PE Exam Exam > PE Exam Notes > Mechanical Engineering for PE > Cheatsheet: Psychrometrics
Cheatsheet: Psychrometrics
1. Fundamental Properties and Definitions
1.1 Key Psychrometric Properties

1.2 Standard Conditions
- Standard atmospheric pressure: 14.696 psia (101.325 kPa)
- Standard temperature: 68°F (20°C)
- Sea level reference for pressure calculations
2. Psychrometric Equations
2.1 Humidity Ratio

2.2 Relative Humidity

2.3 Enthalpy

2.4 Specific Volume

2.5 Degree of Saturation

3. Psychrometric Chart
3.1 Chart Features and Lines

3.2 Reading Chart Procedures
- Two independent properties required to locate state point
- Common pairs: DBT and RH; DBT and WBT; DBT and DPT; DBT and ω
- DPT found by moving horizontally left from state point to saturation curve
- Enthalpy approximated by following constant WBT line
4. Psychrometric Processes
4.1 Sensible Heating/Cooling
- Horizontal line on chart (constant ω)
- No moisture added or removed
- DBT changes; RH changes inversely
- Heating: RH decreases; Cooling: RH increases
- Heat transfer: q = ṁdacpΔT = ṁda(h2 - h1)
4.2 Cooling and Dehumidification
- Process moves down and left on chart
- Air cooled below dew point; condensation occurs
- Both temperature and humidity ratio decrease
- Apparatus dew point (ADP): theoretical temperature of cooling coil surface
- Sensible heat: qs = ṁdacp(T1 - T2)
- Latent heat: ql = ṁdahfg(ω1 - ω2)
- Total heat: qt = ṁda(h1 - h2)
- Moisture removed: ṁw = ṁda(ω1 - ω2)
4.3 Heating and Humidification
- Process moves up and right on chart
- Both temperature and humidity ratio increase
- Sensible and latent heat added
- Steam injection or evaporative methods
4.4 Evaporative Cooling (Adiabatic Saturation)
- Process follows constant WBT line (approximately constant enthalpy)
- DBT decreases; ω increases; RH increases
- Moves toward saturation curve at constant enthalpy
- No external heat added; water evaporation cools air
- h1 ≈ h2
4.5 Mixing of Air Streams
- Final state lies on straight line connecting initial states
- Location determined by mass flow ratio
- ṁda,3 = ṁda,1 + ṁda,2
- ṁda,3ω3 = ṁda,1ω1 + ṁda,2ω2
- ṁda,3h3 = ṁda,1h1 + ṁda,2h2
- Lever rule: distance ratios inversely proportional to mass ratios
4.6 Chemical Dehumidification
- Uses desiccants (silica gel, activated alumina, lithium chloride)
- Removes moisture without cooling below dew point
- ω decreases; DBT increases due to heat of adsorption
- Process moves down and right on chart
5. Equipment and Applications
5.1 Cooling Coil Performance

5.2 Cooling Tower Performance
- Range: difference between entering and leaving water temperatures
- Approach: difference between leaving water temperature and entering air WBT
- Effectiveness: ε = (Tw,in - Tw,out)/(Tw,in - WBTair,in)
- Lower approach indicates better performance
5.3 Air Conditioning Processes
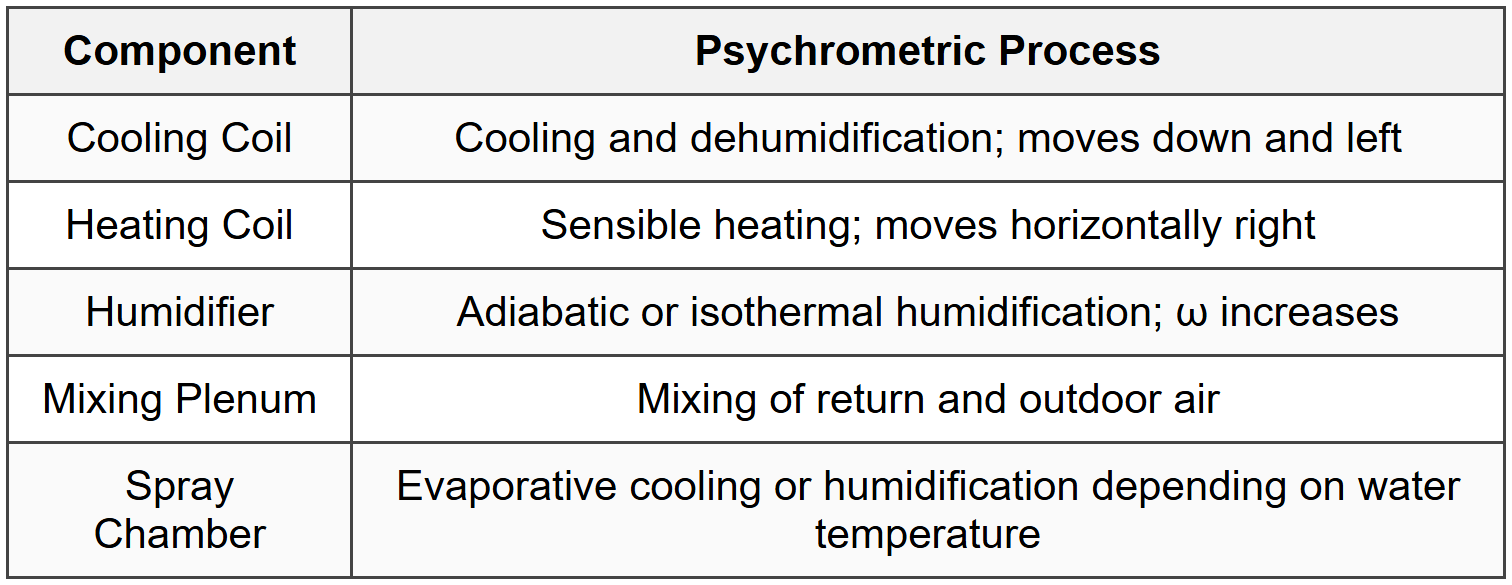
6. Comfort and Load Calculations
6.1 Thermal Comfort Conditions
- Summer comfort: 73-79°F DBT; 30-60% RH
- Winter comfort: 68-75°F DBT; 20-40% RH
- Comfort zone shown on psychrometric chart
6.2 Ventilation Requirements
- Outdoor air required for indoor air quality
- ASHRAE Standard 62.1 specifies ventilation rates
- Minimum outdoor air percentage affects mixed air properties
6.3 Space Load Components
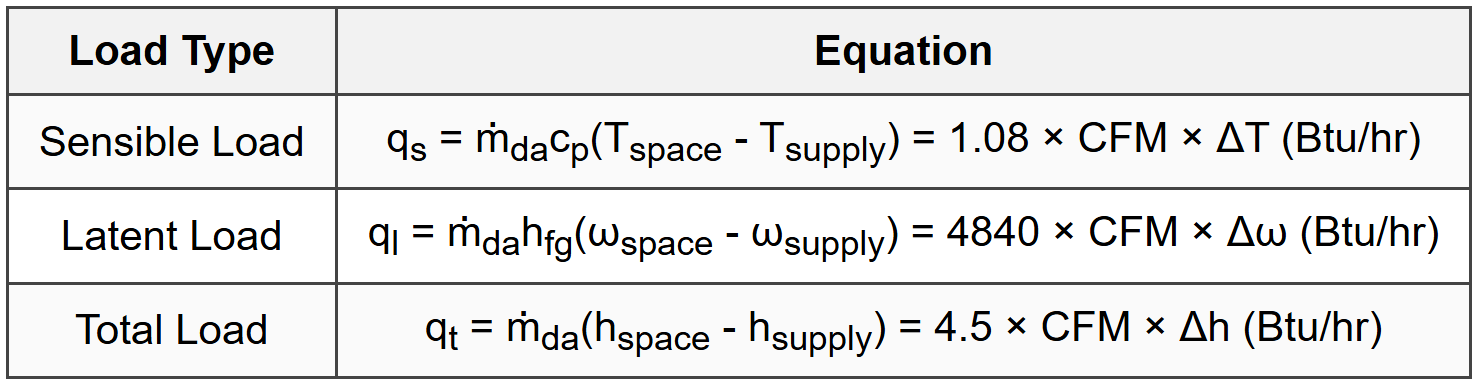
- Factor 1.08 = 60 min/hr × 0.075 lb/ft³ × 0.24 Btu/lb-°F
- Factor 4840 = 60 min/hr × 0.075 lb/ft³ × 1076 Btu/lb
- Factor 4.5 = 60 min/hr × 0.075 lb/ft³
6.4 Room Sensible Heat Factor
- RSHF = qs,room/qt,room
- Used to determine supply air condition on psychrometric chart
- Line from room state point with slope = RSHF intersects supply condition
7. Important Constants and Properties
7.1 Physical Constants

7.2 Molecular Weight Ratio
- Molecular weight water vapor: 18.015 lb/lbmol
- Molecular weight dry air: 28.965 lb/lbmol
- Ratio: 18.015/28.965 = 0.622
- Used in humidity ratio equations
8. Saturation Pressure Correlations
8.1 Antoine Equation

8.2 Hyland-Wexler Equations
- ASHRAE-recommended correlations for saturation pressure
- Valid over wide temperature range
- Used in psychrometric chart generation
9. Common Problem Types
9.1 Problem-Solving Steps
- Identify known and unknown properties
- Plot state points on psychrometric chart
- Determine process path
- Apply mass and energy balances
- Calculate heat transfer and moisture addition/removal
9.2 Mass and Energy Balance Equations
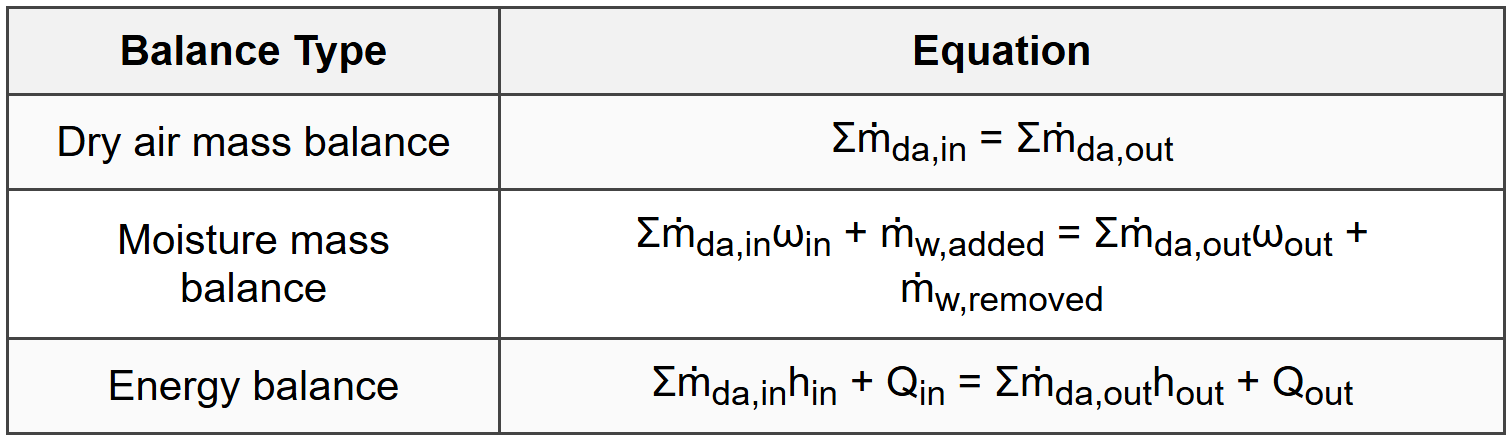
9.3 Key Relationships for Quick Checks
- At saturation: DBT = WBT = DPT
- For any state: DPT ≤ WBT ≤ DBT
- RH increases when air is cooled at constant ω
- Mixing two air streams: result lies on straight line between states
- Sensible cooling: horizontal line to right on chart
- Evaporative cooling: follows constant enthalpy line toward saturation
The document Cheatsheet: Psychrometrics is a part of the PE Exam Course Mechanical Engineering for PE.
All you need of PE Exam at this link: PE Exam
Related Searches
Cheatsheet: Psychrometrics, mock tests for examination, practice quizzes, video lectures, Extra Questions, Summary, Important questions, Free, Semester Notes, MCQs, shortcuts and tricks, Sample Paper, ppt, Exam, Cheatsheet: Psychrometrics, pdf , Objective type Questions, past year papers, Viva Questions, Previous Year Questions with Solutions, Cheatsheet: Psychrometrics, study material;





