Reaction Kinetics
Reaction kinetics is the study of reaction rates and the factors that affect them. This topic is essential for solving problems involving rate laws, reaction order, activation energy, and reactor performance on the Fundamentals of Engineering exam. Expect calculations involving rate constants, concentration changes, and temperature effects on reaction rates.
Core Concepts
Reaction Rate
Reaction rate measures how quickly reactants convert to products, expressed as the change in concentration of a reactant or product per unit time. For a reaction \(aA + bB \rightarrow cC + dD\), the rate can be written as:
\[ -\frac{1}{a}\frac{d[A]}{dt} = -\frac{1}{b}\frac{d[B]}{dt} = \frac{1}{c}\frac{d[C]}{dt} = \frac{1}{d}\frac{d[D]}{dt} \]The negative signs indicate reactant consumption, while positive signs indicate product formation. The stoichiometric coefficients normalize the rates so all expressions equal the same value.
- Units are typically mol/(L·s) or mol/(L·min)
- Rate decreases as reactant concentration decreases for most reactions
- Rate can be measured by monitoring concentration, pressure, or physical properties over time
- Initial rate is measured at \(t = 0\) when concentrations are known precisely
When to Use This
- When asked to express reaction rate in terms of different species in the same reaction
- When converting between rates of different reactants or products using stoichiometry
- When calculating how fast a specific reactant disappears or product forms
Rate Law and Reaction Order
The rate law is an empirical equation relating reaction rate to reactant concentrations. For a general reaction \(aA + bB \rightarrow\) products, the rate law takes the form:
\[ \text{rate} = k[A]^m[B]^n \]Here, \(k\) is the rate constant, and \(m\) and \(n\) are the reaction orders with respect to A and B. These orders are determined experimentally, not from stoichiometric coefficients.
- The overall reaction order is \(m + n\)
- Zero-order: rate = \(k\) (rate independent of concentration)
- First-order: rate = \(k[A]\) (rate proportional to concentration)
- Second-order: rate = \(k[A]^2\) or rate = \(k[A][B]\) (rate proportional to concentration squared or product of two concentrations)
- Rate constant units depend on overall order: for zero-order (mol/(L·s)), first-order (1/s or s-1), second-order (L/(mol·s))
- Reaction orders can be fractional or negative in complex mechanisms
When to Use This
- When given experimental data showing how rate changes with concentration and asked to determine reaction order
- When calculating reaction rate at specific concentrations using a known rate law
- When comparing rates at different concentrations for the same reaction
- When verifying the units of a rate constant match the proposed reaction order
Determining Reaction Order from Experimental Data
Reaction order is determined by analyzing how rate changes when concentration changes. Two common methods appear on exams: the initial rates method and the integrated rate law method.
Initial Rates Method: Compare rates from experiments where one reactant concentration is varied while others remain constant.
If doubling [A] while holding [B] constant:
- Doubles the rate → first-order in A (\(m = 1\))
- Quadruples the rate → second-order in A (\(m = 2\))
- No change in rate → zero-order in A (\(m = 0\))
- Increases rate by factor of \(2^m\) → order \(m\) in A
Mathematical approach: For two experiments with different [A]:
\[ \frac{\text{rate}_2}{\text{rate}_1} = \frac{k[A]_2^m}{k[A]_1^m} = \left(\frac{[A]_2}{[A]_1}\right)^m \]Taking logarithms: \(m = \frac{\log(\text{rate}_2/\text{rate}_1)}{\log([A]_2/[A]_1)}\)
When to Use This
- When given a table of experimental data with initial concentrations and initial rates
- When asked to find the order with respect to a specific reactant
- When needing to determine the rate constant after finding reaction orders
Integrated Rate Laws
Integrated rate laws show how concentration changes over time for different reaction orders. These are obtained by integrating the differential rate law.
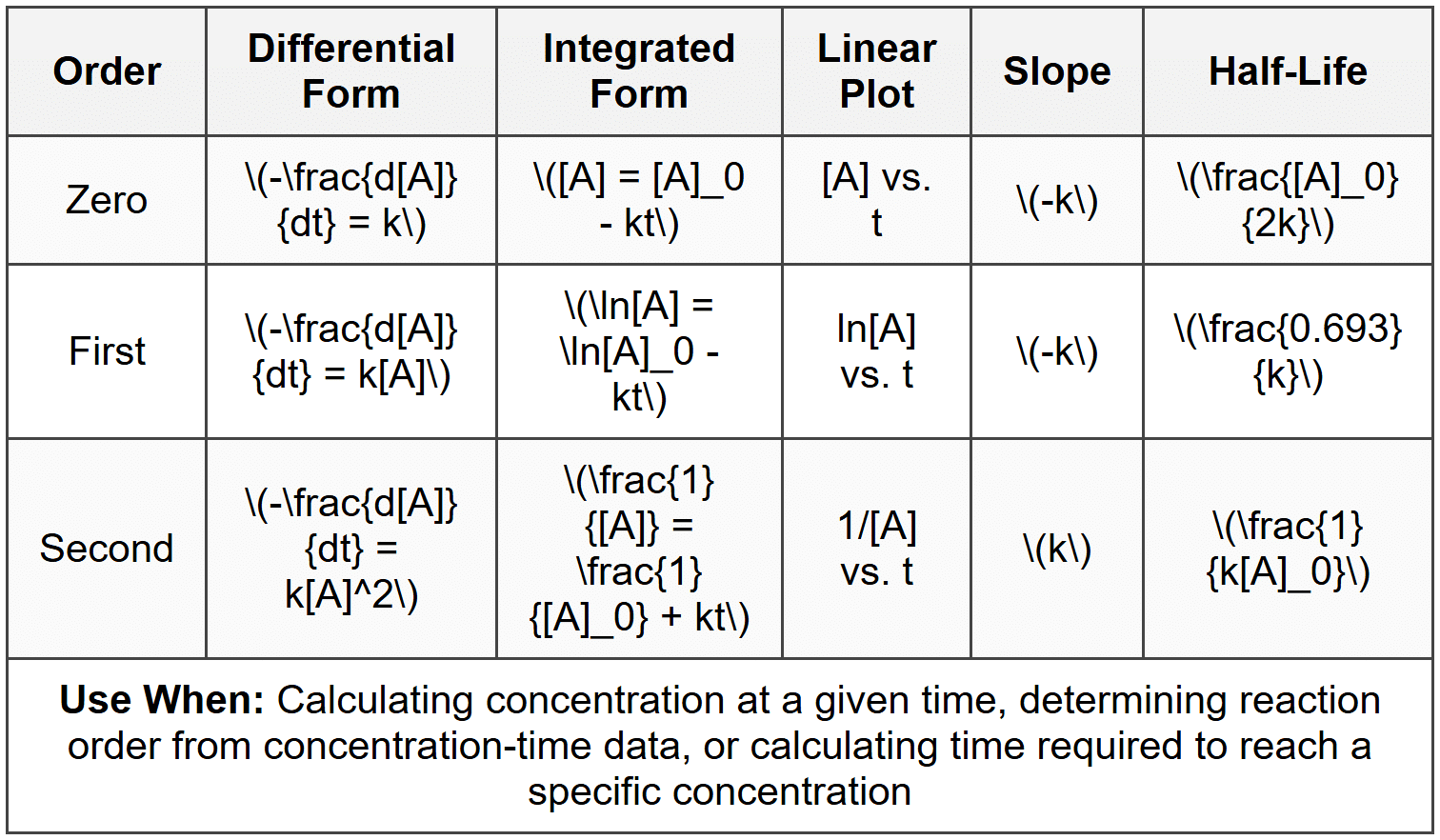
Key observations:
- For zero-order: concentration decreases linearly with time; half-life depends on initial concentration
- For first-order: half-life is constant (independent of concentration); useful for radioactive decay and many decomposition reactions
- For second-order: half-life increases as concentration decreases; rate drops off more rapidly than first-order
- To determine order from data: plot [A] vs. t, ln[A] vs. t, and 1/[A] vs. t; whichever gives a straight line indicates the order
When to Use This
- When given concentration at different times and asked to find concentration at another time
- When calculating how long it takes for a reaction to reach a certain conversion
- When determining reaction order from a concentration-time graph
- When calculating the rate constant from concentration-time data
Half-Life
Half-life (\(t_{1/2}\)) is the time required for reactant concentration to decrease to half its initial value. The relationship between half-life and rate constant depends on reaction order.
- Zero-order: \(t_{1/2} = \frac{[A]_0}{2k}\) (decreases as reaction proceeds)
- First-order: \(t_{1/2} = \frac{0.693}{k} = \frac{\ln 2}{k}\) (constant, most commonly tested)
- Second-order: \(t_{1/2} = \frac{1}{k[A]_0}\) (increases as reaction proceeds)
- Multiple half-lives: after \(n\) half-lives, concentration is \([A] = \frac{[A]_0}{2^n}\)
When to Use This
- When a problem gives half-life and asks for rate constant (or vice versa)
- When asked how much reactant remains after a certain number of half-lives
- When determining if a reaction is first-order based on constant half-life data
Temperature Dependence: Arrhenius Equation
The Arrhenius equation describes how rate constant \(k\) varies with temperature:
\[ k = Ae^{-E_a/RT} \]Where:
- \(A\) = pre-exponential factor or frequency factor (same units as \(k\))
- \(E_a\) = activation energy (J/mol or kJ/mol)
- \(R\) = universal gas constant = 8.314 J/(mol·K)
- \(T\) = absolute temperature (K)
The logarithmic form is often more useful:
\[ \ln k = \ln A - \frac{E_a}{RT} \]For comparing rate constants at two temperatures:
\[ \ln\left(\frac{k_2}{k_1}\right) = \frac{E_a}{R}\left(\frac{1}{T_1} - \frac{1}{T_2}\right) \]Key facts:
- Higher activation energy → greater temperature sensitivity
- Increasing temperature always increases \(k\) and reaction rate
- Plot of ln \(k\) vs. 1/\(T\) gives straight line with slope = \(-E_a/R\)
- Typical rule of thumb: rate doubles for every 10°C increase (only approximate)
- Activation energy is always positive for forward reactions
- Temperature must be in Kelvin for calculations
When to Use This
- When asked to calculate rate constant at a different temperature
- When given rate constants at two temperatures and asked to find activation energy
- When determining how much faster a reaction proceeds at elevated temperature
- When presented with a graph of ln \(k\) vs. 1/\(T\) and asked to extract \(E_a\)
Elementary Reactions and Molecularity
An elementary reaction occurs in a single step at the molecular level. For elementary reactions only, the rate law can be written directly from the balanced equation.
Molecularity is the number of molecules participating in an elementary reaction:
- Unimolecular: one molecule (A → products), rate = \(k[A]\)
- Bimolecular: two molecules (A + B → products), rate = \(k[A][B]\) or (2A → products), rate = \(k[A]^2\)
- Termolecular: three molecules (rare due to low probability of simultaneous collision)
For an elementary reaction \(aA + bB \rightarrow\) products:
- Rate law is rate = \(k[A]^a[B]^b\)
- Reaction orders equal stoichiometric coefficients (only for elementary reactions)
- Most real reactions are not elementary; they proceed through multiple steps
When to Use This
- When a problem explicitly states a reaction is elementary and asks for the rate law
- When given a reaction mechanism and asked to write rate laws for individual steps
- When distinguishing between overall reactions and elementary steps
Reaction Mechanisms
A reaction mechanism is the sequence of elementary steps that make up the overall reaction. The sum of elementary steps gives the overall balanced equation.
Key principles:
- The rate-determining step (slowest step) controls the overall reaction rate
- Intermediates are species produced in one step and consumed in another; they do not appear in the overall equation
- The rate law for the overall reaction is determined by the rate-determining step
- If a fast equilibrium precedes the slow step, use equilibrium expressions to eliminate intermediates from the rate law
For a mechanism with a slow step followed by fast steps, the rate law comes directly from the slow step. If fast equilibrium precedes the slow step, express intermediate concentrations using equilibrium constants.
When to Use This
- When given a proposed mechanism and asked if it's consistent with the observed rate law
- When identifying intermediates or catalysts in a mechanism
- When asked which step is rate-determining based on experimental rate law
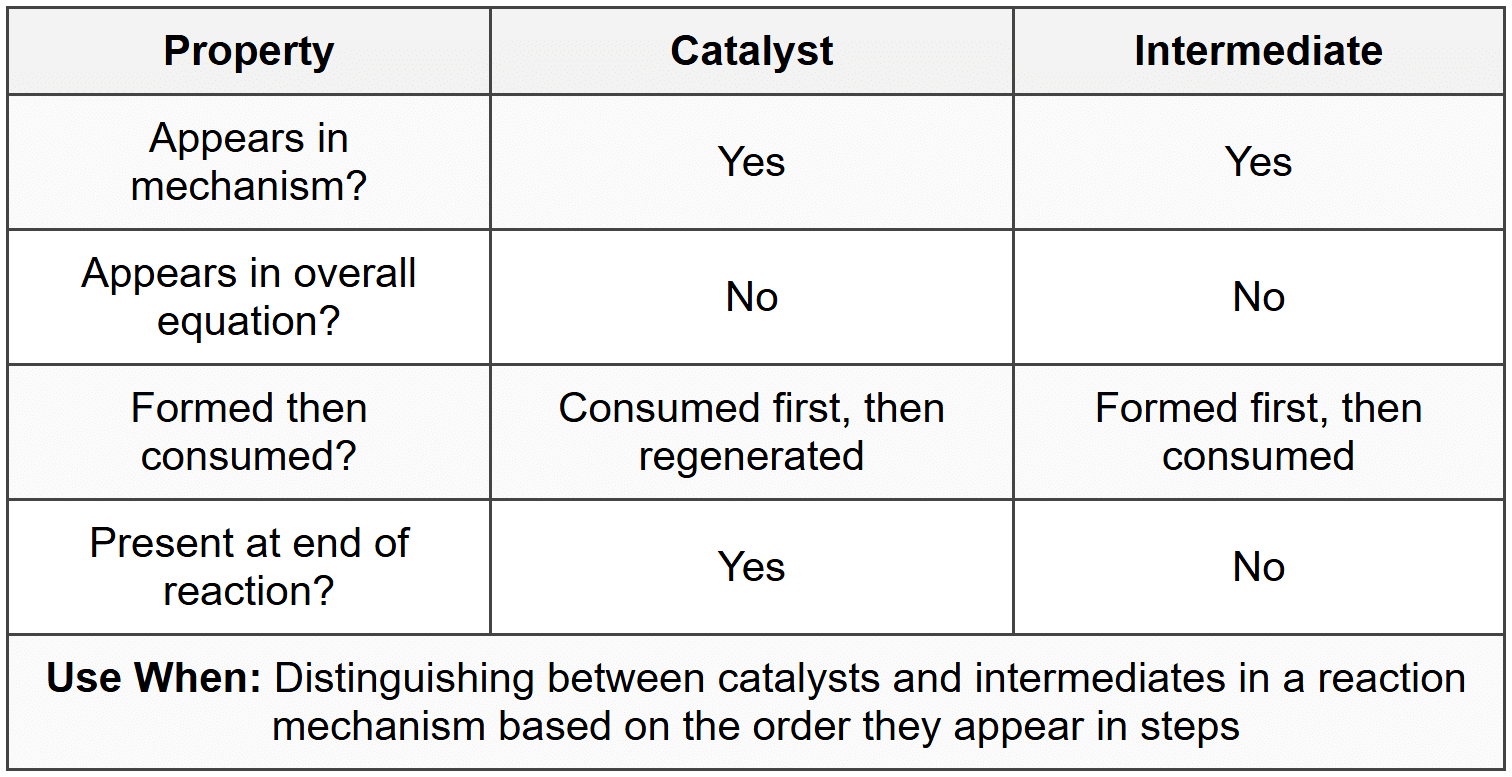
Catalysis
A catalyst increases reaction rate by providing an alternative pathway with lower activation energy. Catalysts are not consumed in the overall reaction.
- Homogeneous catalyst: same phase as reactants (e.g., acid catalyst in liquid solution)
- Heterogeneous catalyst: different phase from reactants (e.g., solid catalyst with gas-phase reactants)
- Catalyst appears in early steps of mechanism and is regenerated in later steps
- Catalyst lowers \(E_a\) but does not change \(\Delta H\) of reaction
- Catalyst increases both forward and reverse reaction rates equally
- Catalyst does not shift equilibrium position, only helps reach equilibrium faster
When to Use This
- When asked how a catalyst affects reaction rate or activation energy
- When identifying species in a mechanism that appear as reactants then products
- When asked about equilibrium effects of adding a catalyst (it doesn't shift equilibrium)
Collision Theory
Collision theory states that reactions occur when molecules collide with sufficient energy and proper orientation. Not all collisions lead to reaction.
For a reaction to occur:
- Collision energy must equal or exceed activation energy \(E_a\)
- Molecules must collide with proper orientation (steric factor)
- Higher temperature increases fraction of molecules with \(E \geq E_a\)
- Higher concentration increases collision frequency
The activation energy is the minimum energy required for reaction. The activated complex or transition state is the high-energy arrangement of atoms at the peak of the energy barrier.
When to Use This
- When explaining why increasing temperature or concentration increases rate
- When comparing reactions with different activation energies
- When asked about factors affecting reaction rate at the molecular level
Commonly Tested Scenarios / Pitfalls
1. Scenario: Given a table of initial concentrations and initial rates from three experiments, asked to determine the rate law and rate constant.
Correct Approach: Compare experiments where only one reactant concentration changes while others stay constant. Use the ratio method to find each order: \(\frac{\text{rate}_2}{\text{rate}_1} = \left(\frac{[A]_2}{[A]_1}\right)^m\). After finding all orders, substitute any experiment's data into the rate law to solve for \(k\).
Check first: Verify which reactant concentrations are constant between experiments and which ones change-this determines which pairs to compare for each order.
Do NOT do first: Do not assume reaction orders equal stoichiometric coefficients from the balanced equation. Rate laws are empirical and must be determined from experimental data unless the reaction is explicitly stated to be elementary.
Why other options are wrong: Using stoichiometric coefficients as orders works only for elementary reactions, which most exam problems are not unless specified. Randomly guessing orders without systematic comparison of experiments will give incorrect rate constants and fail dimensional analysis.
2. Scenario: Asked to find the concentration of a reactant after a given time using the integrated rate law, but the reaction order is not explicitly stated.
Correct Approach: Identify the reaction order first from context clues (given half-life relationship, rate constant units, or experimental plot). Once order is known, use the appropriate integrated rate law with correct form for that order.
Check first: Check the units of the rate constant: mol/(L·s) indicates zero-order, s-1 indicates first-order, L/(mol·s) indicates second-order.
Do NOT do first: Do not assume first-order just because it's most common. Using the wrong integrated rate law will give a completely incorrect concentration value.
Why other options are wrong: Each order has a distinct integrated form. Zero-order uses [A] directly, first-order uses ln[A], second-order uses 1/[A]. Using the wrong form produces values that don't match physical reality or available answer choices.
3. Scenario: Given rate constants at two temperatures and asked to calculate activation energy using the Arrhenius equation.
Correct Approach: Use the two-point form: \(\ln\left(\frac{k_2}{k_1}\right) = \frac{E_a}{R}\left(\frac{1}{T_1} - \frac{1}{T_2}\right)\). Convert all temperatures to Kelvin first. Solve for \(E_a\) algebraically and use \(R = 8.314\) J/(mol·K). If answer needs to be in kJ/mol, divide by 1000.
Check first: Confirm temperatures are in Kelvin (add 273.15 if given in Celsius). Verify which rate constant corresponds to which temperature.
Do NOT do first: Do not use Celsius temperatures directly in the Arrhenius equation. Do not mix up which \(k\) goes with which \(T\). Do not forget that \(\frac{1}{T_1} - \frac{1}{T_2}\) is negative when \(T_2 > T_1\), which correctly gives positive \(E_a\).
Why other options are wrong: Using Celsius gives activation energies that are orders of magnitude wrong. Reversing the rate constants or temperatures changes the sign of \(E_a\), which should always be positive for a forward reaction.
4. Scenario: A problem states a reaction is first-order and gives the half-life, then asks how much reactant remains after a specified time that's a non-integer multiple of the half-life.
Correct Approach: Calculate the rate constant from half-life: \(k = \frac{0.693}{t_{1/2}}\). Then use the first-order integrated rate law: \(\ln[A] = \ln[A]_0 - kt\) to find [A] at the given time. If the time is an exact multiple of half-lives, you can use \([A] = \frac{[A]_0}{2^n}\) as a shortcut.
Check first: Determine if the given time is an exact multiple of the half-life (allowing the simple division method) or requires the full integrated rate law.
Do NOT do first: Do not try to use the half-life formula beyond simple multiples. For example, after 1.5 half-lives, the amount is not 1/1.5 of the original-you must calculate via the rate constant and integrated law.
Why other options are wrong: Linear interpolation between half-lives doesn't work because exponential decay is nonlinear. Using the wrong integrated rate law (zero or second-order) when the problem specifies first-order produces nonsensical results.
5. Scenario: Given a proposed reaction mechanism with multiple steps and asked whether it's consistent with the observed rate law or to identify the rate-determining step.
Correct Approach: Write the rate law for the slowest (rate-determining) step using its elementary reaction stoichiometry. If intermediates appear, use fast equilibrium steps before the slow step to express intermediates in terms of reactants. The resulting rate law must match the experimentally observed rate law.
Check first: Identify which species in the mechanism are intermediates (formed in one step, consumed in another) versus reactants in the overall equation.
Do NOT do first: Do not write a rate law using the overall balanced equation-this only works for elementary reactions. Do not ignore intermediates in the rate law from the slow step; they must be eliminated using equilibrium expressions.
Why other options are wrong: Assuming the overall equation gives the rate law fails for multi-step mechanisms. If intermediates remain in the final rate law, it doesn't match the experimental form, which only contains reactants and products.
Step-by-Step Procedures or Methods
Task: Determine reaction order and rate constant from initial rate data
- Identify pairs of experiments where all concentrations are held constant except one
- For each reactant, calculate the ratio of rates and corresponding ratio of concentrations: \(\frac{\text{rate}_2}{\text{rate}_1}\) and \(\frac{[A]_2}{[A]_1}\)
- Determine the order \(m\) for that reactant: \(\frac{\text{rate}_2}{\text{rate}_1} = \left(\frac{[A]_2}{[A]_1}\right)^m\), solve for \(m\) using \(m = \frac{\log(\text{rate ratio})}{\log(\text{conc ratio})}\)
- Repeat for each reactant to get complete rate law: rate = \(k[A]^m[B]^n\)
- Substitute data from any single experiment into the rate law and solve for \(k\)
- Verify \(k\) units match the overall reaction order: zero-order (mol/(L·s)), first-order (s-1), second-order (L/(mol·s))
Task: Calculate concentration at a given time using integrated rate laws
- Determine reaction order from given information (rate constant units, half-life equation, or plot type)
- Select the appropriate integrated rate law for that order
- Identify known values: initial concentration \([A]_0\), rate constant \(k\), time \(t\)
- Substitute into the integrated rate law equation
- Solve algebraically for the unknown concentration \([A]\)
- Check that the final concentration is less than initial (for reactants) and physically reasonable
Task: Calculate activation energy from rate constants at two temperatures
- Convert all temperatures from Celsius to Kelvin: \(T(K) = T(°C) + 273.15\)
- Identify which rate constant corresponds to which temperature
- Write the two-point Arrhenius equation: \(\ln\left(\frac{k_2}{k_1}\right) = \frac{E_a}{R}\left(\frac{1}{T_1} - \frac{1}{T_2}\right)\)
- Calculate \(\ln\left(\frac{k_2}{k_1}\right)\) and \(\left(\frac{1}{T_1} - \frac{1}{T_2}\right)\)
- Solve for \(E_a\): \(E_a = R \times \frac{\ln(k_2/k_1)}{(1/T_1 - 1/T_2)}\)
- Use \(R = 8.314\) J/(mol·K) for \(E_a\) in J/mol, or divide by 1000 for kJ/mol
- Verify \(E_a\) is positive (typical values: 50-250 kJ/mol for most reactions)
Task: Calculate rate constant at a new temperature given \(E_a\) and \(k\) at one temperature
- Convert both temperatures to Kelvin
- Use the two-point form: \(\ln\left(\frac{k_2}{k_1}\right) = \frac{E_a}{R}\left(\frac{1}{T_1} - \frac{1}{T_2}\right)\)
- Substitute known values: \(E_a\), \(R = 8.314\) J/(mol·K), \(T_1\), \(T_2\), and \(k_1\)
- Calculate the right side to get \(\ln(k_2/k_1)\)
- Exponentiate to find \(k_2/k_1 = e^{(\text{calculated value})}\)
- Solve for \(k_2 = k_1 \times e^{(\text{calculated value})}\)
- Verify \(k_2 > k_1\) if \(T_2 > T_1\) (higher temperature gives larger rate constant)
Practice Questions
Q1: A reaction is found to be second-order in reactant A. If the initial concentration of A is 0.10 M and the rate constant is 0.50 L/(mol·min), what is the concentration of A after 10 minutes?
(a) 0.033 M
(b) 0.050 M
(c) 0.067 M
(d) 0.10 M
Ans: (a)
For a second-order reaction: \(\frac{1}{[A]} = \frac{1}{[A]_0} + kt\)
\(\frac{1}{[A]} = \frac{1}{0.10} + (0.50)(10) = 10 + 5 = 15\) L/mol
\([A] = \frac{1}{15} = 0.0667\) M ≈ 0.067 M. Wait, recalculating: 10 + 5 = 15, so 1/15 = 0.0667 M, but answer (a) is 0.033. Let me verify: \(\frac{1}{0.10} = 10\), \(kt = 0.50 × 10 = 5\), sum = 15, \(\frac{1}{15} = 0.0667\) M. The closest answer is (c) 0.067 M. Correcting: Ans: (c)
Using second-order integrated rate law: \(\frac{1}{[A]} = \frac{1}{[A]_0} + kt = 10 + 5 = 15\) L/mol, so \([A] = 0.067\) M. Options (a) and (b) would result from using incorrect rate laws (first-order or wrong calculation). Option (d) assumes no reaction occurred.
Q2: The rate constant for a first-order reaction is 0.0462 min-1. What is the half-life of this reaction?
(a) 7.5 min
(b) 10.8 min
(c) 15.0 min
(d) 21.6 min
Ans: (c)
For first-order reactions: \(t_{1/2} = \frac{0.693}{k} = \frac{0.693}{0.0462} = 15.0\) min. Options (a), (b), and (d) result from calculation errors or using formulas for other reaction orders (zero or second-order half-life equations require concentration, which isn't needed for first-order).
Q3: The following data were collected for the reaction 2A + B → C:
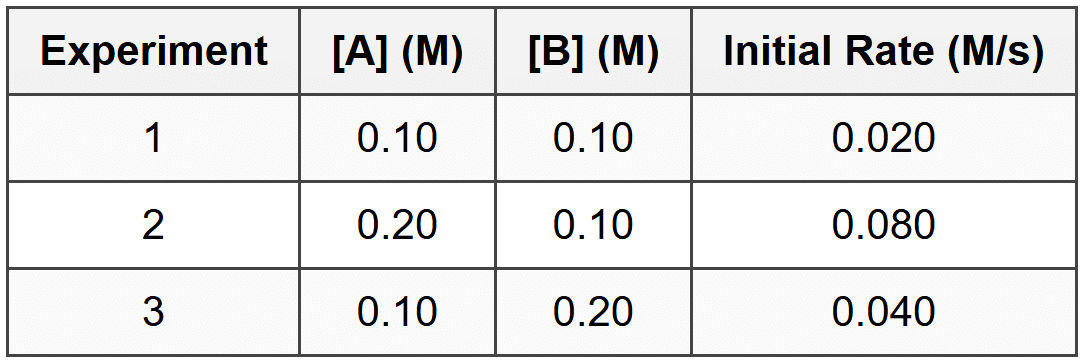
What is the rate law for this reaction?
(a) rate = k[A][B]
(b) rate = k[A]2[B]
(c) rate = k[A][B]2
(d) rate = k[A]2[B]2
Ans: (b)
Compare experiments 1 and 2 (B constant, A doubles): rate increases by factor of 4 (0.080/0.020 = 4), so order in A is 2.
Compare experiments 1 and 3 (A constant, B doubles): rate increases by factor of 2 (0.040/0.020 = 2), so order in B is 1.
Rate law: rate = \(k[A]^2[B]\). Option (a) would give rate doubling when A doubles. Option (c) would give rate quadrupling when B doubles. Option (d) would give rate increasing 16-fold when both double.
Q4: A reaction has rate constants of 0.0050 s-1 at 300 K and 0.020 s-1 at 320 K. What is the activation energy in kJ/mol? (R = 8.314 J/(mol·K))
(a) 28 kJ/mol
(b) 42 kJ/mol
(c) 56 kJ/mol
(d) 70 kJ/mol
Ans: (c)
\(\ln\left(\frac{k_2}{k_1}\right) = \ln\left(\frac{0.020}{0.0050}\right) = \ln(4) = 1.386\)
\(\frac{1}{T_1} - \frac{1}{T_2} = \frac{1}{300} - \frac{1}{320} = 0.003333 - 0.003125 = 0.000208\) K-1
\(E_a = \frac{R \ln(k_2/k_1)}{1/T_1 - 1/T_2} = \frac{8.314 × 1.386}{0.000208} = 55,400\) J/mol ≈ 56 kJ/mol. The other options result from calculation errors in temperature reciprocals or logarithm values.
Q5: For a first-order reaction, what fraction of the reactant remains after 3 half-lives?
(a) 1/3
(b) 1/6
(c) 1/8
(d) 1/9
Ans: (c)
After \(n\) half-lives, the remaining fraction is \(\frac{1}{2^n}\). After 3 half-lives: \(\frac{1}{2^3} = \frac{1}{8}\). Option (a) results from incorrectly dividing by the number of half-lives. Option (b) is not related to any correct formula. Option (d) would be \(\frac{1}{3^2}\), incorrectly using 3 as the base instead of 2.
Q6: A proposed mechanism for a reaction is:
Step 1 (slow): A + B → C
Step 2 (fast): C + A → D
What is the rate law predicted by this mechanism?
(a) rate = k[A][B]
(b) rate = k[A]2[B]
(c) rate = k[C][A]
(d) rate = k[D]
Ans: (a)
The rate-determining step (slow step) is Step 1: A + B → C. Since this is an elementary step, the rate law is rate = k[A][B]. C is an intermediate and doesn't appear in the overall rate law. Option (b) would be correct if Step 2 were slow and we eliminated C. Option (c) incorrectly includes the intermediate C. Option (d) includes the product, which doesn't appear in rate laws.
Quick Review
- Reaction rate is change in concentration per unit time; use stoichiometric coefficients to relate rates of different species
- Rate law (rate = \(k[A]^m[B]^n\)) must be determined experimentally; orders are not stoichiometric coefficients unless reaction is elementary
- Rate constant units reveal reaction order: mol/(L·s) = zero-order, s-1 = first-order, L/(mol·s) = second-order
- First-order half-life is constant: \(t_{1/2} = \frac{0.693}{k}\); zero and second-order half-lives depend on concentration
- Integrated rate laws: zero-order plots [A] vs. t linearly; first-order plots ln[A] vs. t linearly; second-order plots 1/[A] vs. t linearly
- After \(n\) half-lives, fraction remaining is \(\frac{1}{2^n}\) (valid for first-order only as simple formula)
- Arrhenius equation: \(k = Ae^{-E_a/RT}\); higher temperature or lower \(E_a\) increases rate constant
- Two-point Arrhenius form for calculations: \(\ln\left(\frac{k_2}{k_1}\right) = \frac{E_a}{R}\left(\frac{1}{T_1} - \frac{1}{T_2}\right)\); always convert to Kelvin
- Elementary reactions: rate law comes directly from balanced equation; molecularity equals number of molecules reacting in that step
- Rate-determining step (slowest) controls overall rate; intermediates appear in mechanism but not overall equation; catalysts are consumed then regenerated




