PE Exam Exam > PE Exam Notes > Mechanical Engineering for PE > Cheatsheet: Laws Of Thermodynamics
Cheatsheet: Laws Of Thermodynamics
1. Fundamental Concepts
1.1 Thermodynamic Systems
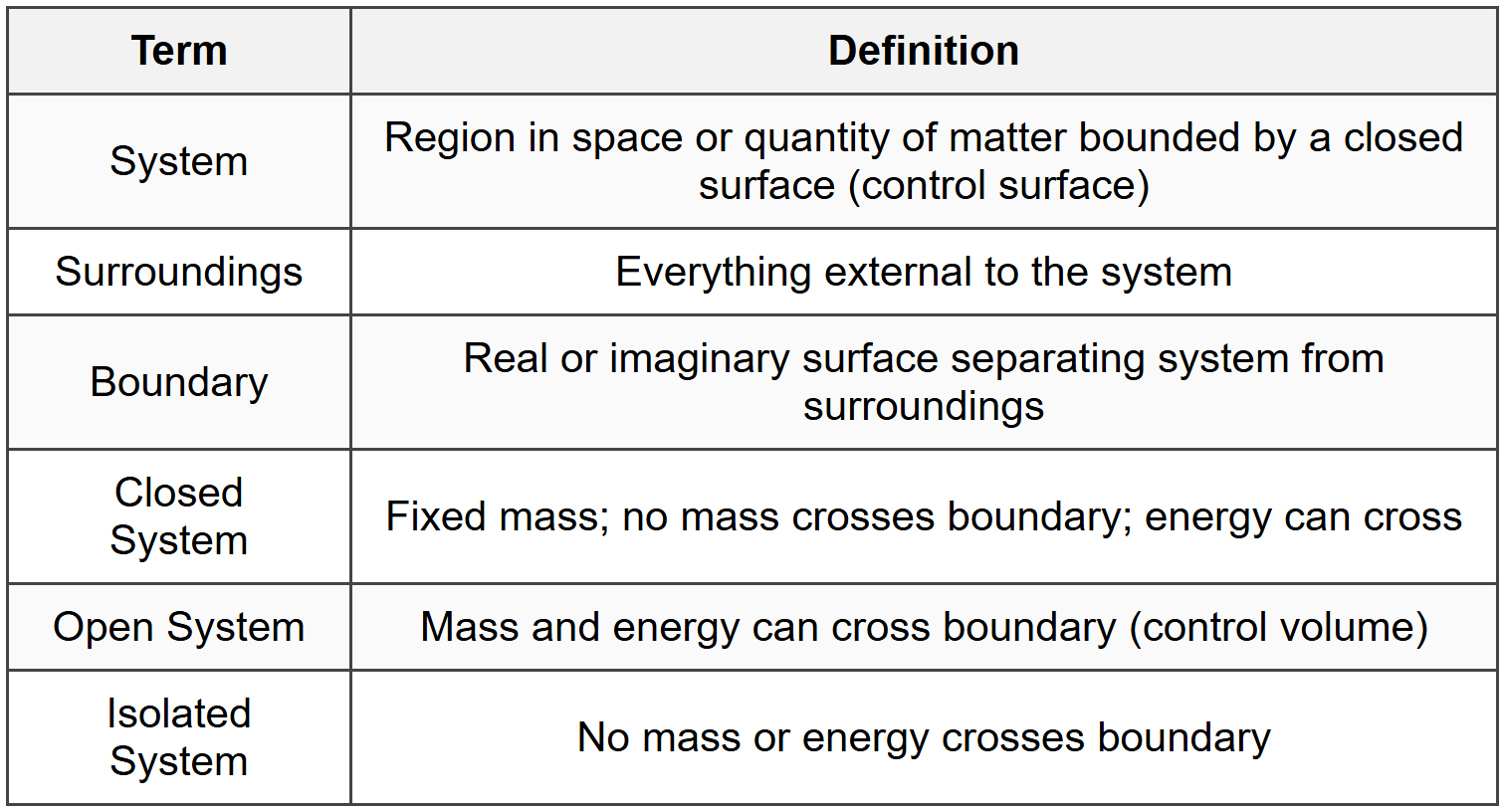
1.2 Properties and State

1.3 Sign Conventions
- Heat added to system: Q > 0 (positive)
- Heat rejected from system: Q < 0="">
- Work done by system: W > 0 (positive)
- Work done on system: W < 0="">
2. Zeroth Law of Thermodynamics
2.1 Statement and Principle

3. First Law of Thermodynamics
3.1 Energy Conservation Principle

3.2 Closed System Formulations
3.2.1 General Energy Equation
- ΔE = ΔU + ΔKE + ΔPE = Q - W
- For stationary systems: ΔKE = 0, ΔPE = 0
- Simplified: ΔU = Q - W
- Per unit mass: Δu = q - w
3.2.2 Work Forms
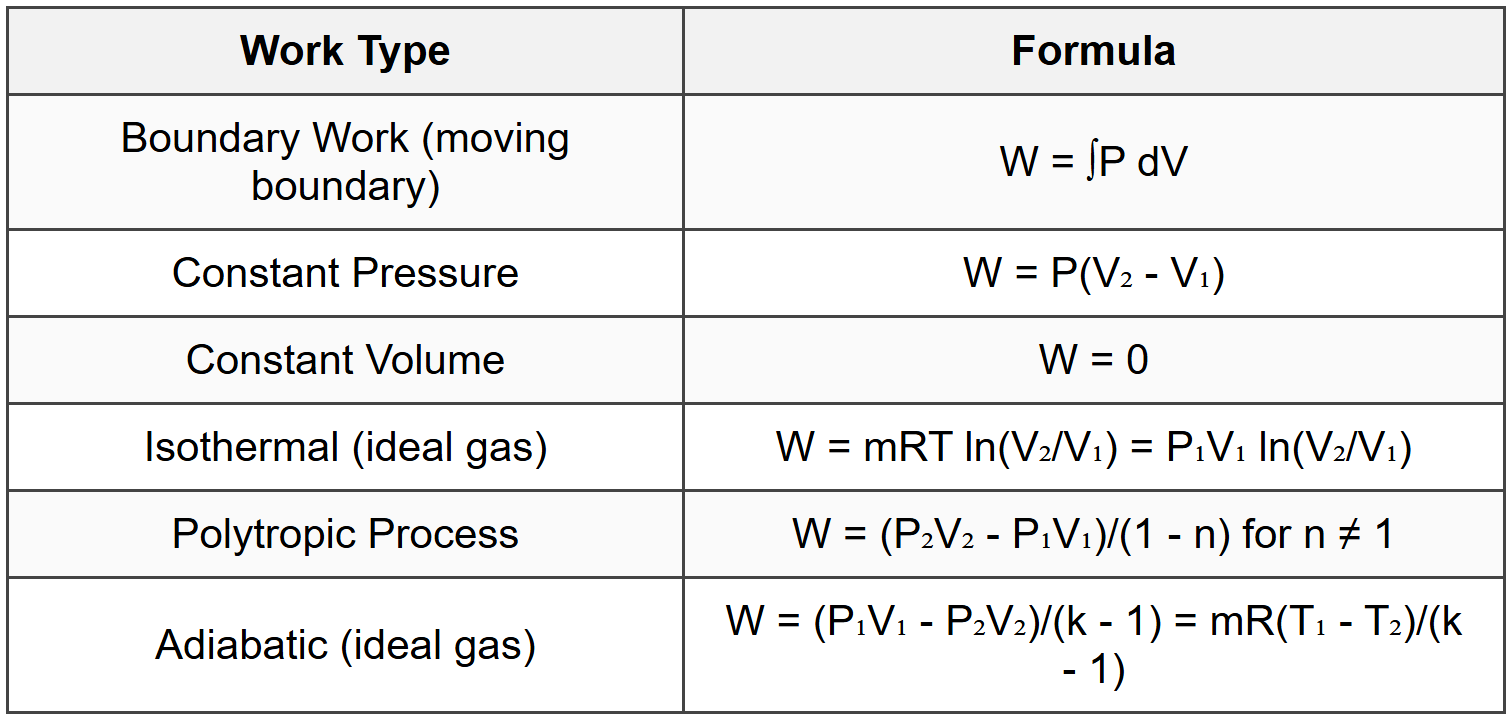
3.3 Open System (Control Volume)
3.3.1 General Energy Balance
- dE_cv/dt = Q̇_cv - Ẇ_cv + Σ_in ṁ(h + V²/2 + gz) - Σ_out ṁ(h + V²/2 + gz)
- h = enthalpy = u + Pv
- Flow work included in enthalpy term
3.3.2 Steady-State Steady-Flow (SSSF)

3.4 Specific Applications
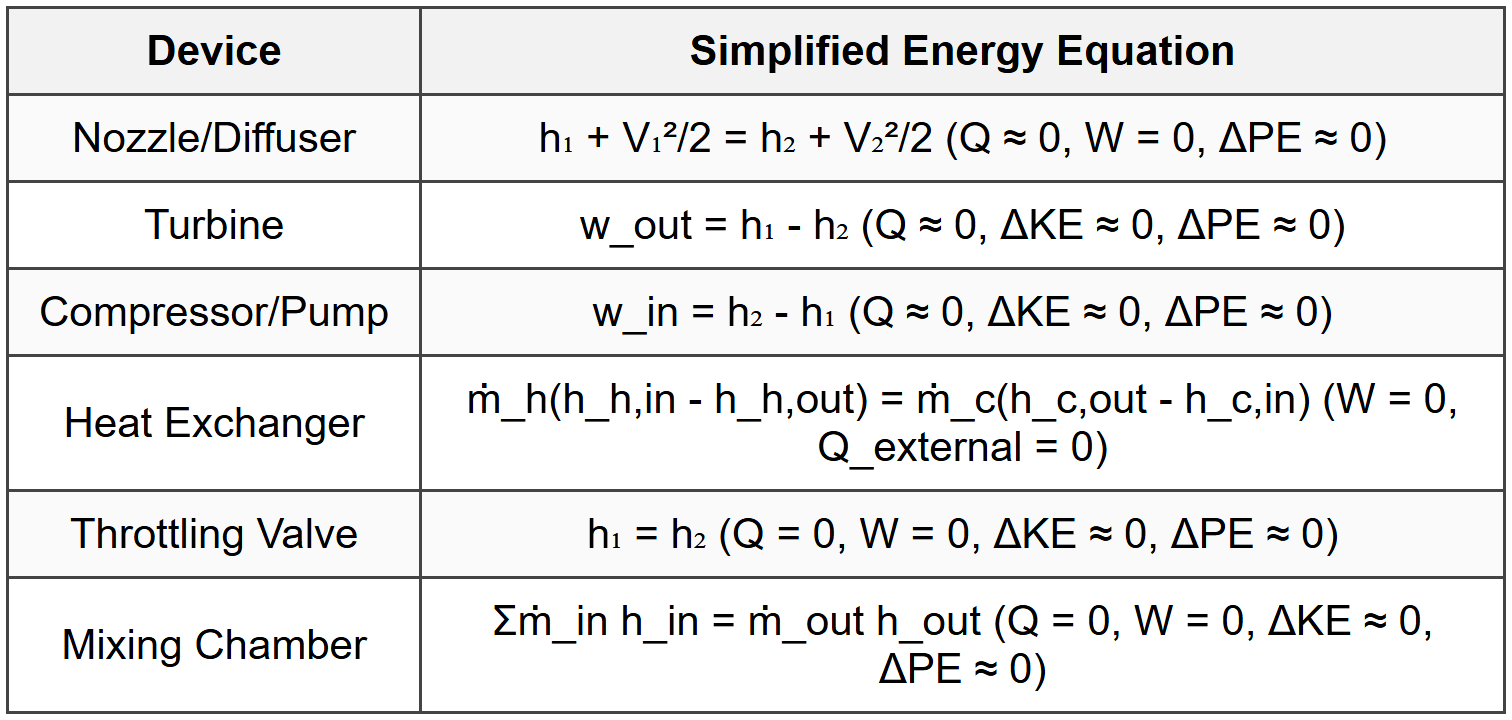
3.5 Enthalpy Relations

4. Second Law of Thermodynamics
4.1 Statements of Second Law

4.2 Entropy
4.2.1 Fundamental Relations

4.2.2 Entropy Change Calculations

4.3 Entropy Balance
4.3.1 Closed System
- ΔS_system = ∫δQ/T_b + S_gen
- S_gen ≥ 0 (irreversibilities measure)
- Isolated system: ΔS_isolated = S_gen ≥ 0
4.3.2 Open System (Control Volume)
- dS_cv/dt = Σ(Q̇_k/T_k) + Σ_in ṁs - Σ_out ṁs + Ṡ_gen
- Steady state: 0 = Σ(Q̇_k/T_k) + Σ_in ṁs - Σ_out ṁs + Ṡ_gen
- Single stream: ṁ(s₂ - s₁) = Q̇/T_b + Ṡ_gen
4.4 Reversibility and Irreversibility
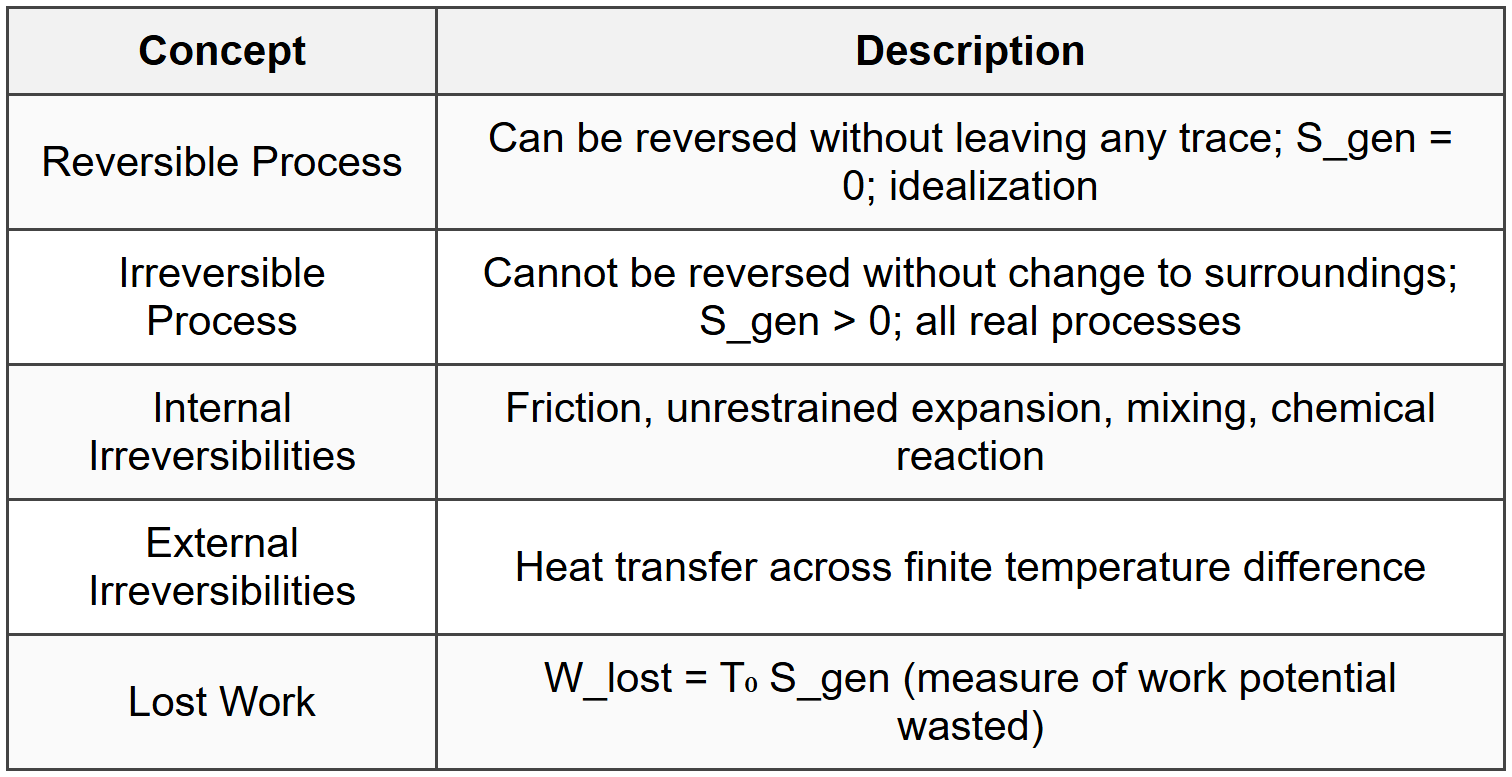
4.5 Isentropic Efficiency

5. Carnot Cycle and Efficiency
5.1 Carnot Cycle

5.2 Carnot Efficiency and COP
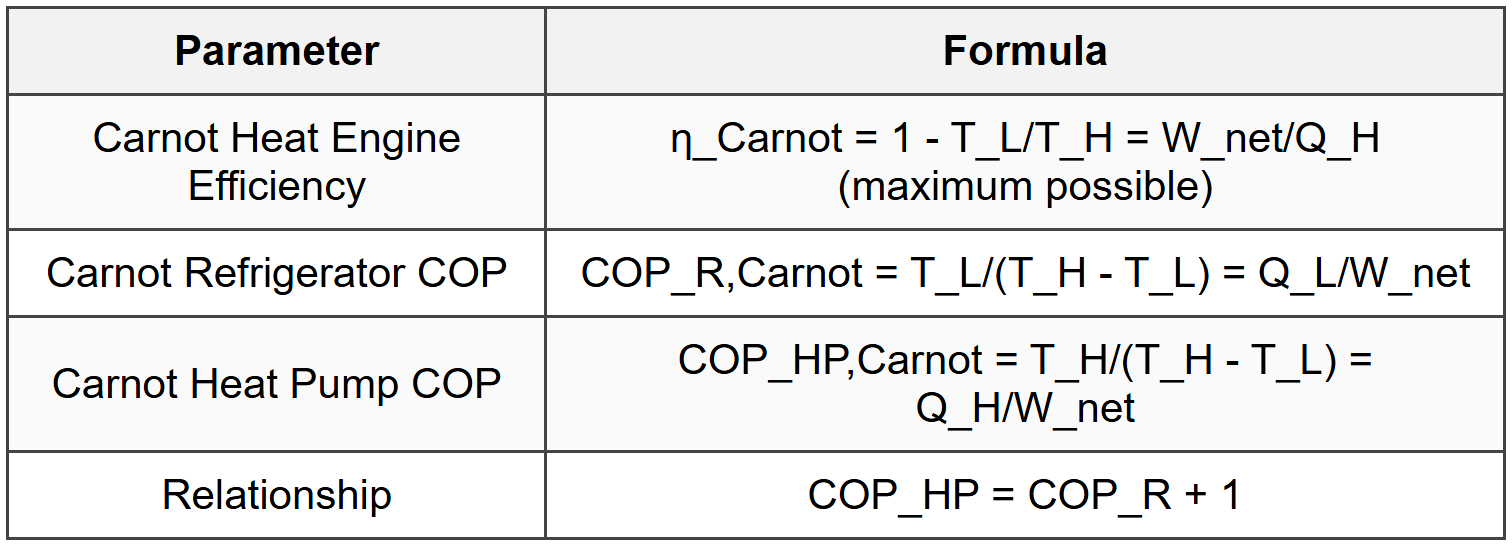
5.3 Carnot Principles
- Efficiency of irreversible engine < efficiency="" of="" reversible="" engine="" (between="" same="">
- All reversible engines between same two reservoirs have same efficiency
- Carnot efficiency depends only on reservoir temperatures
- No engine can be more efficient than Carnot engine operating between same reservoirs
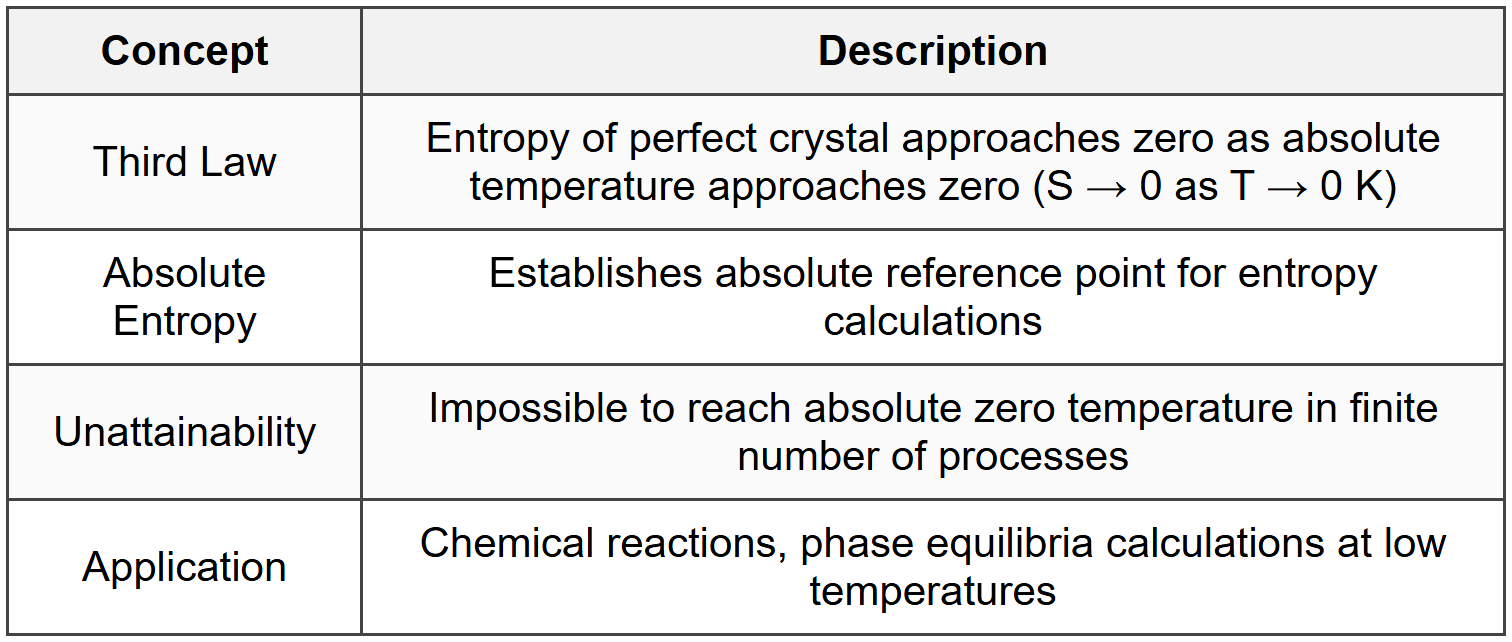
6. Third Law of Thermodynamics
6.1 Statement and Implications
7. Important Thermodynamic Relations
7.1 Maxwell Relations
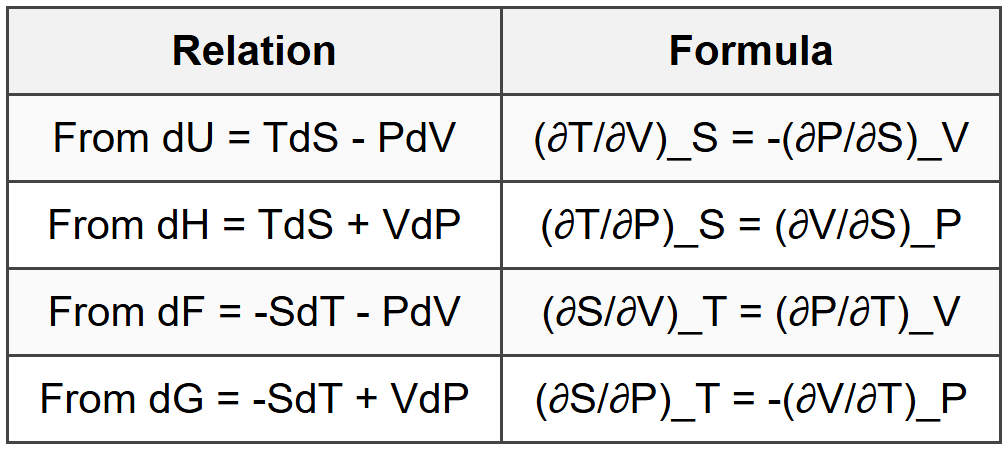
7.2 Tds Relations
- Tds = du + Pdv (first Tds equation)
- Tds = dh - vdP (second Tds equation)
- Both valid for any process (reversible or irreversible)
7.3 Specific Heat Relations

7.4 Gibbs and Helmholtz Functions

8. Key Ideal Gas Relations
8.1 Equations of State
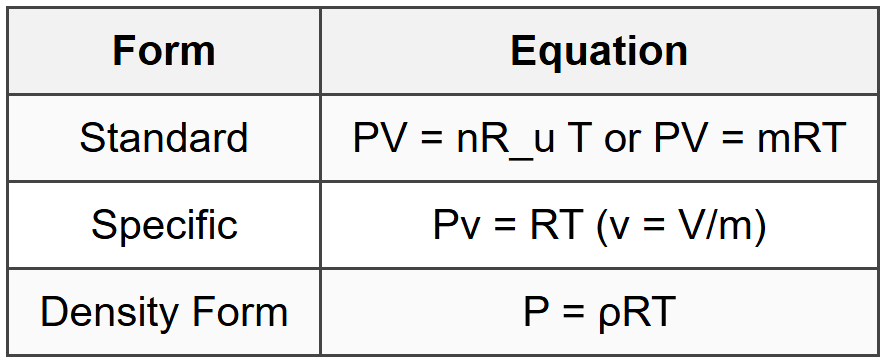
- R = R_u/M where R_u = 8.314 kJ/(kmol·K), M = molecular weight
- R_air = 0.287 kJ/(kg·K)
8.2 Polytropic Process

8.3 Special Cases of Polytropic Process

9. Exergy (Availability)
9.1 Definitions

9.2 Exergy Formulas
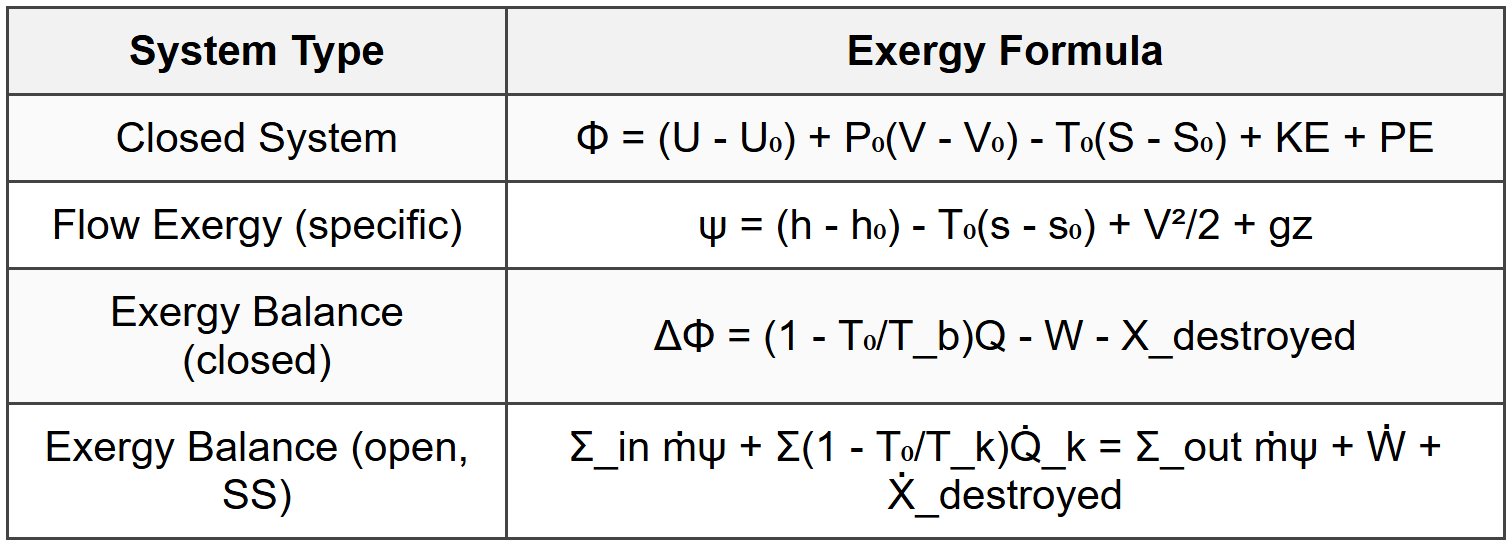
9.3 Second Law Efficiency
- η_II = Exergy recovered/Exergy supplied
- η_II = (Exergy supplied - Exergy destroyed)/Exergy supplied = 1 - X_destroyed/X_supplied
- Measures thermodynamic perfection; 100% for reversible process
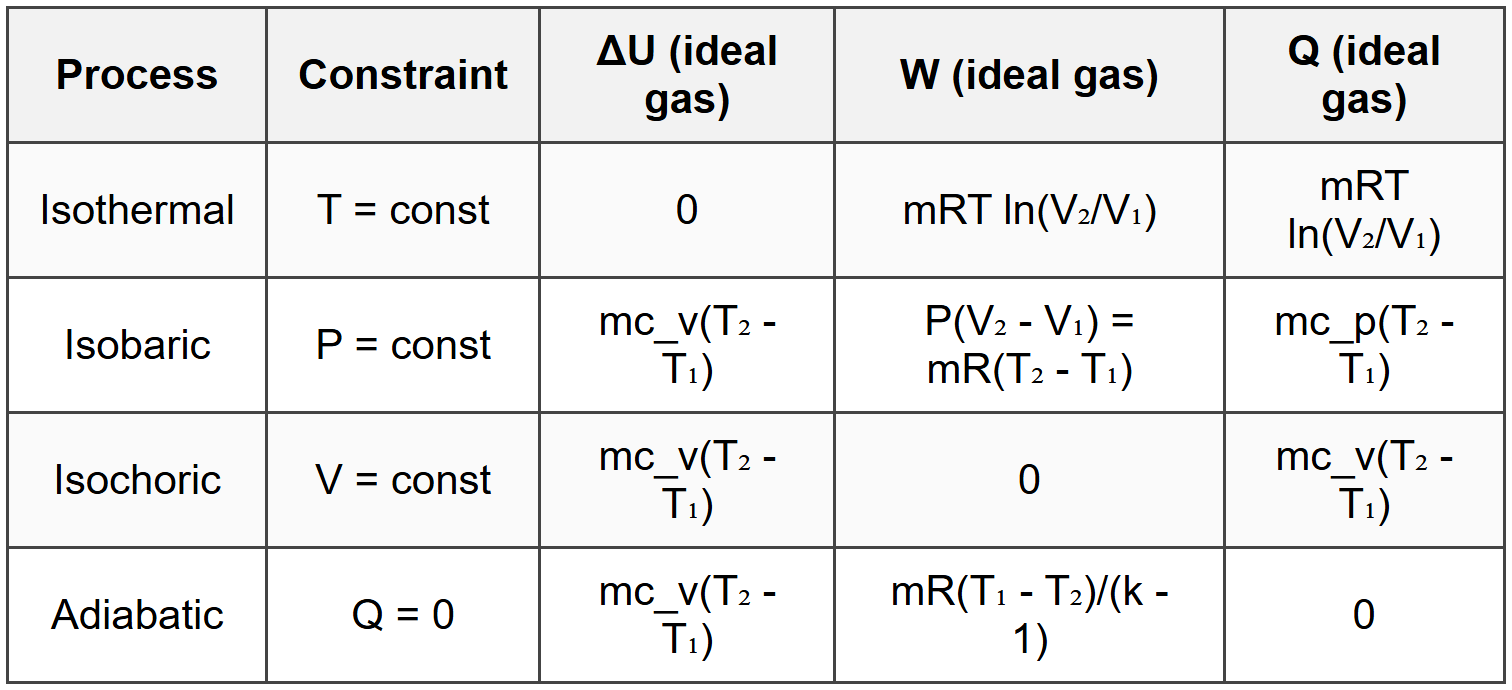
10. Summary of Key Process Equations
10.1 Process Comparison Table
The document Cheatsheet: Laws Of Thermodynamics is a part of the PE Exam Course Mechanical Engineering for PE.
All you need of PE Exam at this link: PE Exam
Related Searches
Cheatsheet: Laws Of Thermodynamics, video lectures, Exam, shortcuts and tricks, Viva Questions, study material, MCQs, Summary, practice quizzes, Cheatsheet: Laws Of Thermodynamics, mock tests for examination, pdf , past year papers, Cheatsheet: Laws Of Thermodynamics, Important questions, Extra Questions, Free, Objective type Questions, ppt, Previous Year Questions with Solutions, Sample Paper, Semester Notes;





