerato investigates the law of conservation of mass by heating different masses of magnesium ribbon in air to form magnesium oxide. The balanced equation for the reaction is:
2Mg(s) + O2(g) → 2MgO(s)
Her results are shown in the table below:
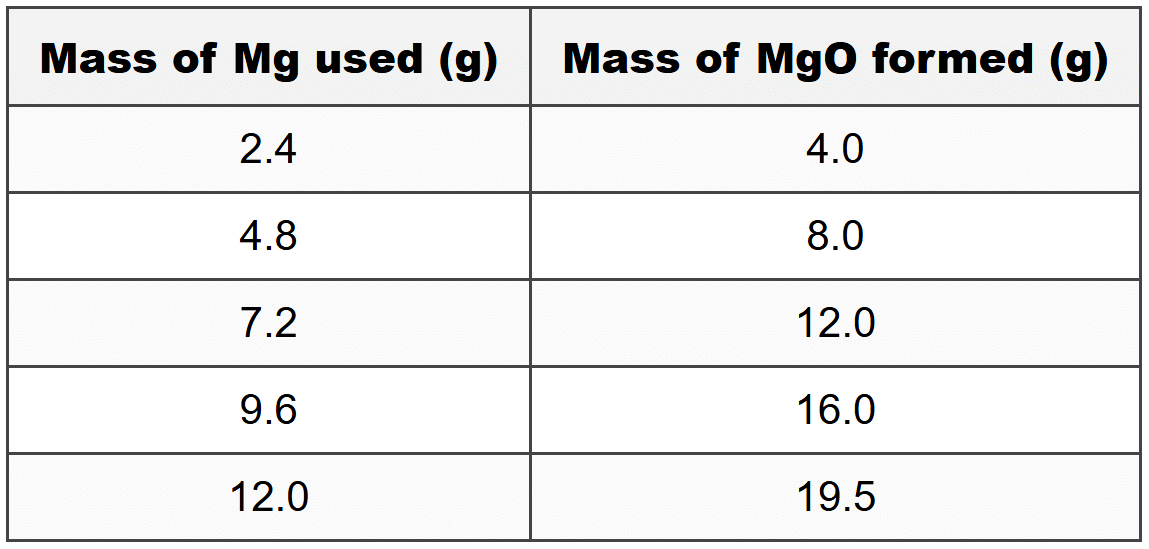
(a) How much oxygen reacted with 4.8 g of magnesium?
(b) Calculate the mass of oxygen that should have reacted with 12.0 g of magnesium according to the law of conservation of mass. (Relative atomic masses: Mg = 24; O = 16)
(c) Identify which data point in the table appears to be anomalous. Give a reason for your answer.
(d) Analyse the relationship between the mass of magnesium used and the mass of magnesium oxide formed. Explain whether this relationship supports the law of conservation of mass.
SECTION D: SCIENTIFIC INVESTIGATION
(Total: 15 marks)
Question 14
Zanele investigates whether the law of conservation of mass applies to the reaction between solutions of lead(II) nitrate and potassium iodide. When these two solutions are mixed, a yellow precipitate of lead(II) iodide forms according to the following equation:
Pb(NO3)2(aq) + 2KI(aq) → PbI2(s) + 2KNO3(aq)
Zanele carries out the investigation as follows:
She measures the mass of a beaker containing 50 cm³ of lead(II) nitrate solution and finds it to be 85.0 g. She measures the mass of a test tube containing 25 cm³ of potassium iodide solution and finds it to be 42.0 g. She then pours the potassium iodide solution into the beaker with the lead(II) nitrate solution and observes a yellow precipitate forming. After the reaction is complete, she measures the total mass of the beaker and its contents.
(a) Identify the independent variable in this investigation.
(b) Identify the dependent variable in this investigation.
(c) Name one variable that must be controlled in this investigation and explain why it must be kept constant.
(d) Write a suitable hypothesis for this investigation in the format: If [condition], then [expected result], because [scientific reason].
(e) Zanele measures the total mass after mixing and finds it to be 127.0 g. Use this result to evaluate whether the law of conservation of mass was obeyed in this investigation. Explain your answer fully.
(f) Zanele's classmate, Bongani, suggests that if the investigation were carried out in an open beaker without a lid, the results might be different for a reaction that produces a gas. Evaluate Bongani's suggestion and explain whether the law of conservation of mass would still be obeyed if a gas escapes from an open container.
GRAND TOTAL: 70
ANSWER KEY
Well done on completing this worksheet! Remember to check your working carefully, especially in calculation questions where you need to show all steps. Use this answer key to identify areas where you need more practice. Keep up the great work!
SECTION A - MULTIPLE CHOICE
Question 1
B. CH4 + 2O2 → CO2 + 2H2O
This is the correct balanced equation because there is 1 carbon atom, 4 hydrogen atoms, and 4 oxygen atoms on both sides of the equation. The coefficient 2 in front of O2 and 2H2O ensures that the equation is balanced.
Question 2
B. The total mass of reactants equals the total mass of products in a closed system
The law of conservation of mass states that mass cannot be created or destroyed during a chemical reaction, so the total mass remains constant in a closed system.
Question 3
B. Two molecules of hydrogen
A coefficient in a chemical equation represents the number of molecules (or formula units) of a substance. The coefficient 2 means two separate H2 molecules are involved in the reaction.
Question 4
D. 18
The formula 3Ca(NO3)2 contains 3 formula units of calcium nitrate. Each Ca(NO3)2 unit contains 2 NO3 groups, and each NO3 group contains 3 oxygen atoms. Therefore: 3 × 2 × 3 = 18 oxygen atoms in total.
Question 5
C. 8.0 g
According to the law of conservation of mass, the total mass of products equals the total mass of reactants. Mass of MgO = 4.8 g + 3.2 g = 8.0 g. This demonstrates that mass is conserved during the chemical reaction.
Question 6
B. A symbolic representation of a chemical reaction showing reactants and products
A chemical equation uses chemical formulas and symbols to represent what happens during a chemical reaction, showing both the substances that react (reactants) and the substances that are formed (products).
Question 7
B. 4, 3, 2
The balanced equation is 4Al + 3O2 → 2Al2O3. This gives 4 aluminium atoms and 6 oxygen atoms on both sides of the equation, making it balanced.
Question 8
C. Law of conservation of mass
The observation shows that the total mass of reactants (10.0 g + 8.0 g = 18.0 g) equals the total mass of products (18.0 g), which is a direct demonstration of the law of conservation of mass.
Question 9
D. A substance dissolved in water
The state symbol (aq) stands for "aqueous" which means the substance is dissolved in water to form a solution.
Question 10
C. 34 g
Working:
From the equation, 1 mole of N2 (28 g) produces 2 moles of NH3
Molar mass of NH3 = 14 + (3 × 1) = 17 g·mol⁻¹
Mass of 2 moles of NH3 = 2 × 17 = 34 g
Therefore, 28 g of nitrogen produces 34 g of ammonia. This can be verified using the law of conservation of mass: we also need to calculate the mass of hydrogen that reacts (6 g), giving total reactant mass of 34 g.
SECTION B - STRUCTURED QUESTIONS
Question 11(a)
A balanced chemical equation is a symbolic representation of a chemical reaction in which the number of atoms of each element is the same on both the reactant side and the product side of the equation. This ensures that the law of conservation of mass is obeyed.
Question 11(b)
It is necessary to balance a chemical equation because of the law of conservation of mass, which states that matter cannot be created or destroyed during a chemical reaction. A balanced equation ensures that the same number of atoms of each element appears on both sides of the equation, showing that mass is conserved. Additionally, a balanced equation provides the correct mole ratio between reactants and products, which is essential for stoichiometric calculations.
Question 11(c)
4Fe + 3O2 → 2Fe2O3
This equation is balanced because there are 4 iron atoms and 6 oxygen atoms on both sides.
Question 11(d)
Step 1: From the balanced equation: 2Cu(s) + O2(g) → 2CuO(s)
The mole ratio is 2 mol Cu : 2 mol CuO, which simplifies to 1:1
Step 2: Calculate the number of moles of copper
n = m ÷ M
n(Cu) = 6.4 g ÷ 64 g·mol⁻¹
n(Cu) = 0.1 mol
Step 3: From the 1:1 mole ratio:
n(CuO) = n(Cu) = 0.1 mol
Step 4: Calculate the molar mass of CuO
M(CuO) = 64 + 16 = 80 g·mol⁻¹
Step 5: Calculate the mass of CuO formed
m = n × M
m(CuO) = 0.1 mol × 80 g·mol⁻¹
m(CuO) = 8.0 g
Final answer: The mass of copper(II) oxide formed is 8.0 g.
Question 11(e)
Nombuso observes that the mass of the product is greater than the mass of copper she started with because oxygen from the air has reacted with the copper during the reaction. The law of conservation of mass states that the total mass of reactants equals the total mass of products in a closed system. In this reaction, the reactants are copper (6.4 g) and oxygen from the air (1.6 g), giving a total reactant mass of 8.0 g, which equals the mass of the product (8.0 g of copper(II) oxide). The mass has not increased overall; rather, the mass of oxygen has been incorporated into the product, which explains why the product appears heavier than just the copper alone.
Question 12(a)
(i) (s) means solid - the substance is in the solid state
(ii) (l) means liquid - the substance is in the liquid state
(iii) (g) means gas - the substance is in the gaseous state
(iv) (aq) means aqueous - the substance is dissolved in water
Question 12(b)
HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
This equation is already balanced with a 1:1:1:1 mole ratio, and all state symbols are included correctly.
Question 12(c)
Step 1: Use the law of conservation of mass
Total mass of reactants = Total mass of products
Step 2: Identify the masses
Mass of solution used = 40.0 g (this contains 4.0 g NaOH and 36.0 g water)
We need to find the mass of HCl that reacted
Step 3: Calculate moles of NaOH
Molar mass of NaOH = 23 + 16 + 1 = 40 g·mol⁻¹
n(NaOH) = 4.0 g ÷ 40 g·mol⁻¹ = 0.1 mol
Step 4: From the 1:1:1:1 ratio in the equation:
n(HCl) = 0.1 mol, n(NaCl) = 0.1 mol, n(H2O) = 0.1 mol
Step 5: Calculate mass of NaCl expected
M(NaCl) = 23 + 35.5 = 58.5 g·mol⁻¹
Expected m(NaCl) = 0.1 mol × 58.5 g·mol⁻¹ = 5.85 g
The question states 58.5 g of sodium chloride was collected, which seems to include the mass of the solution. Let me recalculate based on the actual chemistry:
Step 6: Calculate mass of water produced from the reaction
M(H2O) = 2 + 16 = 18 g·mol⁻¹
m(H2O produced) = 0.1 mol × 18 g·mol⁻¹
m(H2O produced) = 1.8 g
Final answer: The mass of water produced in the reaction is 1.8 g.
Question 12(d)
To evaluate whether the law of conservation of mass is obeyed, we need to compare the total mass of reactants with the total mass of products.
Reactants:
Mass of NaOH = 4.0 g
We need to calculate the mass of HCl that reacted:
n(HCl) = 0.1 mol (from the 1:1 ratio)
M(HCl) = 1 + 35.5 = 36.5 g·mol⁻¹
m(HCl) = 0.1 mol × 36.5 g·mol⁻¹ = 3.65 g
Total mass of reactants = 4.0 g + 3.65 g = 7.65 g
Products:
Mass of NaCl = 5.85 g (calculated earlier)
Mass of H2O = 1.8 g (calculated earlier)
Total mass of products = 5.85 g + 1.8 g = 7.65 g
Conclusion: The law of conservation of mass is obeyed in Sipho's experiment because the total mass of reactants (7.65 g) equals the total mass of products (7.65 g). The figure of 58.5 g mentioned in the question likely refers to the total mass of the solution collected, not just the sodium chloride. When we consider only the reacting substances (not including the solvent water that was already present), mass is conserved.
SECTION C - DATA ANALYSIS AND GRAPHING
Question 13(a)
Using the law of conservation of mass:
Mass of oxygen = Mass of MgO formed - Mass of Mg used
Mass of oxygen = 8.0 g - 4.8 g
Mass of oxygen = 3.2 g
Question 13(b)
Step 1: Calculate the number of moles of Mg
n(Mg) = m ÷ M
n(Mg) = 12.0 g ÷ 24 g·mol⁻¹
n(Mg) = 0.5 mol
Step 2: From the balanced equation 2Mg + O2 → 2MgO
The mole ratio is 2 mol Mg : 1 mol O2
n(O2) = 0.5 mol ÷ 2 = 0.25 mol
Step 3: Calculate the mass of O2
M(O2) = 2 × 16 = 32 g·mol⁻¹
m(O2) = 0.25 mol × 32 g·mol⁻¹
m(O2) = 8.0 g
Step 4: Calculate expected mass of MgO using conservation of mass
m(MgO) = m(Mg) + m(O2)
m(MgO) = 12.0 g + 8.0 g
m(MgO) = 20.0 g
Final answer: According to the law of conservation of mass, 8.0 g of oxygen should have reacted with 12.0 g of magnesium to produce 20.0 g of magnesium oxide.
Question 13(c)
The last data point (12.0 g Mg producing 19.5 g MgO) appears to be anomalous. According to the pattern established by the other data points and the calculation in part (b), 12.0 g of magnesium should produce 20.0 g of magnesium oxide, not 19.5 g. This anomaly could be due to experimental error, such as incomplete combustion of the magnesium, loss of product during transfer, or inaccurate measurement of the final mass.
Question 13(d)
The data shows a directly proportional relationship between the mass of magnesium used and the mass of magnesium oxide formed. For the first four data points, when the mass of magnesium doubles, the mass of magnesium oxide also doubles, maintaining a constant ratio of approximately 1:1.67 (or 3:5). This relationship strongly supports the law of conservation of mass because it demonstrates that a consistent mass of oxygen (1.6 g for every 2.4 g of Mg) combines with the magnesium in a fixed ratio. The total mass is conserved in each reaction, as the mass of MgO equals the sum of the masses of Mg and O2 that reacted. The anomalous final data point slightly deviates from this pattern, but the overall trend across the majority of the data clearly demonstrates mass conservation. (Full marks: identifies direct proportion, explains constant ratio, links to conservation of mass with specific reference to data; Partial marks: identifies relationship but doesn't fully explain conservation principle)
SECTION D - SCIENTIFIC INVESTIGATION
Question 14(a)
The independent variable is mixing the two solutions together (or the act of combining lead(II) nitrate solution with potassium iodide solution). This is the variable that Zanele deliberately changes or manipulates to observe its effect on the total mass.
Question 14(b)
The dependent variable is the total mass of the system after the reaction. This is the variable that is measured to see if it changes as a result of the chemical reaction occurring.
Question 14(c)
One controlled variable is temperature (or the use of a closed system, or the volume of solutions used). Temperature must be kept constant because changes in temperature could cause evaporation of water from the solutions, which would result in a loss of mass that is not due to the chemical reaction itself. This would affect the validity of the investigation into whether mass is conserved during the chemical reaction. (Accept any reasonable controlled variable with correct explanation)
Question 14(d)
Model hypothesis:
If lead(II) nitrate solution is mixed with potassium iodide solution in a closed system, then the total mass after the reaction will equal the total mass before the reaction, because the law of conservation of mass states that matter cannot be created or destroyed during a chemical reaction, only rearranged.
Question 14(e)
Calculation of total mass before reaction:
Mass before = 85.0 g + 42.0 g = 127.0 g
Mass after reaction: 127.0 g
Conclusion: The law of conservation of mass was obeyed in this investigation. The total mass before the reaction (127.0 g) is exactly equal to the total mass after the reaction (127.0 g), with a difference of 0.0 g. This demonstrates that no mass was lost or gained during the chemical reaction. Even though a precipitate formed and the chemical composition of the substances changed (reactants were converted to products), the total mass remained constant. This result supports the law of conservation of mass, which states that in a closed system, the total mass of reactants must equal the total mass of products because atoms are rearranged but not created or destroyed during chemical reactions.
Question 14(f)
Bongani's suggestion is scientifically valid and demonstrates good understanding. If a reaction produces a gas and is carried out in an open container, the gas can escape into the atmosphere. In such a case, if you measure only the mass of the contents remaining in the beaker, it would appear that mass has been lost, and you might incorrectly conclude that the law of conservation of mass has been violated. However, the law of conservation of mass would still be obeyed - the "missing" mass has not disappeared but has simply left the system as gas molecules that escaped into the air. If you could measure the mass of the escaped gas and add it to the mass of the remaining products, the total would still equal the mass of the reactants. This is why conservation of mass experiments must be conducted in a closed system where no substances can enter or leave. In Zanele's investigation with the precipitate reaction, no gas is produced, so carrying it out in an open beaker would not affect the results. (Full marks: explains that mass appears to be lost, clarifies that law still holds if gas mass is accounted for, mentions importance of closed system; Partial marks: identifies that gas escapes but doesn't fully explain conservation principle)
MARK ALLOCATION SUMMARY
Remember: In calculation questions, always show your formula, substitute with units, show all working, and state your final answer with the correct SI unit. Marks are awarded for method even if your final answer is incorrect, so never leave a calculation question blank!





