Chapter Notes: Potentiometry - 1
Potentiometry
Introduction
When a metal M is immersed in a solution containing its ions Mn+, an electrode potential is established. The potential of such an electrode is given by the Nernst equation. For the half-reaction involving n electrons, at temperature T,
E = E° + (RT/nF) ln a(Mn+)
where E° is the standard electrode potential, R is the gas constant, T the absolute temperature and F the Faraday constant. By combining the electrode under test with a stable reference electrode and measuring the cell e.m.f., the activity a(Mn+) of the metal ion in solution can be deduced. For dilute solutions activity ≈ concentration; for more concentrated solutions the activity coefficient must be used to convert activity to concentration.
The method of determining an ion concentration from a single electrode-potential measurement is called direct potentiometry. The electrode whose potential depends on the analyte concentration is called the indicator electrode. When the analyte ion participates directly in the electrode reaction, the electrode is called an electrode of the first kind. If the analyte ion influences the electrode potential indirectly (e.g., via a sparingly soluble salt), the electrode is an electrode of the second kind.
Example of a second-kind electrode: a silver-silver chloride electrode. The silver electrode potential is affected by the Ag+ activity determined by the silver chloride solubility and thus by the chloride activity. Using the solubility equilibrium the electrode potential can be expressed in terms of chloride activity.
At 25 °C the factor (RT/nF)·ln(10) = 0.0591 V when n = 1. Thus a ten-fold change in activity of a singly charged ion changes the electrode potential by ≈ 59.1 mV. For a doubly charged ion (n = 2) the same ten-fold change produces ≈ 29.6 mV shift. High precision in concentration determination therefore requires very precise potential measurement.
A practical complication in measuring cell e.m.f. is the liquid junction potential established where the test solution contacts the reference electrode electrolyte. This can be minimised by using concentrated electrolytes in which cation and anion mobilities are similar (e.g., saturated KCl), or by using concentration cells where both compartments contain the same ionic species at different concentrations.
Because direct potentiometry requires accurate absolute potentials, potentiometric titration is often preferred. In potentiometric titration the change in electrode potential during addition of titrant is monitored; the end point corresponds to the region where the potential changes most rapidly, and the liquid junction potential is largely irrelevant because only potential changes-not absolute values-are used.
Reference electrodes
The hydrogen electrode
The standard hydrogen electrode (SHE) is the primary reference. A practical hydrogen electrode uses a platinum surface (often plated with platinum black) in contact with hydrogen gas and the test solution. Preparation of a hydrogen electrode requires careful cleaning and platinising of the platinum, removal of chlorine traces, and storage in distilled water. The electrode must be operated under oxygen-free conditions and with hydrogen at known pressure when used as a standard.
Disadvantages of the hydrogen electrode include sensitivity to poisoning of the platinum black (by substances such as arsenic, sulphides, cyanide, mercury, surfactants), and practical difficulties in routine operation. Therefore, more convenient subsidiary reference electrodes are widely used (e.g., calomel and silver-silver chloride electrodes).
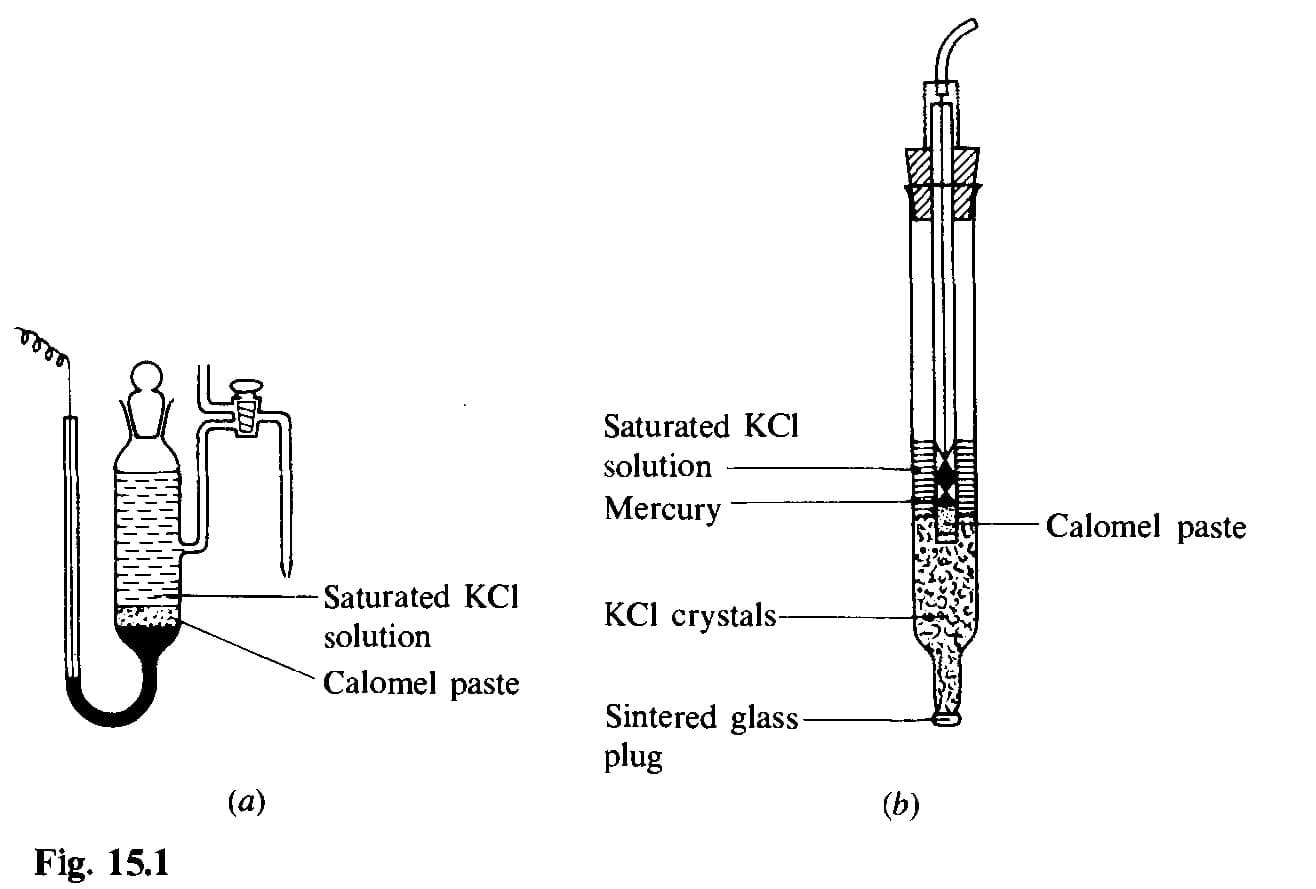
The calomel electrode
The calomel electrode (mercury-mercurous chloride) is a widely used reference electrode because it is convenient and has a fairly stable potential. It consists of metallic mercury in equilibrium with mercury(I) chloride (calomel) and a potassium chloride solution of known concentration (commonly 0.1 M, 1.0 M or saturated). These are commonly termed 0.1 M (decimolar), 1.0 M (molar) and saturated calomel electrodes (S.C.E.). Typical potentials vs SHE at 25 °C are approximately:
- 0.1 M KCl (decimolar): about +0.3358 V
- 1.0 M KCl (molar): about +0.2824 V
- Saturated KCl (S.C.E.): about +0.2444 V
The electrode reaction is:
Hg2Cl2(s) + 2e- ⇌ 2Hg(l) + 2Cl-
The potential depends on chloride activity in the internal solution. The S.C.E. is commonly used because saturated KCl suppresses liquid junction potentials, but its potential is sensitive to temperature (KCl solubility varies with temperature). Decimolar and molar calomel electrodes have potentials less sensitive to temperature changes.
Practical calomel electrodes use a calomel paste layer above mercury and a porous contact (sintered glass) to the external solution. Commercial electrodes often include ion-exchange junctions or double junctions to reduce contamination of the test solution. Where chloride must be avoided, alternatives such as mercury(I) sulphate electrodes can be used.
The silver-silver chloride electrode
The silver-silver chloride electrode consists of a silver wire coated with silver chloride and immersed in a KCl solution whose chloride activity controls the electrode potential. Saturated KCl is often used; 1.0 M and 0.1 M KCl are also employed. The electrode is robust and widely used, particularly in combination glass electrodes for pH measurement. Commercial electrodes mirror calomel electrode construction features (porous junctions, double junctions, ion-exchange membranes).
| Electrode | Solution / condition | 15 °C | 20 °C | 25 °C | 30 °C |
|---|---|---|---|---|---|
| Calomel | KCl (saturated) (S.C.E.) | 0.2512 | 0.2477 | 0.2444 | 0.2409 |
| 1.0 M KCl | 0.2852 | 0.2838 | 0.2824 | 0.2810 | |
| 0.1 M KCl | 0.3365 | 0.3360 | 0.3358 | 0.3356 | |
| Mercury(I) sulphate | K2SO4 (saturated) | 0.656 | |||
| 0.05 M H2SO4 | 0.680 | ||||
| Silver-silver chloride | KCl (saturated) | 0.2091 | 0.2040 | 0.1989 | 0.1939 |
| 1.0 M KCl | 0.2272 | ||||
| 0.1 M KCl | 0.2901 |
Indicator electrodes - general discussion
The indicator electrode is chosen so that its potential is a function of the activity of the ion to be measured. For metal ions, simple metal rods (e.g., Ag, Cu) or metal-plated platinum wires (clean, oxide-free) are commonly used. For hydrogen ion measurements the most common indicator is the glass electrode. Membrane electrodes (glass or other solid membranes) whose potential varies linearly with pH (or other ion activities) are the basis of a wide class of ion-selective electrodes (ISEs).
Anionic determinations can use gas electrodes (e.g., oxygen electrode for O2 or chlorine electrode for Cl2) or second-kind electrodes (e.g., silver-silver chloride for Cl-). Choice of indicator electrode depends on the titration or determination type: glass electrode for acid-base, silver electrode for halide-silver precipitation titrations, platinum wire for redox titrations.
An example of a rugged hydrogen-ion sensor is the antimony electrode. The surface oxide equilibrates according to:
Sb2O3(s) + 6H+ + 6e- ⇌ 2Sb(s) + 3H2O
giving a potential approximately proportional to pH over a limited range (about pH 3-8). Antimony electrodes are cheap and robust but need calibration and have limitations in strongly oxidising or complexing media.
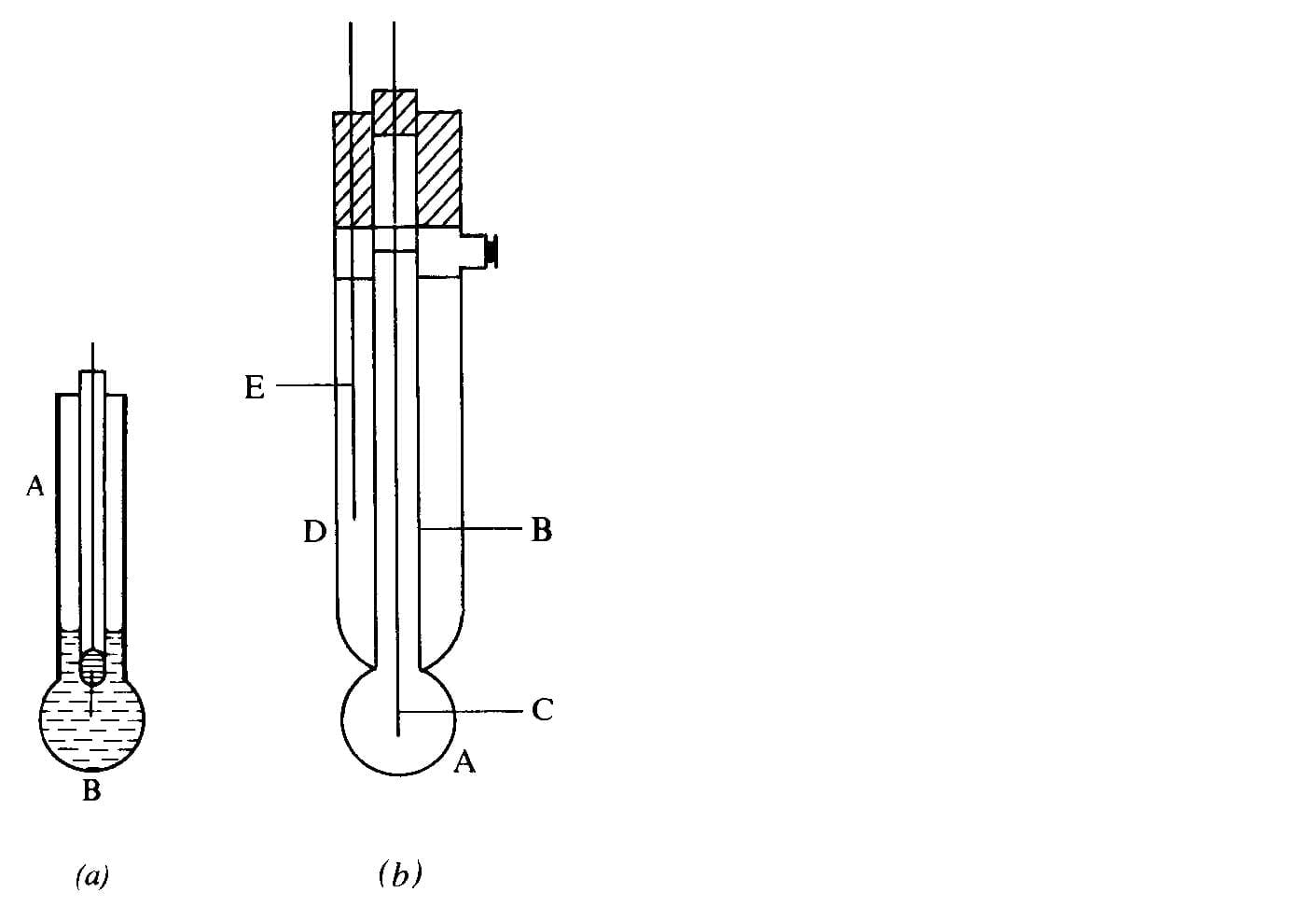
The glass electrode
The glass electrode develops a potential across a thin hydrated surface layer of glass that depends on hydrogen ion activity. A typical arrangement is a thin glass bulb filled with an internal solution (commonly 0.1 M HCl) and an internal Ag/AgCl reference. The external surface contacts the sample. Combination electrodes include both the glass bulb (indicator) and a reference electrode in one body.
Because the glass membrane has high resistance, measurement requires specialised high-impedance instrumentation. The electrode potential can be written as
E = K + (RT/F) ln aH+
and at 25 °C
E = K + 0.0591·pH
Here K is an electrode constant that depends on the glass composition and the electrode's asymmetry potential; practically every glass electrode must be calibrated frequently using buffer solutions. The glass response originates from ion-exchange equilibria in the hydrated glass layer, for example
H+soln + Na+glass ⇌ H+glass + Na+soln
Glass composition is important. Traditional lime-soda glass (approx. SiO2 72%, Na2O 22%, CaO 6%) gave good response over pH 1-9 but suffered an alkaline error at high pH in presence of alkali-metal ions. Lithium-containing glasses (Li2O-rich) reduce alkaline error and are used for high-pH applications. Glass electrodes must be stored wet and soaked before use; they tolerate oxidants and reductants better than many other electrodes and are useful in viscous media and biological samples.
Alkali metal ion-selective glass electrodes
By modifying glass composition (introducing Al2O3, Li2O etc.), glass electrodes selective for other alkali metals (Li+, Na+, K+) are possible. These electrodes still show some H+ sensitivity, so the hydrogen ion concentration of the sample must be kept sufficiently low relative to the analyte.
| Glass composition (approx.) | For determination of |
|---|---|
| Na2O 22%, CaO 6%, SiO2 72% | H+ (subject to alkaline error) |
| Li2O 28%, Cs2O 2%, BaO 4%, La2O3 3%, SiO2 63% | H+ (alkaline error reduced) |
| Li2O 15%, Al2O3 25%, SiO2 60% | Li+ |
| Na2O 11%, Al2O3 18%, SiO2 71% | Na+, Ag+ |
| Na2O 27%, Al2O3 5%, SiO2 68% | K+, NH4+ |
The indicator response follows the Nernst equation:
E = k + (RT/nF) ln aM+
For a singly charged cation at 25 °C: E = k - 0.0591·pM, where pM = -log10aM+.
Interference from another cation C+ is described using an exchange equilibrium between ions in the glass and in solution. The electrode potential in presence of an interfering ion can be expressed as a function of aM+, aC+ and a selectivity coefficient kpot(M,C). The selectivity coefficient quantifies how much the interfering ion affects the electrode response; smaller values indicate less interference.
Other solid-membrane electrodes
Membranes other than glass can be used: single crystals, pressed pellets of crystalline salts, or crystalline salt dispersed in a polymer. Examples:
- Silver iodide in polymer: iodide-selective electrodes made by dispersing AgI in silicone rubber; the membrane supports an Ag/AgCl or Ag/AgI inner reference and produces a potential dependent on external I- activity.
- Lanthanum fluoride (LaF3) single-crystal electrode: a fluoride-selective electrode where F- migration through the crystal produces a potential proportional to fluoride activity.
- Pressed-pellet silver sulphide electrodes: respond to Ag+ activity or (as electrodes of the second kind) to S2- activity via the solubility product of Ag2S. Mixed pellets (e.g., Ag2S + AgCl) produce halide-selective electrodes.
Ion-exchange (PVC membrane) electrodes
Ion-selective membranes can be prepared by incorporating an ion-exchange material or ionophore into a polymer matrix (typically poly(vinyl chloride), PVC). The membrane is cast from a solvent; a disc is cut and sealed to form the membrane end of an electrode body. Such membranes can be selective for many ions depending on the exchanger or neutral carrier used.
Examples:
- Calcium electrode: uses a dialkyl phosphate exchanger dissolved in a plasticiser; the membrane reacts with Ca2+ to form surface species whose activity controls the electrode potential. Acidic media can reverse the reaction and limit the electrode use. Changing the membrane composition gives cross-sensitivity to Mg2+ and other divalent ions, useful for 'water hardness' measurements.
- Nitrate electrode: uses quaternary ammonium salts (e.g., tridodecylmethylammonium nitrate) as exchanger in the membrane.
- Perchlorate electrode: membranes containing suitable complexing salts respond to ClO4-.
Neutral ionophores (e.g., valinomycin for K+) are widely used. Valinomycin selectively complexes K+, extracting it into the membrane and producing a Nernstian response for potassium with high selectivity over Na+.
Enzyme-based electrodes
Enzyme electrodes convert a non-ionic or neutral analyte into an ionic species that can be detected by an ion-selective electrode. The enzyme can be immobilised in a gel or on a support applied to the sensing membrane.
Example: urease-based urea electrode. Urease catalyses:
CO(NH2)2 + H2O + 2H+ (urease) → 2NH4+ + CO2
The produced NH4+ is sensed by an ammonium-responsive electrode (glass or NH4+ ISE), and the result is related to urea concentration. Other enzyme-electrode combinations are used for penicillin, glucose (via oxidase producing H+ or H2O2), etc.
For such enzyme electrodes the general electrode response can be written in the same Nernst form appropriate to the ion produced by the enzymatic reaction; for urease-based electrode at 25 °C one often finds
E = k + 0.0295·log aurea
Gas-sensing electrodes
Gas-sensing electrodes analyse gases that are absorbed into solution (NH3, CO2, NO2, SO2, H2S). The gas is passed through or contacted with a small internal liquid compartment across a gas-permeable membrane; the internal solution composition is chosen so that the dissolved gas produces a measurable ionic change detected by an ion-selective electrode (often a pH glass electrode). Membranes can be microporous (PTFE, polypropylene) or thin homogeneous films (silicone rubber).
Ion-selective field-effect transistors (ISFETs)
ISFETs replace the metal gate of a MOSFET with a thin ion-sensitive membrane. Ion activity at the membrane surface modulates the transistor channel current. ISFETs are compact, require very small sample volumes, and are used in biochemical and miniaturised sensing applications. Calibration against standards is required as with classical ISEs.
Commercial ion-selective electrodes - detection limits and considerations
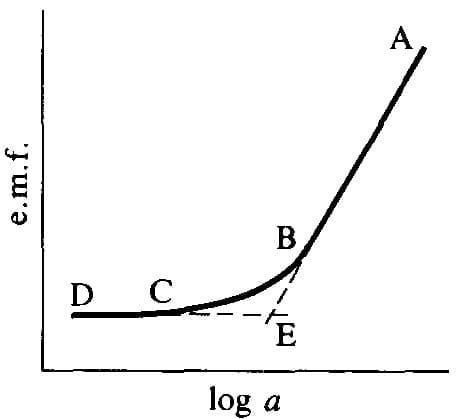
Many ISEs are commercially available for a wide range of ions. Two key practical aspects are the usable concentration (activity) range and the response time. The typical response curve (E vs log a) has a Nernstian linear region at moderate-to-high activities, a curved low-activity region, and a plateau at very low activities. The IUPAC detection limit is where the extrapolated linear portion intersects the extrapolated Nernstian portion.
Response time is defined as the time for the e.m.f. to reach within 1 mV of the equilibrium value; it depends on membrane, interfering ions, stirring, and temperature. Sample and standard solutions should have comparable ionic strength; when this is not possible, add a TISAB (total ionic strength adjuster buffer) to both sample and standards to fix ionic strength and reduce activity-related errors.
| Ion (membrane type) | Lower limit of detection (pX = -log10[X]) | Ion (membrane type) | Lower limit of detection (pX) |
|---|---|---|---|
| Glass membrane - H+ | 13 | Crystalline solid-state - Ag+ | 8 |
| Glass membrane - Na+ | 6 | Crystalline solid-state - Cd2+ | 7 |
| Glass membrane - K+ | 4 | Crystalline solid-state - Cu2+ | 8 |
| Glass membrane - Ag+ | 5 | Crystalline solid-state - Pb2+ | 6 |
| Glass membrane - NH4+ | 5 | Crystalline solid-state - F- | 6 |
| Ion-exchange membrane - Cl- | 4.3 | Ion-exchange membrane - Ca2+ | 6.3 |
| Ion-exchange membrane - Br- | 5.0 | Ion-exchange membrane - K+ (alternative) | 5.0 |
| Ion-exchange membrane - I- | 7.3 | Ion-exchange membrane - Ca2+ + Mg2+ | 5.3 |
| Ion-exchange membrane - CN- | 6 | Ion-exchange membrane - NO3- | 5.3 |
| Ion-exchange membrane - SCN- | 5.3 | Ion-exchange membrane - ClO3- | 5.3 |
| Ion-exchange membrane - S2- | 8 |
Instrumentation and measurement of cell e.m.f.
pH meters and ion meters
Traditional potentiometric measurement used Poggendorff compensation (null) methods. Modern practice uses high-input-impedance electronic voltmeters (pH meters, ion meters) that read directly in pH or activity (or millivolt). Such instruments require electrode calibration and temperature compensation because the Nernst slope depends on temperature.
Important instrument features:
- Calibration (Set buffer): adjust instrument reading with electrode assembly placed in a buffer of known pH.
- Temperature compensation: manual set or automatic using a temperature probe; corrects slope and potential for measurement temperature.
- Slope control: used to correct deviations from ideal Nernst slope between two calibration buffers.
- Isopotential pH: the pH at which cell e.m.f. is independent of temperature; some meters include an isopotential control to compensate electrode system differences.
Typical procedure for pH measurement
- Switch on the instrument and allow warm-up if needed. Prepare buffer solutions for calibration.
- Set temperature control (manual) or insert temperature probe into a buffer (automatic compensation).
- Insert electrode assembly into the first buffer and set the 'Set buffer' control to match the known pH.
- Rinse electrode, place in the second buffer. Adjust 'Slope' so the instrument reads the known pH of the second buffer.
- Re-check first buffer. Repeat until calibration is stable.
- Rinse electrode and measure the test solution. After use rinse and store electrode appropriately (keep moist).
Direct-reading ion meters for ISEs operate similarly; two or more standards with known pX (-log activity) are used to calibrate slope and offset. Most ion meters have a millivolt mode and can be scaled to ion activity once slope and offset are determined.
Practical considerations and care of electrodes
- Always clean and rinse electrodes with distilled/deionised water between measurements.
- Store glass and many ISEs moist (in suitable storage solution) to preserve the hydrated surface layer or membrane conditioning.
- Replace clogged porous junctions (sintered discs) or use double-junction electrodes to protect samples from internal electrode additives (e.g., Hg+, Cl-).
- Be aware of interfering ions and adjust ionic strength (TISAB) and pH to minimise interferences.
- Regularly calibrate electrodes and check slope; replace or refurbish membranes or internal solutions as recommended by manufacturers.
Key reactions and equations (collected)
Hg2Cl2(s) + 2e- ⇌ 2Hg(l) + 2Cl-
Sb2O3(s) + 6H+ + 6e- ⇌ 2Sb(s) + 3H2O
E (antimony) = E°Sb2O3,Sb - 0.0591·pH (at 25 °C, approximate relation)
E = K + (RT/F) ln aH+
E = K + 0.0591·pH (25 °C)
H+soln + Na+glass ⇌ H+glass + Na+soln
E = k + (RT/nF) log aM+
E = k - 0.0591·pM (for singly charged cation at 25 °C)
Mglass+ + C+soln ⇌ Cglass+ + M+soln (exchange equilibrium)
Kex = (aM+·a′C+)/(a′M+·aC+)
E = KM + (2.303 RT/nF) ln (aM+ + kpot(M,C)·aC+)
kM,C = (Response to C+)/(Response to M+)
Urease reaction: CO(NH2)2 + H2O (urease) → 2NH4+ + CO2
E (urea electrode) = k + 0.0295·log aurea (indicative relation)

