Physical and Chemical Change
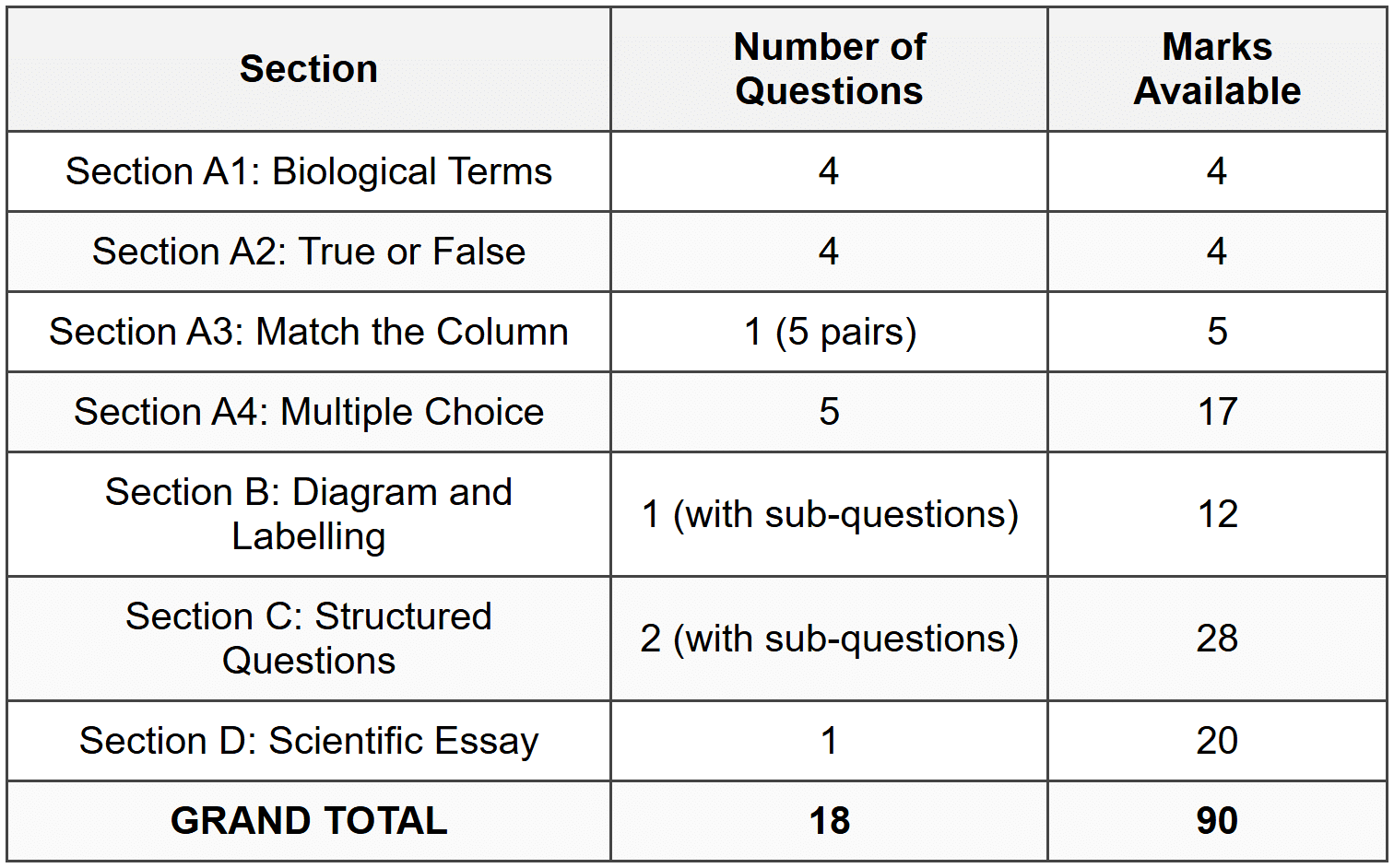
SECTION A: SHORT QUESTIONS
(Total: 30 marks)
A1: Give the correct biological term
(Total: 4 marks)
1. The process by which large complex food molecules are broken down into smaller, simpler molecules that can be absorbed by cells.
2. The type of change that occurs when a substance is altered in form or state but retains its chemical composition and can be reversed.
3. The biological catalysts that speed up the rate of chemical reactions in living organisms without being consumed in the process.
4. The energy required to initiate a chemical reaction by breaking the bonds in reactant molecules.
A2: True or False
(Total: 4 marks)
5. Physical changes in living organisms involve the formation of new substances with different chemical properties from the original substance.
6. Enzymes lower the activation energy required for chemical reactions in cells, thereby increasing the rate at which metabolic processes occur.
7. The digestion of starch by amylase in the mouth is an example of a physical change because the food is being broken down.
8. Denaturation of enzymes is a permanent chemical change that occurs when the primary structure of the protein is altered by extreme temperature or pH.
A3: Match Column A to Column B
(Total: 5 marks)
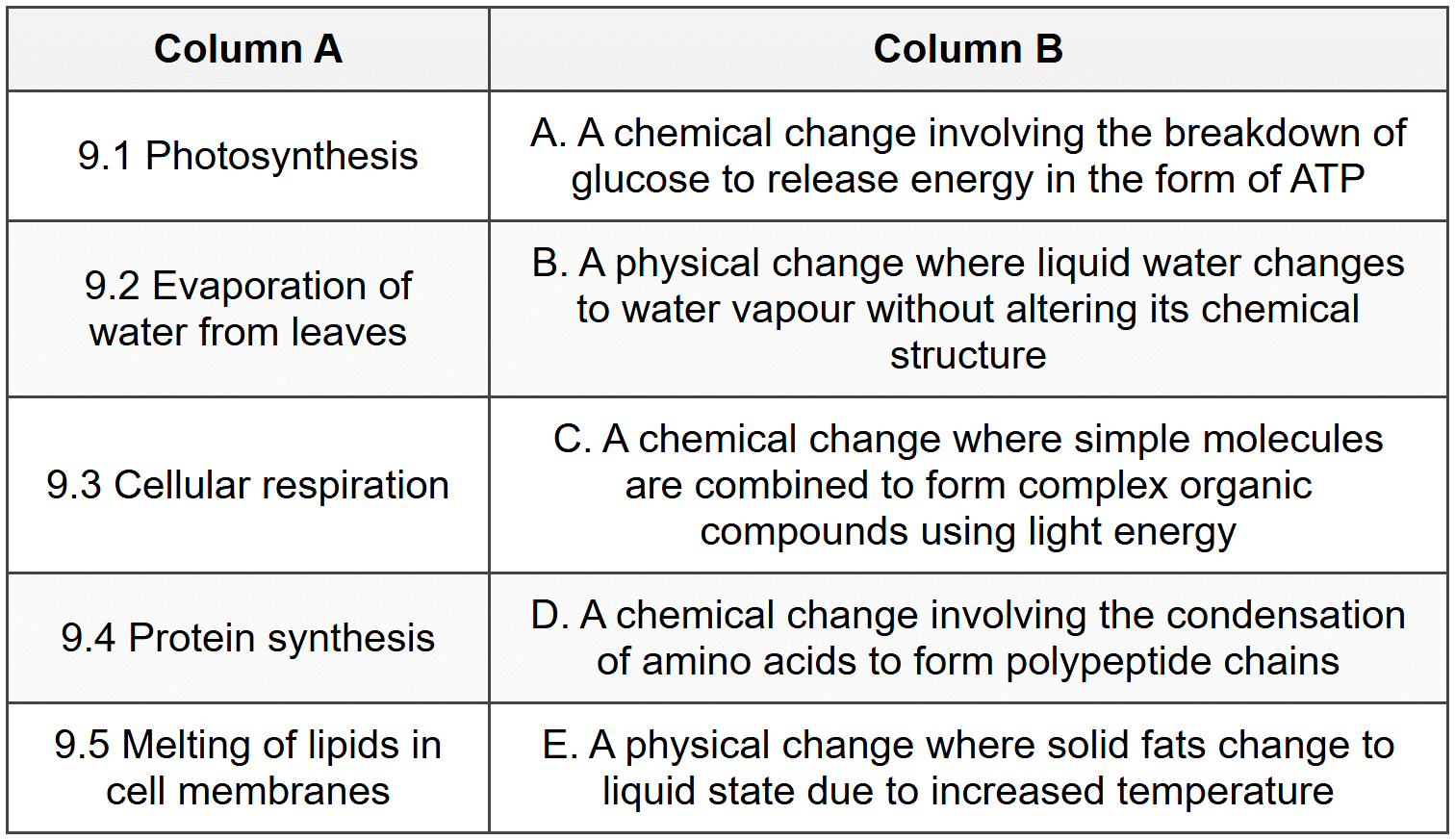
9. Match the biological processes in Column A with their correct descriptions in Column B. Write only the letter (A-E) next to the question number.
A4: Multiple Choice
(Total: 17 marks)
10. Thandi conducted an experiment where she added hydrogen peroxide to liver tissue. Bubbles formed immediately and the liver tissue remained unchanged. Which statement best explains this observation?
A. The liver tissue underwent a physical change and released oxygen gas
B. The enzyme catalase in the liver catalysed the chemical breakdown of hydrogen peroxide into water and oxygen
C. The liver tissue dissolved in the hydrogen peroxide causing a physical reaction
D. The bubbles indicate a physical change as no new substances were formed
11. A student observes that when bread is toasted, it turns brown and cannot be changed back to its original state. This observation indicates that toasting bread is:
A. A physical change because the bread changed colour
B. A physical change because the bread became harder
C. A chemical change because new substances with different properties were formed
D. A reversible change because water can be added back to the bread
12. During photosynthesis in Pelargonium plants, carbon dioxide and water are converted into glucose and oxygen. Which statement correctly identifies the type of change and provides valid reasoning?
A. Physical change, because the plant uses physical energy from sunlight
B. Chemical change, because new substances with different molecular structures are formed from the reactants
C. Physical change, because carbon dioxide and oxygen are both gases
D. Chemical change, because the mass of the products equals the mass of the reactants
13. Sipho investigated the effect of temperature on enzyme activity. He found that at 25°C the enzyme worked efficiently, but at 65°C the enzyme stopped working completely. The loss of enzyme function at high temperature is best explained by:
A. A physical change where the enzyme melted and became liquid
B. A reversible physical change where the enzyme can regain its shape when cooled
C. A chemical change where the heat broke the peptide bonds in the primary structure
D. A chemical change where hydrogen bonds and other weak bonds maintaining the tertiary structure were disrupted, altering the active site permanently
14. In the human digestive system, lipase catalyses the breakdown of lipids into fatty acids and glycerol. Students were given four scenarios and asked to identify which represents the same type of change as lipid digestion. Which scenario should they select?
A. Ice melting to form liquid water in a cold drink
B. Amylase breaking down starch into maltose in the mouth
C. Salt dissolving in water to form a solution
D. Oxygen diffusing from the alveoli into blood capillaries
SECTION B: DIAGRAM AND LABELLING QUESTIONS
(Total: 12 marks)
Study the figure described below showing a biological process involving chemical change.

15. (a) Identify structures 1 to 6 by providing the correct biological term for each numbered label.
(b) Explain the function of structure 1 in this chemical change and describe the function of structure 2 in ensuring specificity of the reaction.
(c) Predict what would happen to the rate of product formation (structure 5) if structure 2 was permanently altered by exposure to extreme pH. Explain your answer using the concept of enzyme specificity.
SECTION C: STRUCTURED QUESTIONS
(Total: 28 marks)
Question 16: Digestive Processes
Nomsa investigated the digestion of different food types. She placed equal amounts of cooked egg white (protein), cooking oil (lipid), and bread (starch) into three separate test tubes. She added the enzyme pepsin to the tube containing egg white, lipase to the tube containing oil, and amylase to the tube containing bread. All tubes were maintained at 37°C. After 30 minutes, she tested the contents of each tube.
16. (a) State whether the changes occurring in each of the three test tubes represent physical or chemical changes. Justify your answer with reference to the molecular structure of the substances.
(b) Explain why Nomsa maintained all three test tubes at 37°C during the investigation.
(c) The test tube containing egg white and pepsin showed that the solid egg white had broken down into a clear liquid. Describe the chemical change that occurred using correct biological terminology, naming the type of bonds broken and the products formed.
(d) Nomsa repeated the experiment but added pepsin to all three test tubes instead of using different enzymes. Predict the results she would observe in the tubes containing oil and bread. Explain your prediction using the concept of enzyme specificity.
Question 17: Photosynthesis and Cellular Respiration
Lerato set up an investigation using Elodea canadensis, an aquatic plant. She placed the plant in water containing a pH indicator that turns yellow in acidic conditions and purple in alkaline conditions. Initially, the indicator was yellow due to dissolved carbon dioxide forming carbonic acid. She then placed the setup under a bright light for several hours.
17. (a) After several hours under bright light, Lerato observed that the indicator turned purple. Explain this observation by describing the chemical change that occurred in the plant and how it affected the carbon dioxide concentration in the water.
(b) Identify the type of chemical change represented by photosynthesis. Support your answer by referring to the formation of new substances.
(c) Write a balanced chemical equation for the photosynthetic process occurring in Elodea canadensis, using correct chemical formulae.
(d) Lerato then moved the plant to complete darkness for 12 hours. The indicator gradually changed from purple back to yellow. Analyse this result by explaining which metabolic process became dominant in the absence of light and why this would cause the observed colour change in the indicator.
SECTION D: SCIENTIFIC ESSAY
(Total: 20 marks)
18. Write a scientific essay on the role of enzymes in facilitating chemical changes in living organisms.
Your essay must include the following:
- Define enzymes and explain their chemical nature as biological catalysts
- Describe how enzymes lower activation energy and increase the rate of metabolic reactions, using the lock-and-key model or induced-fit model
- Explain the difference between physical and chemical changes, providing one example of each from metabolic processes
- Discuss how temperature and pH affect enzyme structure and function, distinguishing between reversible and irreversible changes
- Provide a specific example of an enzyme-catalysed reaction in a named organism using correct binomial nomenclature, stating the substrate, enzyme, and products
Mark allocation guide:
- Definition of enzymes and their chemical nature (3 marks)
- Description of how enzymes lower activation energy with reference to a model (5 marks)
- Explanation of physical versus chemical changes with metabolic examples (4 marks)
- Discussion of temperature and pH effects on enzyme structure (5 marks)
- Specific example with correct nomenclature, substrate, enzyme, and products (3 marks)
GRAND TOTAL: 90 marks
ANSWER KEY
Well done on completing this worksheet on Physical and Chemical Change in Life Sciences. Use this answer key to check your understanding and identify areas for improvement. Pay special attention to the correct biological terminology required for full marks.
SECTION A1 - Question 1
Digestion
Digestion is the process that breaks down large, complex food molecules such as proteins, carbohydrates, and lipids into smaller, simpler molecules like amino acids, glucose, and fatty acids that can be absorbed across cell membranes.
SECTION A1 - Question 2
Physical change
A physical change alters the form, state, or appearance of a substance without changing its chemical composition, and it is generally reversible. Examples include melting, freezing, and dissolving.
SECTION A1 - Question 3
Enzymes
Enzymes are biological catalysts composed of proteins that accelerate chemical reactions in living organisms by lowering the activation energy required, without being permanently altered or consumed in the process.
SECTION A1 - Question 4
Activation energy
Activation energy is the minimum energy required to initiate a chemical reaction by breaking the existing bonds in reactant molecules, allowing new bonds to form in the products.
SECTION A2 - Question 5
FALSE
The corrected statement: Physical changes in living organisms involve no formation of new substances with different chemical properties from the original substance.
Physical changes do not alter the chemical composition of substances. Only chemical changes result in the formation of new substances with different chemical properties. The phrase "formation of new substances" should be replaced with "changes in form or state only" or similar.
SECTION A2 - Question 6
TRUE
Enzymes function as biological catalysts by lowering the activation energy barrier, thereby increasing the rate of metabolic reactions such as cellular respiration, photosynthesis, and digestion.
SECTION A2 - Question 7
FALSE
The corrected statement: The digestion of starch by amylase in the mouth is an example of a chemical change because the food is being broken down.
The enzymatic breakdown of starch into maltose involves breaking glycosidic bonds and forming new, smaller molecules with different chemical properties, which defines a chemical change, not a physical change.
SECTION A2 - Question 8
FALSE
The corrected statement: Denaturation of enzymes is a permanent chemical change that occurs when the tertiary structure of the protein is altered by extreme temperature or pH.
Denaturation disrupts the weak hydrogen bonds and other interactions that maintain the tertiary (three-dimensional) structure of the enzyme, not the primary structure which consists of the sequence of amino acids held together by strong peptide bonds.
SECTION A3 - Question 9

9.1: Photosynthesis is an anabolic chemical change in which simple inorganic molecules (carbon dioxide and water) are combined using light energy to form complex organic compounds (glucose), representing a chemical change with new substances formed.
9.2: Evaporation is a physical change where water changes state from liquid to gas (water vapour) without any alteration in its chemical structure (H2O remains H2O).
9.3: Cellular respiration is a catabolic chemical change involving the breakdown of glucose molecules through the breaking of covalent bonds to release energy stored in ATP molecules.
9.4: Protein synthesis is a chemical change involving condensation reactions where amino acids are joined by peptide bonds to form polypeptide chains with new chemical properties.
9.5: Melting of lipids is a physical change where lipid molecules change from solid to liquid state due to increased kinetic energy from heat, but the chemical structure of the lipid molecules remains unchanged.
SECTION A4 - Question 10
B. The enzyme catalase in the liver catalysed the chemical breakdown of hydrogen peroxide into water and oxygen
Liver tissue contains high concentrations of the enzyme catalase, which catalyses the decomposition of hydrogen peroxide (H2O2) into water (H2O) and oxygen gas (O2). This is a chemical change because new substances with different properties are formed. The bubbles observed are oxygen gas, and the liver tissue remains unchanged because enzymes are not consumed in the reactions they catalyse.
SECTION A4 - Question 11
C. A chemical change because new substances with different properties were formed
Toasting bread involves a chemical change called the Maillard reaction, where sugars and amino acids react under heat to form new compounds that give the bread its brown colour and different taste. The change is irreversible because the new substances formed cannot be converted back to the original bread composition. The formation of new substances with different chemical properties is the defining characteristic of a chemical change.
SECTION A4 - Question 12
B. Chemical change, because new substances with different molecular structures are formed from the reactants
Photosynthesis is a chemical change because the reactants (carbon dioxide and water) are converted into products (glucose and oxygen) with completely different molecular structures and chemical properties. The covalent bonds in CO2 and H2O are broken, and new bonds are formed to create C6H12O6 and O2. This represents the formation of new substances, which is the hallmark of chemical change.
SECTION A4 - Question 13
D. A chemical change where hydrogen bonds and other weak bonds maintaining the tertiary structure were disrupted, altering the active site permanently
At high temperatures such as 65°C, denaturation occurs. This is a chemical change in which the weak hydrogen bonds, ionic bonds, and hydrophobic interactions that maintain the enzyme's tertiary structure are permanently disrupted. The active site changes shape and can no longer bind to the substrate, resulting in complete loss of enzyme function. This change is irreversible and is therefore a chemical, not physical, change. The primary structure (peptide bonds) remains intact.
SECTION A4 - Question 14
B. Amylase breaking down starch into maltose in the mouth
Both lipase breaking down lipids and amylase breaking down starch represent enzyme-catalysed chemical changes in which large, complex molecules (polymers) are hydrolysed into smaller molecules (monomers or dimers). Both involve breaking covalent bonds (ester bonds in lipids, glycosidic bonds in starch) and forming new substances with different chemical properties. The other options represent physical changes: melting, dissolving, and diffusion do not involve the formation of new chemical substances.
SECTION B - Question 15(a)
1. Enzyme
2. Active site
3. Substrate
4. Enzyme-substrate complex
5. Products
6. Activation energy
Students must use the exact biological terms listed above for full marks. "Catalyst" alone is insufficient for structure 1; "enzyme" must be stated. "Binding site" is insufficient for structure 2; "active site" is required.
SECTION B - Question 15(b)
The enzyme (structure 1) functions as a biological catalyst that lowers the activation energy required for the chemical breakdown of the substrate, thereby increasing the rate of the reaction without being consumed or permanently altered. The active site (structure 2) has a specific three-dimensional shape that is complementary to the shape of the substrate molecule, ensuring that only specific substrate molecules can bind to the enzyme, which provides enzyme specificity and prevents the enzyme from catalysing unwanted reactions.
Required for full marks: Students must mention that the enzyme lowers activation energy, increases reaction rate, and is not consumed. For the active site, they must mention specific three-dimensional shape, complementary to substrate, and enzyme specificity.
SECTION B - Question 15(c)
If structure 2 (the active site) was permanently altered by exposure to extreme pH, the rate of product formation would decrease to zero or stop completely. This is because extreme pH causes denaturation of the enzyme, which disrupts the weak bonds maintaining the tertiary structure of the enzyme protein. The altered active site would no longer have the specific complementary shape required to bind to the substrate, preventing the formation of the enzyme-substrate complex. Without the formation of this complex, the chemical reaction cannot be catalysed, and no products would be formed.
Full marks: Student explains that product formation stops/decreases to zero, mentions denaturation, states that the active site shape changes, explains that the substrate can no longer bind, and concludes that no enzyme-substrate complex forms so no reaction occurs.
Partial marks: Student states that the reaction rate decreases and mentions that the active site changes shape, but does not explain the mechanism of denaturation or link it to loss of enzyme-substrate complex formation.
SECTION C - Question 16(a)
All three test tubes demonstrate chemical changes. In the test tube containing egg white and pepsin, the peptide bonds in the protein molecules are hydrolysed, breaking the long polypeptide chains into shorter peptide chains and amino acids, which are chemically different from the original protein. In the test tube containing oil and lipase, the ester bonds in the triglyceride molecules are broken, producing fatty acids and glycerol, which have different chemical properties from the original lipid. In the test tube containing bread and amylase, the glycosidic bonds in the starch polymer are hydrolysed, producing maltose and eventually glucose molecules, which are chemically distinct from starch. All three processes result in the formation of new substances with different molecular structures, which is the defining characteristic of a chemical change.
Required for full marks: Student must state that all three are chemical changes, identify the specific bonds broken in at least two cases (peptide bonds, ester bonds, glycosidic bonds), name the products formed, and explain that new substances with different properties are produced.
SECTION C - Question 16(b)
Nomsa maintained all three test tubes at 37°C because this is the optimum temperature for enzyme activity in the human body. At optimum temperature, enzymes have the maximum kinetic energy for substrate collisions while maintaining the correct three-dimensional tertiary structure of the active site. Temperatures significantly higher or lower than 37°C would either denature the enzyme (if too high) or reduce the kinetic energy and frequency of enzyme-substrate collisions (if too low), both of which would decrease the rate of the chemical reactions being investigated.
Required for full marks: Student must mention optimum temperature, state that it maintains enzyme structure and function, and explain the effect on reaction rate.
SECTION C - Question 16(c)
The enzyme pepsin catalysed the hydrolysis of the egg white protein. During this chemical change, peptide bonds (also called amide bonds) that link amino acids together in the polypeptide chains were broken through the addition of water molecules. This catabolic reaction broke down the large, complex, insoluble protein molecules into smaller, soluble polypeptides and amino acids, which appeared as a clear liquid. The products (polypeptides and amino acids) have different chemical properties from the original protein, confirming that a chemical change occurred.
Required for full marks: Student must use the terms pepsin, hydrolysis, peptide bonds, and name the products as polypeptides and amino acids.
SECTION C - Question 16(d)
In the tube containing oil, no chemical change would occur and the oil would remain unchanged. In the tube containing bread, no chemical change would occur and the starch would remain as a solid. This is because pepsin is a protease enzyme with an active site that has a specific three-dimensional shape complementary only to protein substrates. Due to enzyme specificity, pepsin cannot catalyse the breakdown of lipids or carbohydrates because these substrates have different molecular shapes that do not fit into the active site of pepsin. The lock-and-key model or induced-fit model explains that only substrates with the correct complementary shape can bind to a specific enzyme's active site to form an enzyme-substrate complex.
Required for full marks: Student must predict that no change occurs in oil and bread tubes, explain enzyme specificity, mention the active site has a specific shape complementary only to proteins, and state that lipids and carbohydrates cannot bind to pepsin's active site.
SECTION C - Question 17(a)
The indicator turned purple because the Elodea canadensis plant was undergoing photosynthesis under the bright light. During photosynthesis, the plant absorbed carbon dioxide from the water to use as a raw material for synthesising glucose. As the carbon dioxide concentration in the water decreased, the concentration of carbonic acid (H2CO3) also decreased, making the water less acidic and more alkaline. The pH indicator responded to this increase in pH by changing colour from yellow (acidic) to purple (alkaline). This demonstrates that photosynthesis is a chemical change that consumes carbon dioxide and produces oxygen and glucose.
Required for full marks: Student must state that photosynthesis occurred, explain that carbon dioxide was absorbed/consumed by the plant, state that carbonic acid concentration decreased, explain that the water became more alkaline/less acidic, and link this to the colour change of the indicator.
SECTION C - Question 17(b)
Photosynthesis is a chemical change (specifically an anabolic or synthesis reaction). This is because the reactants-carbon dioxide (CO2) and water (H2O)-are converted into completely different products-glucose (C6H12O6) and oxygen (O2)-through the breaking and reforming of covalent bonds. The products have different molecular structures, different chemical properties, and different functions compared to the reactants. The formation of these new substances confirms that photosynthesis is a chemical change, not a physical change.
Required for full marks: Student must identify photosynthesis as a chemical change, state that new substances are formed, identify the reactants and products, and explain that they have different molecular structures and chemical properties.
SECTION C - Question 17(c)
6CO2 + 6H2O → C6H12O6 + 6O2
Or, showing light energy and chlorophyll:
6CO2 + 6H2O + light energy → C6H12O6 + 6O2
Required for full marks: Correct chemical formulae, correct balancing of the equation, and appropriate use of the arrow symbol. Accept either version of the equation.
SECTION C - Question 17(d)
In complete darkness, the Elodea canadensis plant could not perform photosynthesis because light energy is essential for the light-dependent reactions. However, the plant continued to perform cellular respiration to generate ATP for metabolic processes. Cellular respiration is a chemical change that consumes glucose and oxygen and releases carbon dioxide and water. As cellular respiration became the dominant metabolic process, carbon dioxide was released into the water, increasing the concentration of carbonic acid and making the water more acidic. This decrease in pH caused the indicator to change from purple (alkaline) back to yellow (acidic). This demonstrates that in the absence of photosynthesis, the plant is a net producer of carbon dioxide rather than a net consumer.
Required for full marks: Student must explain that photosynthesis stopped due to lack of light, state that cellular respiration continued or became dominant, explain that respiration releases carbon dioxide into the water, state that this increases acidity or decreases pH, and link this to the colour change to yellow.
SECTION D - Question 18: Model Essay Answer
1. Definition of enzymes and their chemical nature as biological catalysts
Enzymes are biological catalysts that increase the rate of chemical reactions in living organisms without being permanently altered or consumed in the process. Chemically, enzymes are globular proteins composed of one or more polypeptide chains folded into a specific three-dimensional tertiary structure. This structure includes a region called the active site, which has a shape complementary to specific substrate molecules.
2. Description of how enzymes lower activation energy with reference to a model
All chemical reactions require activation energy, which is the minimum energy needed to break existing bonds in reactant molecules so that new bonds can form. Enzymes function by lowering this activation energy barrier, thereby allowing reactions to proceed more rapidly at the temperatures found in living cells. According to the lock-and-key model, the active site of the enzyme has a rigid, fixed shape that is exactly complementary to the shape of the substrate, much like a lock and key. When the substrate binds to the active site, an enzyme-substrate complex forms. Alternatively, the induced-fit model proposes that the active site is flexible and changes shape slightly when the substrate approaches, moulding itself around the substrate to achieve a perfect fit. In both models, the formation of the enzyme-substrate complex strains and weakens the bonds in the substrate, making them easier to break. This reduces the activation energy required and increases the rate at which products are formed. After the reaction, the products are released and the enzyme returns to its original shape, ready to catalyse another reaction.
3. Explanation of physical versus chemical changes with metabolic examples
A physical change alters the form, state, or appearance of a substance without changing its chemical composition, and it is usually reversible. An example from metabolism is the diffusion of oxygen from the alveoli into the blood capillaries during gaseous exchange. The oxygen molecules (O2) move from an area of high concentration to an area of low concentration, but their chemical structure remains unchanged. In contrast, a chemical change involves the breaking and forming of covalent bonds, resulting in new substances with different chemical properties, and it is generally irreversible. An example is cellular respiration, where glucose (C6H12O6) is broken down in the presence of oxygen to form carbon dioxide (CO2), water (H2O), and ATP. The products have completely different molecular structures and properties from the reactants, confirming that a chemical change has occurred.
4. Discussion of temperature and pH effects on enzyme structure and function
Enzyme activity is significantly affected by temperature. At low temperatures, enzyme and substrate molecules have low kinetic energy, resulting in fewer collisions between them and a slower reaction rate. As temperature increases towards the optimum temperature (usually around 37°C for human enzymes), kinetic energy increases, collisions become more frequent, and the reaction rate increases. However, if the temperature rises significantly above the optimum, the enzyme undergoes denaturation. This is an irreversible chemical change in which the heat energy breaks the weak hydrogen bonds, ionic bonds, and hydrophobic interactions that maintain the enzyme's tertiary structure. The active site changes shape permanently and can no longer bind to the substrate, causing enzyme activity to cease completely. Similarly, pH affects enzyme function. Each enzyme has an optimum pH at which it functions most efficiently. Extreme pH values (very acidic or very alkaline) disrupt the ionic and hydrogen bonds in the enzyme's structure, causing denaturation and permanent loss of function. However, small deviations from optimum pH may cause only temporary, reversible changes in enzyme shape that reduce activity but do not permanently destroy the enzyme. Once conditions return to optimum, the enzyme can regain its functional shape.
5. Specific example with correct nomenclature, substrate, enzyme, and products
An example of an enzyme-catalysed reaction occurs in humans, Homo sapiens. In the mouth, the enzyme salivary amylase (also called ptyalin) catalyses the breakdown of starch, a polysaccharide substrate, into maltose, a disaccharide. The reaction involves the hydrolysis of glycosidic bonds that link glucose monomers in the starch molecule. The products, maltose molecules, have different chemical properties from starch: they are smaller, soluble, and sweet-tasting, whereas starch is a large, insoluble molecule. This represents a chemical change because new substances with different molecular structures are formed.
Full marks criteria for section 1: Student defines enzymes as biological catalysts, states they are proteins, mentions they are not consumed, and refers to the active site.
Partial marks criteria for section 1: Student defines enzymes as catalysts and states they are proteins, but omits reference to the active site or the fact that they are not consumed.
Full marks criteria for section 2: Student explains that enzymes lower activation energy, describes either the lock-and-key or induced-fit model with reference to the active site and enzyme-substrate complex, and explains how the complex strains substrate bonds.
Partial marks criteria for section 2: Student states that enzymes lower activation energy and mentions the active site, but does not describe a model or explain the mechanism of bond weakening.
Full marks criteria for section 3: Student defines both physical and chemical changes, provides a correct metabolic example of each, and explains why each example represents that type of change with reference to molecular structure.
Partial marks criteria for section 3: Student defines physical and chemical changes and provides examples, but does not explain the molecular basis for the classification.
Full marks criteria for section 4: Student discusses both temperature and pH, explains the concept of optimum conditions, describes denaturation as an irreversible chemical change caused by disruption of weak bonds in the tertiary structure, and distinguishes between reversible and irreversible changes.
Partial marks criteria for section 4: Student discusses temperature and pH effects and mentions denaturation, but does not explain the molecular mechanism or distinguish between reversible and irreversible changes.
Full marks criteria for section 5: Student provides a specific example with correct binomial nomenclature in italics, names the enzyme, substrate, and products, and identifies the type of bond broken.
Partial marks criteria for section 5: Student provides an example with enzyme, substrate, and products, but uses incorrect or missing binomial nomenclature or does not identify the bond type.
Marker's note: Common errors include using non-specific language such as "breaks down food" instead of "hydrolyses peptide bonds," failing to use correct binomial nomenclature with proper formatting, confusing physical and chemical changes in metabolic processes, stating that denaturation affects the primary structure instead of the tertiary structure, and omitting the key concept that enzymes are not consumed in reactions. Students must use precise biological terminology throughout to achieve full marks.
MARK ALLOCATION SUMMARY