Chapter Notes: Exploring Mixtures and their Separation

Introduction
This chapter explores mixtures in greater depth - their properties, behaviour, and the various techniques used to separate them. From industrial processes like sugar production to life-saving medical tests, the separation of mixtures plays a crucial role in our daily lives.
1. How Can We Classify Mixtures?
Homogeneous Mixture (Solution)
A homogeneous mixture has a uniform composition throughout. Every part of the mixture looks and tastes the same.
- Example: Sugar dissolved in water - equally sweet from first sip to last.
- Other examples: Vinegar (acetic acid in water), aerated drinks (carbon dioxide in water).
- A solution is always homogeneous.
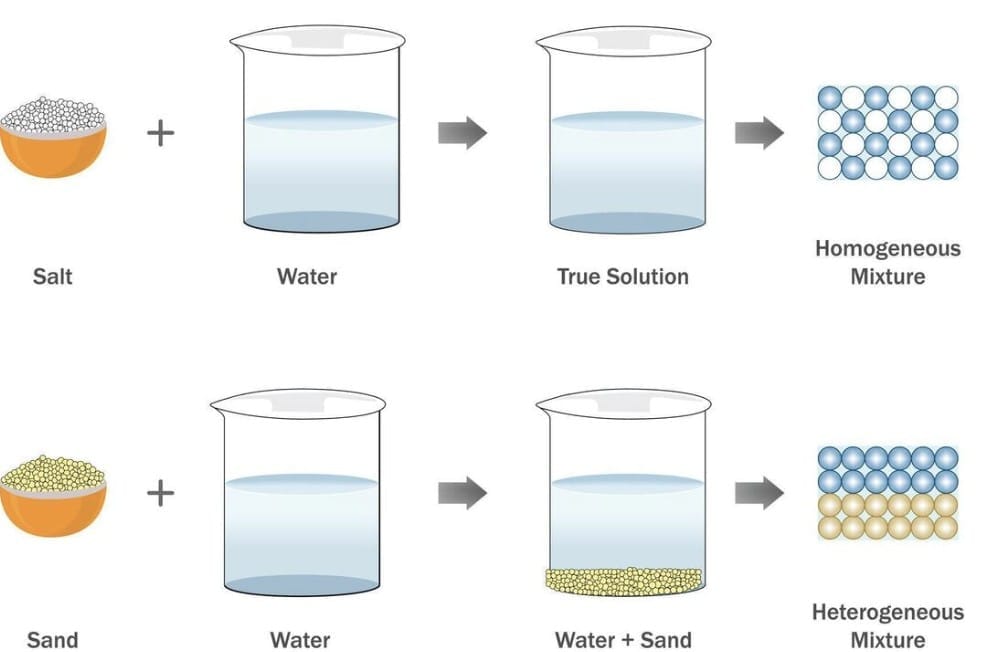
Heterogeneous Mixture
A heterogeneous mixture is non-uniform. Its composition varies from one part to another.
- Example: Sand and water - sand particles are visible and settle with time.
- Other examples: Muddy water, oil and water, chalk powder in water.
2. Solutions
A solution is a homogeneous mixture of two or more substances.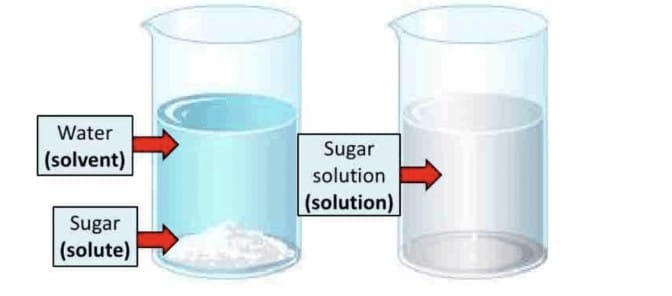
- Solute: The substance that gets dissolved (e.g., sugar).
- Solvent: The substance that dissolves the solute (e.g., water).
2.1 Concentration of a Solution
The amount of solute dissolved in a given amount of solvent or solution is called the concentration of the solution. The right proportion of solute and solvent is always essential when making a solution.
Example: ORS (Oral Rehydration Solution) must have specified amounts of salt and sugar in water. Too little or too much can make it ineffective or harmful.
Note: Not all sugary drinks prepared at home or sold in the market are ORS.
Meet a Scientist
Dilip Mahalanabis
An Indian paediatrician first developed and implemented the treatment for dehydration caused by diseases such as diarrhoea and cholera. He formulated Oral Rehydration Solution (ORS), which revolutionised rehydration therapy. It has saved millions of lives after being popularised worldwide by the World Health Organization (WHO).
2.2 How Do We Express Concentration?
There are three main ways to express concentration in terms of percentage:
A. Mass by Mass Percentage (% m/m or % w/w)
Tells us how many grams of solute are present in 100 grams of the total solution.
Formula: % m/m = (Mass of solute / Mass of solution) x 100
Used for: Milk powder, spice mixtures, packaged food labels.
Example: If 10 g of salt is dissolved in 90 g of water, calculate the mass by mass percentage of the solution formed.
Mass of salt (solute) = \( 10\,\text{g} \), Mass of water (solvent) = \( 90\,\text{g} \), Total mass of solution = \( 10 + 90 = 100\,\text{g} \), Mass by mass percentage = \( \frac{\text{Mass of solute}}{\text{Mass of solution}} \times 100 \) = \( \frac{10}{100} \times 100 = 10\%\,(m/m) \)
B. Mass by Volume Percentage (% m/v or % w/v)
Tells us how many grams of solute are present in 100 mL of the solution.
Formula: % m/v = (Mass of solute / Volume of solution) x 100
Used for: Medicines, laboratories (e.g., 5% glucose IV solution).
Example: If 5 g of glucose is dissolved in water to make 100 mL of solution, calculate its concentration in mass by volume percentage.
If 5 g of glucose is dissolved in water to make 100 mL of solution, calculate its concentration in mass by volume percentage. Mass of glucose (solute) = \( 5\,\text{g} \), Volume of solution = \( 100\,\text{mL} \), Mass by volume percentage = \( \frac{\text{Mass of solute}}{\text{Volume of solution}} \times 100 = \frac{5}{100} \times 100 = 5\%\,(m/v) \)
C. Volume by Volume Percentage (% v/v)
Tells us how many mL of solute are present in 100 mL of the solution.
Formula: % v/v = (Volume of solute / Volume of solution) x 100
Used for: Perfumes, cosmetics, vinegar.
Example: If 1 mL of a liquid pesticide is mixed with a sufficient amount of water to form 100 mL of a pesticide spray for rice crop, calculate its volume by volume percentage.
Volume of pesticide (solute) = \( 1\,\text{mL} \), Total volume of solution = \( 100\,\text{mL} \), Volume by volume percentage = \( \frac{\text{Volume of solute}}{\text{Volume of solution}} \times 100 = \frac{1}{100} \times 100 = 1\%\,(v/v) \)
Note: % m/m and % w/w are numerically equal and used interchangeably.
2.3 Solubility of Substances
Solubility is the maximum amount of solute that can dissolve in 100 mL (or 100 g) of solvent at a given temperature.
- A solution that cannot dissolve any more solute at a given temperature is called a saturated solution.
- Solubility of solid solutes in liquid solvents generally increases with temperature.
- Solubility of gases in liquids generally decreases with increase in temperature.
A solubility curve is a graph showing solubility vs temperature for a substance.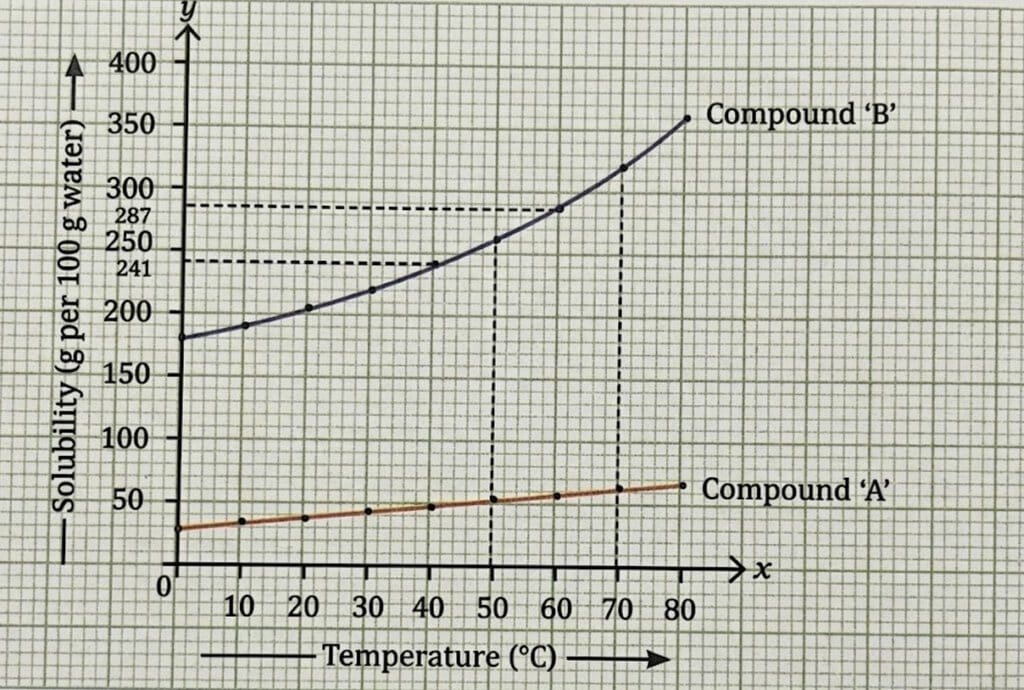
Different substances have different solubilities, and generally, solubility increases with an increase in temperature. From the graph, compound B is more soluble than compound A, and its solubility increases more rapidly with temperature.
When a hot saturated solution is cooled, the excess solute may separate out in the form of crystals.
3. Methods of Separation of Homogeneous Mixtures
3.1 Crystallization
Crystallization is the process of forming crystals from a saturated solution.
- A crystal is a solid made up of particles arranged in a regular geometric pattern.
- Steps involved in the process of crystallization:
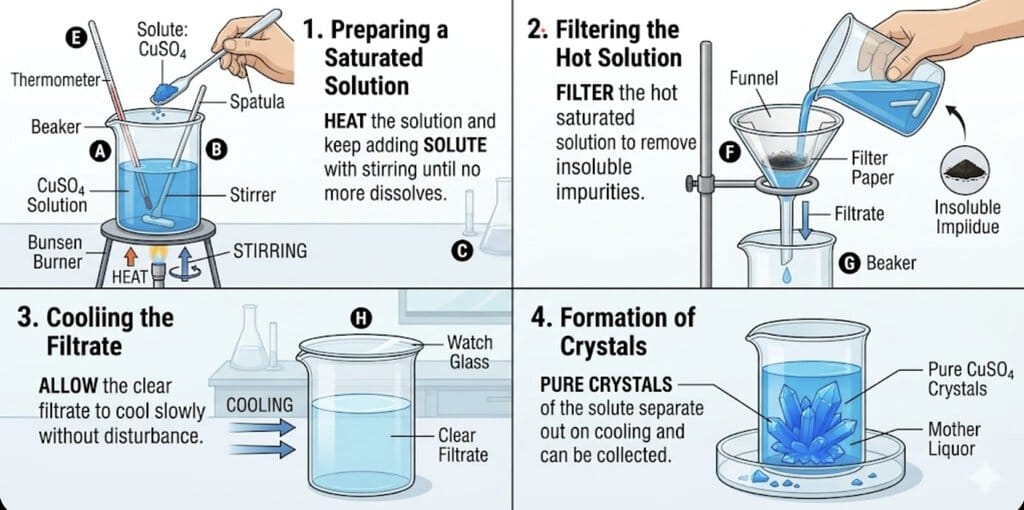 1. Preparing a saturated solution:
1. Preparing a saturated solution:
Heat the solution (e.g., copper sulphate in water) and keep adding solute with stirring until no more dissolves.
2. Filtering the hot solution:
Filter the hot saturated solution to remove insoluble impurities.
3. Cooling the filtrate:
Allow the clear filtrate to cool slowly without disturbance.
4. Formation of crystals:
Pure crystals of the solute separate out on cooling and can be collected. - Principle: Based on differences in solubility of a substance at different temperatures.
- Used for: Separating two solids when one is present in small quantity and both are soluble in the same solvent; also for purification of solids.
- Example: Salt crystals from seawater; copper sulfate (blue vitriol) crystals in lab.
- Large natural crystals are found in mines, caves, and within the Earth's crust, such as in Mawsmai Cave. Minerals like Quartz are common examples of such crystals.
Naturally occurring crystals: Rock salt, candy sugar (mishri), snowflakes, frost on windows, quartz. Fig: Rock Salt
Fig: Rock Salt
Tip: Slow cooling gives larger, well-shaped crystals. Rapid cooling gives smaller, less well-formed crystals.
Note: Sulfuric acid is required for the crystallization of only some salts.
India's Scientific Contributions
Crystallization of salt was an ancient process used by the local communities of the coastal areas in India. The panga salt was obtained by boiling concentrated sea brines,while the evaporation of sea water produced the karkatch salt. Salt crystals of different sizes were produced by these methods.
3.2 Distillation
Distillation is the process of separating a homogeneous mixture of two miscible liquids by heating until the liquid with the lower boiling point vaporises, then cooling the vapour back to liquid (condensation). Fig: Distillation set-up
Fig: Distillation set-up
- It allows recovery of the solvent or separation of liquids that differ in boiling point by at least 25°C.
- Can also be used to separate a liquid from a solution containing dissolved solids.
- Example: Separating acetone (boiling point 56°C) and water (boiling point 100°C).
Historical note: Distillation was used in India for extracting fragrances from flowers to make perfumes. The Deg-Bhapka method used in Kannauj (Uttar Pradesh) for making Mitti ka Ittar is a famous traditional distillation method.
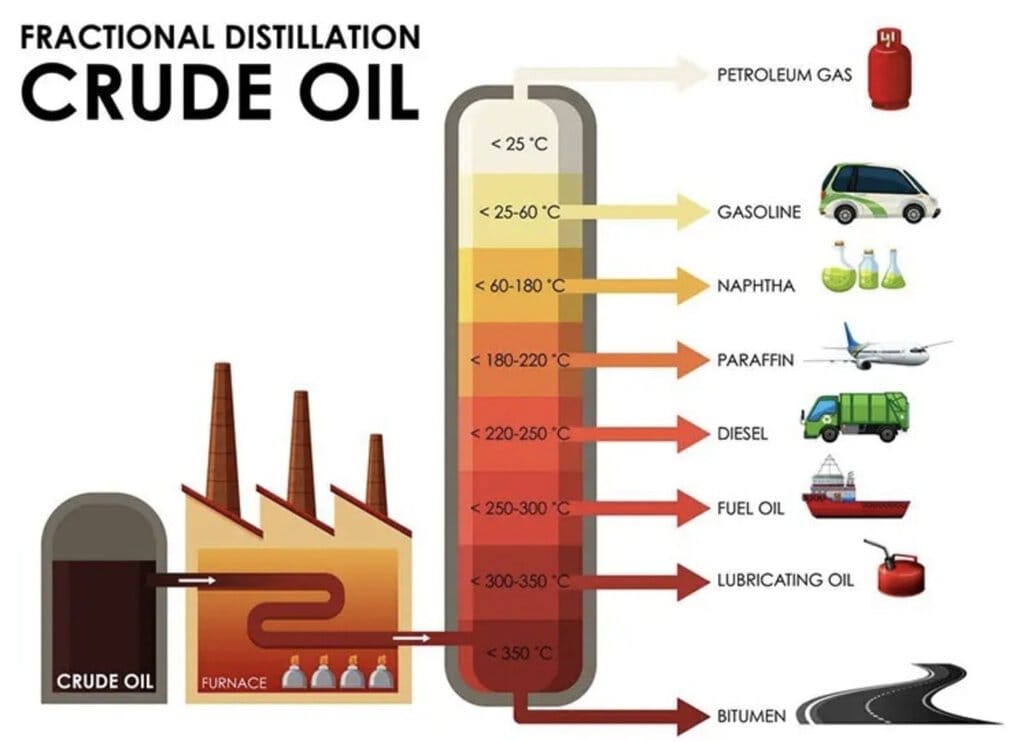
Fractional Distillation: A petroleum refinery is an industrial unit where crude oil extracted from the Earth's crust is processed into useful products like petroleum gas, petrol, kerosene, and diesel. This is done by fractional distillation, which separates components with small differences in boiling points (less than 25 °C). The lighter gaseous fraction is collected first and compressed into Liquefied Petroleum Gas (LPG) for domestic use.
3.3 Paper Chromatography
Paper chromatography is a method of separating the components of a mixture by using differences in their interactions with the solvent and the paper.
- The liquid (solvent) carries substances up the paper, separating them based on how fast they move.
- Used for: Separating coloured substances like dyes, inks, pigments; separating pigments from plant extracts.
 Fig: Paper chromatography
Fig: Paper chromatography - The word 'chromatography' comes from Greek: chroma (colour) + graphein (to write).
How it works: A spot of the mixture is placed on chromatographic paper; the paper is dipped in a solvent. As the solvent rises, it carries different components at different speeds, separating them into distinct spots.
Try yourself: What is crystallization used for?
4. How Can We Separate Components of Heterogeneous Mixtures?
4.1 Separation of Two Immiscible Liquids
Immiscible liquids do not mix and form separate layers (e.g., oil and water, mustard oil and water).
- Tool used: Separating funnel.
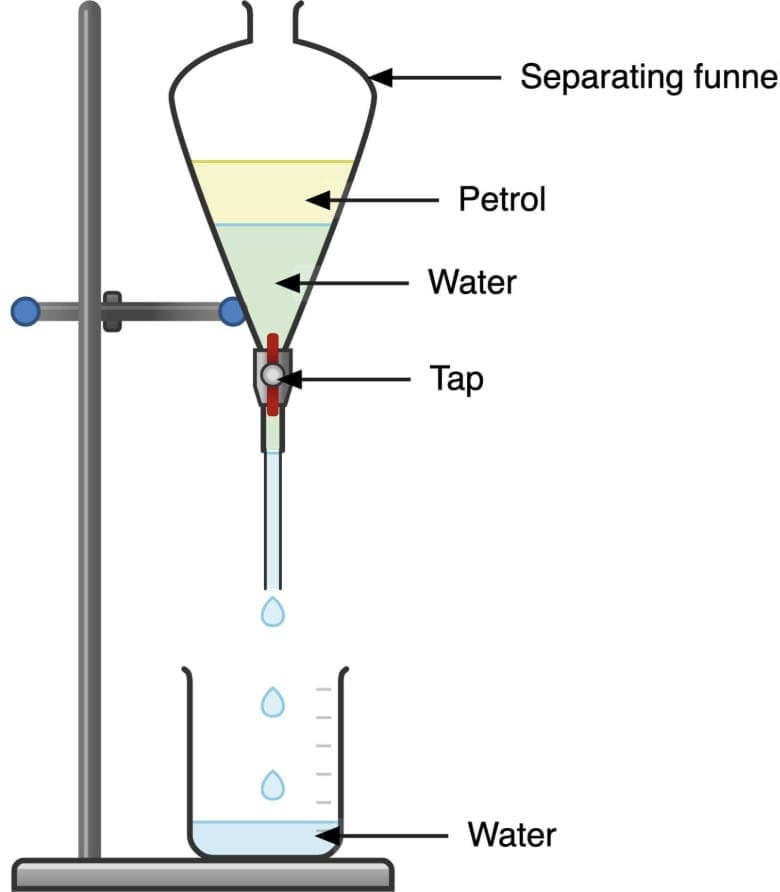 Fig: Separating funnel.
Fig: Separating funnel. - Principle: The denser liquid settles to the bottom; the less dense liquid forms the upper layer.
- Method: Allow layers to form, then open the stopcock to drain the lower layer into a flask; collect the upper layer separately.
- Example: Mustard oil (less dense, yellow, upper layer) separated from water (more dense, lower layer).
Gas mixtures: Most gas-gas mixtures are homogeneous (e.g., hydrogen + oxygen used as rocket fuel). Smoke, fog, and dust in air are heterogeneous mixtures with gas as one component.
4.2 Sublimation
Sublimation is the process in which a solid changes directly into vapour (gas) without passing through the liquid state, on heating below its melting point. On cooling, the vapour changes directly back to solid - this is called deposition.
- Used for: Separating a sublimable solid from a non-sublimable solid.
- Example: Camphor sublimes; sand does not. So camphor can be separated from sand by sublimation.
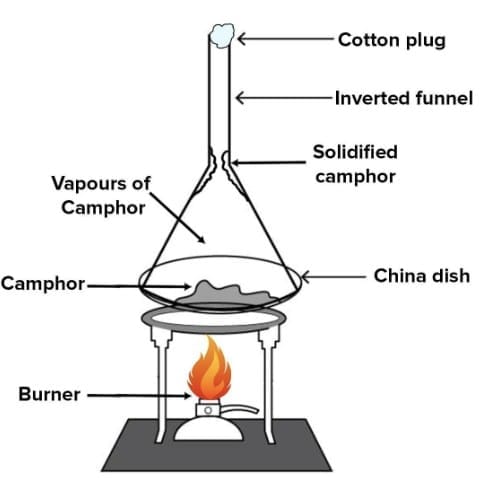 Fig: Sublimation of camphor
Fig: Sublimation of camphor - Other examples: Naphthalene, dry ice (solid CO2) also sublime.
Alloys
An alloy is a homogeneous mixture of two or more metals, or a metal and a non-metal.
- Physical methods cannot separate alloy components.
- Alloys are stronger, more rigid, or more corrosion-resistant than pure metals.

| Alloy | Composition |
|---|---|
| Brass | ~80% copper + ~20% zinc |
| Bronze | ~80% copper + ~20% tin |
| Stainless Steel | Iron + carbon (0.03-0.8%) + chromium (16-18%) + nickel (10-14%) + molybdenum (2-3%) |
4.3 Suspensions
A suspension is a heterogeneous mixture in which solid particles do not dissolve but remain suspended throughout the liquid.
- Particles of a suspension are larger than 1000 nm in diameter.
- Particles are visible to the naked eye.
- Particles settle to the bottom when left undisturbed.
- Can be separated by filtration.
- Examples: Muddy water, sawdust in water, tea leaves in water.
Separating Mud from Water
Centrifugation and/or coagulation are used when filtration alone is insufficient.
A. Centrifugation
Centrifugation is the process of spinning a mixture in a tube at high speed. The centrifugal force causes heavier particles to move outward and settle at the bottom, while the lighter liquid remains at the top.
- Used in: Laboratories to separate blood components (red blood cells, plasma); chemical industries.
- Paperfuge: A hand-powered low-cost device made from cardboard that works like a centrifuge. Useful in remote areas for detecting diseases like malaria and anaemia.
 Fig: Centrifugation machine
Fig: Centrifugation machine
B. Coagulation
Coagulation is the process of adding a substance called a coagulant to make fine suspended particles clump together. These larger clumps settle down by gravity (sedimentation) and can be separated by decantation or filtration. Process of coagulation
Process of coagulation
- Coagulant used: Powdered alum (fitkari) - a white crystalline chemical.
- Example: Adding alum to muddy water to make fine particles clump and settle.
- Another example: Making paneer (cheese) from milk using lemon juice or vinegar as a coagulant.
4.4 Colloids
A colloid is a type of mixture that is neither a true solution nor a true suspension. It appears homogeneous but is actually heterogeneous.
- Particle size: 1 to 1000 nm in diameter.
- Particles do not settle over time.
- Particles are uniformly dispersed throughout the mixture.
- Cannot be separated by filtration.
- Shows the Tyndall Effect.
- Examples: Milk, tomato sauce, ice cream, blood, fog, smoke.
| Property | Solution | Colloid | Suspension |
|---|---|---|---|
| Nature | Homogeneous | Appears homogeneous | Heterogeneous |
| Particle Size | < 1 nm | 1-1000 nm | > 1000 nm |
| Visibility of Particles | Not visible | Not visible | Visible to naked eye |
| Settling | Does not settle | Does not settle | Settles on standing |
| Filtration | Cannot be separated | Cannot be separated | Can be separated |
| Tyndall Effect | No | Yes | Yes |
Components of a Colloid
- Dispersed phase: The solute-like component (the particles that are dispersed).
- Dispersion medium: The component in which the dispersed phase is suspended (like the solvent).
Emulsions
Emulsions are colloids where both the dispersed phase and dispersion medium are liquids.
- Oil-in-water emulsions: Milk, vanishing creams.
- Water-in-oil emulsions: Butter, body lotions, cold cream.
- Emulsifying agents (e.g., proteins in milk) stabilise emulsions.
5. Tyndall Effect
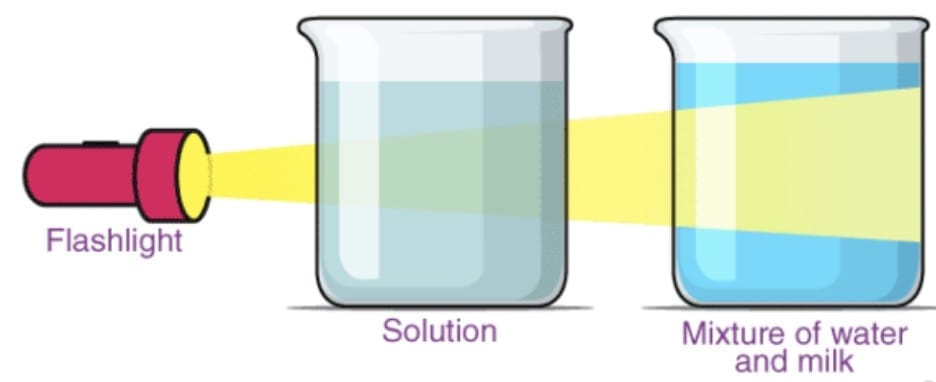
The Tyndall Effect is the scattering of light by particles in a colloid or suspension, making the path of the light beam visible.
Named after: Scientist John Tyndall, who first studied this phenomenon.
Occurs in: Colloids and suspensions (NOT in true solutions).
Examples in daily life:
- A beam of light entering a dark room through a small hole (scattered by dust particles).
- Light from floodlights in a sports stadium.
- Sunlight passing through gaps in leaves of a dense tree.
- Milk (colloid) shows Tyndall effect; a copper sulfate solution (true solution) does not.
Quick Summary: Separation Methods
| Method | Type of Mixture | Principle | Example |
|---|---|---|---|
| Crystallization | Homogeneous (solid-liquid) | Difference in solubility at different temperatures | Purifying copper sulfate; salt from seawater |
| Distillation | Homogeneous (liquid-liquid) | Difference in boiling points (>25°C) | Separating acetone and water |
| Fractional Distillation | Homogeneous (liquid-liquid) | Difference in boiling points (<25°C) | Crude oil refining |
| Paper Chromatography | Homogeneous (mixture of solutes) | Difference in movement rates on paper | Separating ink dyes, plant pigments |
| Separating Funnel | Heterogeneous (two immiscible liquids) | Difference in density | Oil from water |
| Sublimation | Heterogeneous (sublimable + non-sublimable solid) | One solid sublimes, other does not | Camphor from sand |
| Centrifugation | Heterogeneous (solid-liquid suspension) | Centrifugal force separates by density | Blood components |
| Coagulation | Suspension (fine particles in liquid) | Particles clump together and settle | Mud from water using alum |
Key Terms to Remember
| Term | Meaning |
|---|---|
| Homogeneous mixture | Uniform composition throughout (e.g., saltwater) |
| Heterogeneous mixture | Non-uniform composition (e.g., sand and water) |
| Solution | Homogeneous mixture of solute and solvent |
| Solute | Substance that gets dissolved |
| Solvent | Substance that dissolves the solute |
| Concentration | Amount of solute in a given amount of solvent/solution |
| Saturated solution | Cannot dissolve any more solute at a given temperature |
| Solubility | Maximum amount of solute that dissolves in 100g/mL of solvent at a given temperature |
| Solubility curve | Graph of solubility vs temperature |
| Crystal | Solid with particles in a regular geometric pattern |
| Distillation | Separating liquids by vaporisation and condensation |
| Fractional distillation | Distillation for liquids with small differences in boiling points |
| Chromatography | Separation by differences in movement rates on paper |
| Immiscible liquids | Liquids that do not mix (e.g., oil and water) |
| Sublimation | Solid to gas without passing through liquid state |
| Deposition | Gas to solid without passing through liquid state |
| Suspension | Heterogeneous mixture with particles >1000 nm that settle |
| Colloid | Mixture with particles 1-1000 nm; appears homogeneous |
| Emulsion | Colloid with both liquid dispersed phase and dispersion medium |
| Centrifugation | Separation using rapid spinning (centrifugal force) |
| Coagulation | Clumping of fine particles using a coagulant (e.g., alum) |
| Tyndall Effect | Scattering of light by particles in colloid/suspension |
| Dispersed phase | Solute-like component in a colloid |
| Dispersion medium | Solvent-like component in a colloid |
| Alloy | Homogeneous mixture of two or more metals |
FAQs on Chapter Notes: Exploring Mixtures and their Separation
| 1. What are the main classifications of mixtures? |  |
| 2. What is a solution, and how is it formed? |  |
| 3. What are some methods used to separate homogeneous mixtures? |  |
| 4. How can the components of heterogeneous mixtures be separated? |  |
| 5. What is the Tyndall effect, and how does it relate to mixtures? |  |