NCERT Solutions: Exploring Mixtures and their Separation
Page 72 - Think It Over
Q1: Why do suspended particles settle in muddy water over time but not in milk?
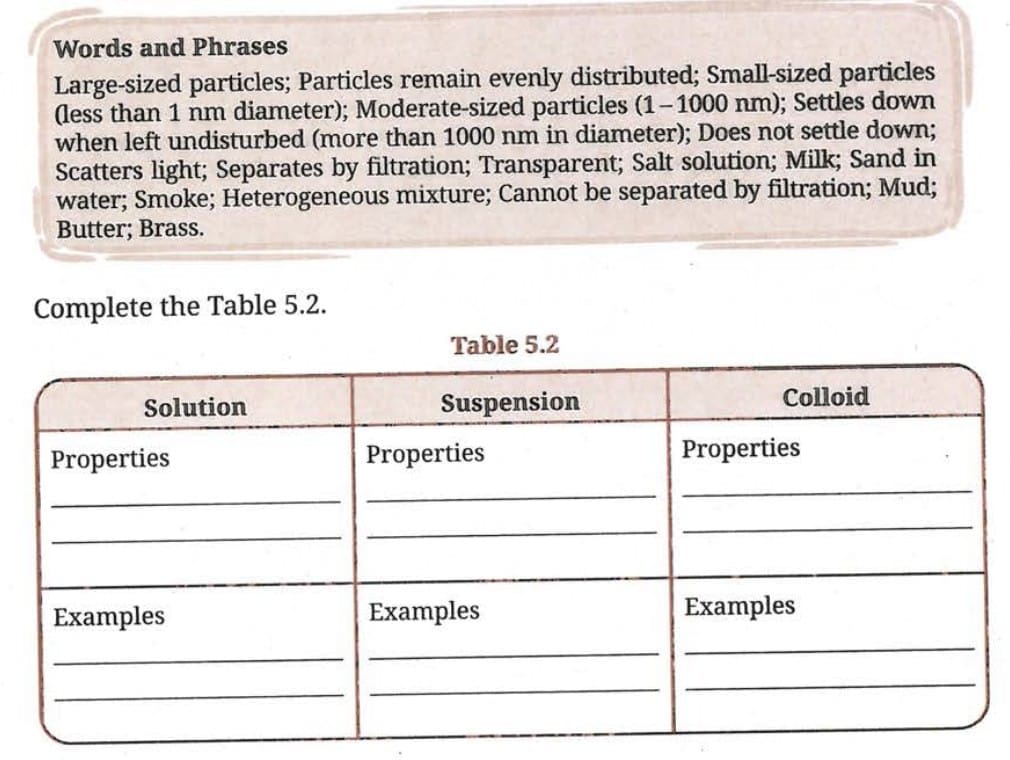
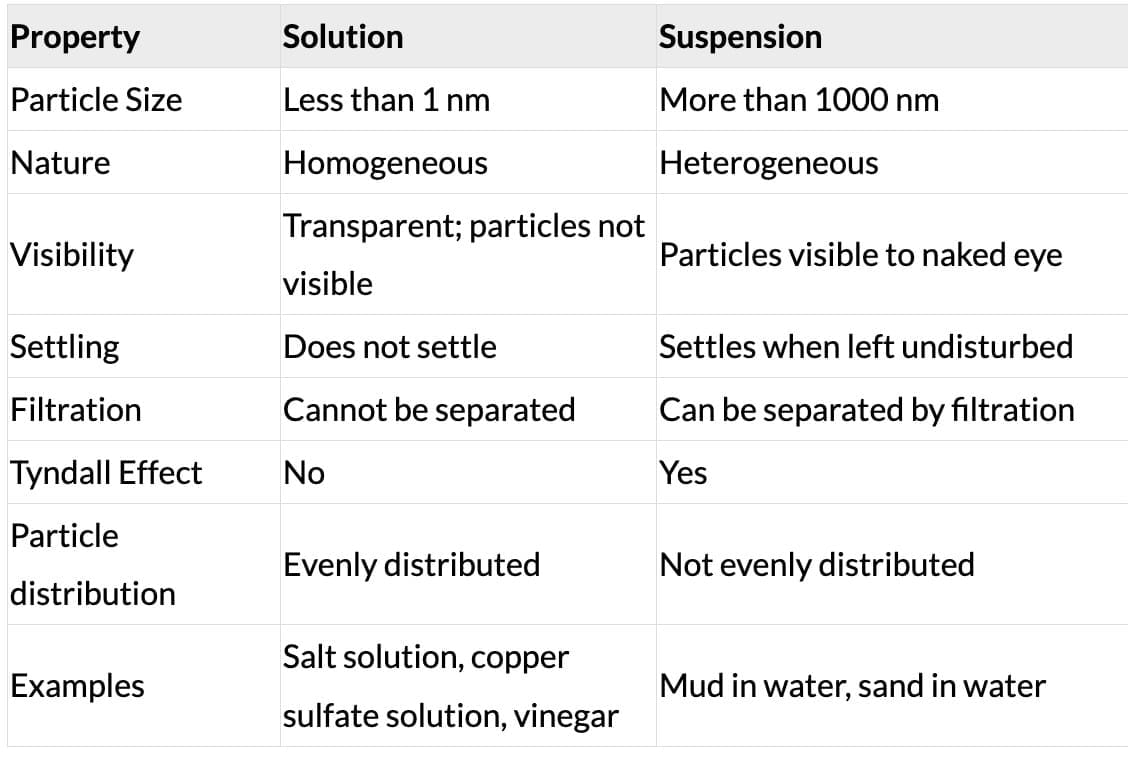
Ans: Suspended particles in muddy water are large and heavy, so they settle down due to gravity when left undisturbed. Milk is a colloid, and its particles are very small and remain uniformly dispersed, so they do not settle.
Q2: How is evaporation different from boiling?
Ans: Evaporation is a slow process that occurs at the surface of a liquid at any temperature, whereas boiling is a rapid process that occurs throughout the liquid at a fixed temperature (boiling point)>
Q3: Why do you see bright rays of sunlight when it passes through small gaps between the leaves of a dense tree?
Ans: This happens due to the scattering of light by tiny particles like dust and water droplets present in air (Tyndall effect), which makes the path of light visible.
Page 75 - Pause and Ponder
Q 1. A common talcum powder contains 4% m/m zinc oxide. How much zinc oxide is present in 300 g of the talcum powder?
Ans: 4% m/m means 4 g of zinc oxide in 100 g of talcum powder. In 300 g: Zinc oxide = (4/100) x 300 = 12 g.
So 12 g of zinc oxide is present in 300 g of talcum powder.
Q 2. Your mother gives you a bottle of orange juice concentrate. She asks you to mix two tablespoons (each 15 mL) with water to make 150 mL of juice per person. What is the % v/v of orange juice concentrate in the mixture?
Ans: Volume of concentrate = 2 x 15 mL = 30 mL.
Total volume of juice = 150 mL. % v/v = (30/150) x 100 = 20% v/v.
Q 3. Vinegar contains 5% v/v acetic acid. Glacial acetic acid is 100% acetic acid. If you want to make vinegar from glacial acetic acid, how would you proceed?
Ans: 5% v/v means 5 mL of glacial acetic acid in every 100 mL of vinegar. To make 100 mL of vinegar: Take 5 mL of glacial acetic acid and slowly add water to make the total volume 100 mL. Mix carefully (always add acid to water, never water to acid).
Page 79 - Pause and Ponder
Q 4. Refer to solubility curves in Activity 5.2. If equal masses of hot, saturated solutions of compounds 'A' and 'B' are cooled from 80°C to 60°C, which solution is likely to deposit more solid?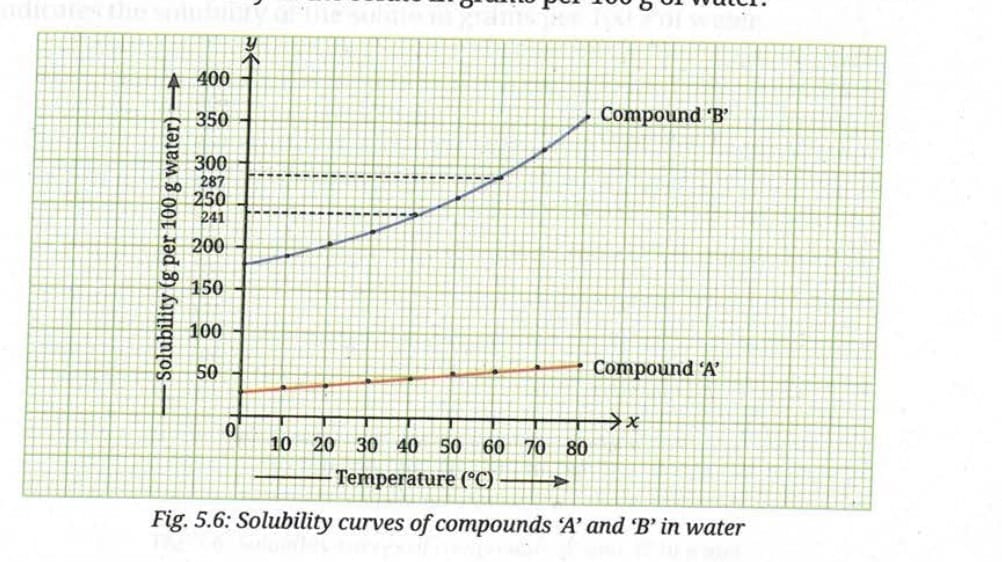
Ans: Compound B will deposit more solid. Looking at the solubility curves, Compound B shows a steeper decrease in solubility when cooled from 80°C to 60°C compared to Compound A. Therefore, more of Compound B will crystallise out.
Q 5. Will there be any change in the size of common salt crystals if the rate of evaporation is increased or decreased? Explain.
Ans: Yes. If evaporation rate is increased (fast evaporation), crystals form quickly and are smaller in size. If evaporation rate is decreased (slow evaporation), crystals grow slowly and are larger, well-shaped crystals. This is because slow cooling/evaporation gives particles enough time to arrange themselves in a proper geometric pattern.
Page 82 - Pause and Ponder
Q 6 (i). Salt can be separated from a salt solution by evaporation or distillation.
Ans: True. Salt can be separated from salt solution by evaporation (the water evaporates, leaving salt behind). Distillation can also be used if we want to recover the water as well.
Q 6 (ii). Distillation can be used for separation of two liquids even when these have the same boiling point.
Ans: False. Distillation works only when the two liquids differ in boiling points (by at least about 25°C). If two liquids have the same boiling point, they cannot be separated by distillation.
Q 6 (iii). In paper chromatography, the solvent level should be above the sample spot at the beginning of the experiment.
Ans: False. In paper chromatography, the solvent level should be below the sample spot. If the solvent level is above the spot, the sample will dissolve directly into the solvent instead of being carried up the paper by capillary action.
Q 6 (iv). Evaporation and crystallization are the same processes.
Ans: False. Evaporation is the process of removing the solvent by heating, leaving the solute behind (but may not give pure crystals). Crystallization is a more precise process that gives pure crystals of the solute by controlled cooling of a saturated solution. Crystallization gives purer and better-formed crystals than simple evaporation.
Page 84 - Pause and Ponder
Q 7. Why do immiscible liquids form two separate layers in a separating funnel?
Ans: Immiscible liquids do not mix with each other because they have different chemical properties (polarity). They also have different densities. The denser liquid sinks to the bottom and the less dense liquid floats on top, forming two distinct, separate layers. For example, water (denser) forms the lower layer and mustard oil (less dense) forms the upper layer.
Q 8. Is sublimation different from evaporation? Justify.
Ans: Yes, sublimation is different from evaporation. Evaporation is the process by which a liquid converts into vapour at the surface, below its boiling point. Sublimation is the process by which a solid converts directly into vapour without passing through the liquid state. So in evaporation, the substance first exists as a liquid (e.g., water evaporating from a wet cloth). In sublimation, the substance goes from solid directly to gas (e.g., camphor, naphthalene balls disappearing without leaving any liquid behind).
Page 85- Threads of Curiosity
Q: The spinning game is a folk dance called phugadi in Marathi and kikli in Punjabi. What is this called in your local language?
Ans: It is commonly called "Ghoomar" or "Chakkar wala khel" in many Hindi-speaking regions. (The name may vary depending on the local language and region.
Page 88 - Pause and Ponder
Q 9. Clouds are made up of tiny water droplets or ice crystals floating in the air. Based on what you know about solutions, suspensions and colloids, what type of mixture do you think clouds are, and why?Ans: Clouds are colloids. The tiny water droplets or ice crystals (dispersed phase) are suspended in air (dispersion medium). The particle size is in the colloid range (1-1000 nm). The particles do not settle, the mixture appears uniform, and it shows the Tyndall Effect (the beam of sunlight is visible through clouds). All these are properties of a colloid.
Q 10. Why do cities with a lot of smoke and dust in the air often look hazy?
Ans: Smoke and dust form colloidal mixtures with air. These fine particles (1-1000 nm) scatter light in all directions - this is the Tyndall Effect. The scattering of light by these particles makes the atmosphere look hazy or foggy. This is why polluted cities appear hazy, especially in sunlight.
Revise, Reflect, Refine
Ans: Option (iv) is correct.
- Muddy water - Ht (heterogeneous; particles visible, settle on standing).
- Milk - Ht (heterogeneous/colloid; appears uniform but is actually heterogeneous at particle level).
- Blood - Ht (heterogeneous/colloid; contains cells suspended in plasma).
- Brass - Hm (homogeneous alloy of copper and zinc with uniform composition throughout).
(a) air and dust particles
(b) copper sulfate and water
(c) starch and water
(d) acetone and water
- (a) Air and dust particles: This is a colloid - dust particles (1-1000 nm) dispersed in air. Colloids show the Tyndall Effect. CORRECT.
- (b) Copper sulfate and water: This is a true solution (particles < 1 nm). True solutions do NOT show the Tyndall Effect. INCORRECT.
- (c) Starch and water: Starch forms a colloid in water (starch particles are in the colloid range). Colloids show the Tyndall Effect. CORRECT.
- (d) Acetone and water: This is a homogeneous solution. Solutions do NOT show the Tyndall Effect. INCORRECT.
(i)A cake recipe uses dry ingredients, namely75 g of sugar for 420 g of all-purpose flour and 5 g of sodium hydrogencarbonate. Express the concentration of each component in the mixture using an appropriate method.
Ans: Total mass of mixture = 75 + 420 + 5 = 500 g. Since all components are solids, we use mass by mass percentage (% m/m).
- Sugar: % m/m = (75/500) x 100 = 15% m/m
- All-purpose flour: % m/m = (420/500) x 100 = 84% m/m
- Sodium hydrogencarbonate: % m/m = (5/500) x 100 = 1% m/m
(ii). A brass alloy contains 70% copper by mass. Calculate the quantities of copper and zinc present in 120 g of brass.
Ans: Brass contains 70% copper and (100 - 70) = 30% zinc by mass.
- Mass of copper = (70/100) x 120 = 84 g
- Mass of zinc = (30/100) x 120 = 36 g
- Verification: 84 + 36 = 120 g
Ans: Yes, cooking oil and water are immiscible liquids - they do not mix and will form two separate layers. Oil is less dense than water, so oil will form the upper layer. Water (denser) will form the lower layer.
Method of separation: Using a separating funnel.
Procedure: Pour the mixture into the separating funnel and allow it to stand until two distinct layers form. Open the stopcock slowly to drain the lower layer (water) into a beaker. Close the stopcock when water is almost fully drained. Then open the stopcock again to collect the upper oil layer in a separate container.
Apparatus: Separating funnel fixed on a laboratory stand with a stopcock at the bottom and a conical flask to collect each layer.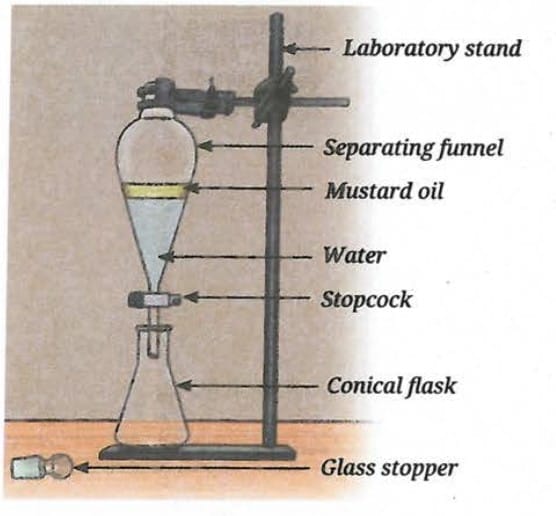
(i) Both A and R are true, and R is the correct explanation of A.
(ii) Both A and R are true, but R is not the correct explanation of A.
(iii) A is true, but R is false.
(iv) A is false, but R is true.
Ans: Option (iii): A is true, but R is false.
Explanation: The assertion is true - solutions do not show the Tyndall Effect. However, the reason given is false. The particles in a solution are SMALLER than 1 nm (not larger than 100 nm). It is because the particles are so small (less than 1 nm) that they cannot scatter light effectively, so solutions do not show the Tyndall Effect.
Q 7. How would you separate the mixtures given in Table 5.3? Mention the reason for choosing your method.
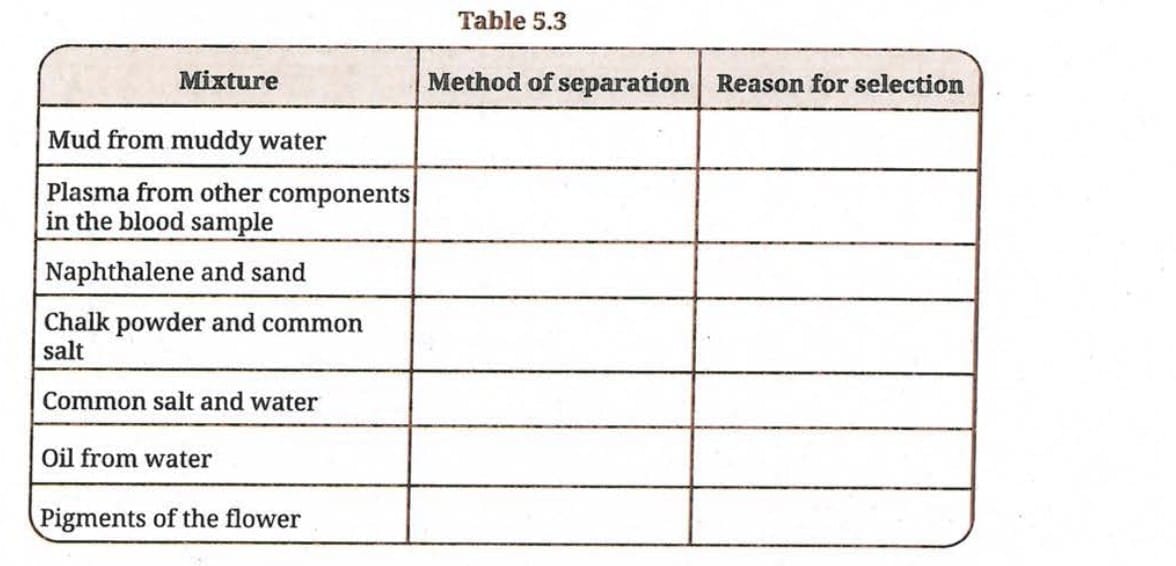
Ans:
Ans: The boiling points of A (60°C) and B (90°C) differ by 30°C (which is more than 25°C), so simple Distillation can be used to separate them.
Process: Heat the mixture in a distillation flask. Liquid A, having the lower boiling point (60°C), vaporises first. The vapour of A passes through the condenser, where it cools and condenses back into liquid and is collected in a conical flask as the distillate. Liquid B, with the higher boiling point (90°C), remains in the distillation flask.
 Q 9. Compare evaporation, crystallization and distillation. In which situation would you prefer each of these over the others?
Q 9. Compare evaporation, crystallization and distillation. In which situation would you prefer each of these over the others?
Ans: Evaporation: Removes solvent by heating; solute is left behind as a solid. Prefer when we want to obtain the solute and do not need the solvent. Example: Getting salt from saltwater, concentrating a solution. Not suitable when solute decomposes on heating.
Crystallization: Controlled cooling of a saturated solution to obtain pure crystals of the solute. Prefer when we want pure crystals of the solute, or when two solids are both dissolved in the same solvent and need to be separated. Best when solute's solubility changes significantly with temperature. Example: Purifying copper sulfate, separating potassium nitrate from sodium chloride.
Distillation: Separates two miscible liquids with different boiling points (>25°C), or separates solvent from solute while recovering both. Prefer when we need to separate two miscible liquids, OR when we want to recover both the solute (in the flask) and the solvent (as distillate). Example: Separating acetone and water; purifying water.
Q 10 (i). What would happen if blood behaved like a true suspension inside the body?
Ans: If blood behaved like a true suspension, its particles (red blood cells, white blood cells, platelets) would settle down to the bottom due to gravity, just like mud particles settle in muddy water. This would make blood unable to flow properly through the blood vessels. The cells would not be distributed throughout the bloodstream, which would prevent the delivery of oxygen and nutrients to body tissues. This would be fatal - the body's organs would fail due to lack of oxygen and nutrients.
Q 10 (ii). In a blood sample, identify the dispersed phase and the dispersion medium.
Ans:
- Dispersed phase: Blood cells - red blood cells (RBCs), white blood cells (WBCs), and platelets.
- Dispersion medium: Blood plasma (the yellowish liquid that makes up most of the blood).

Ans: The correct sequence of separation techniques is:
- Step 1 - Sublimation: Heat the mixture gently. Naphthalene sublimes (converts directly to vapour) and is collected on the cooler funnel, leaving behind sand and salt.
- Step 2 - Dissolve in water and Filter: Add water to the remaining mixture of sand and salt. Salt dissolves in water (filtrate), while sand does not dissolve and is collected on the filter paper as residue.
- Step 3 - Evaporation/Distillation: Evaporate the filtrate (salt solution) to recover common salt. All three components - naphthalene, sand, and common salt - are now separated.
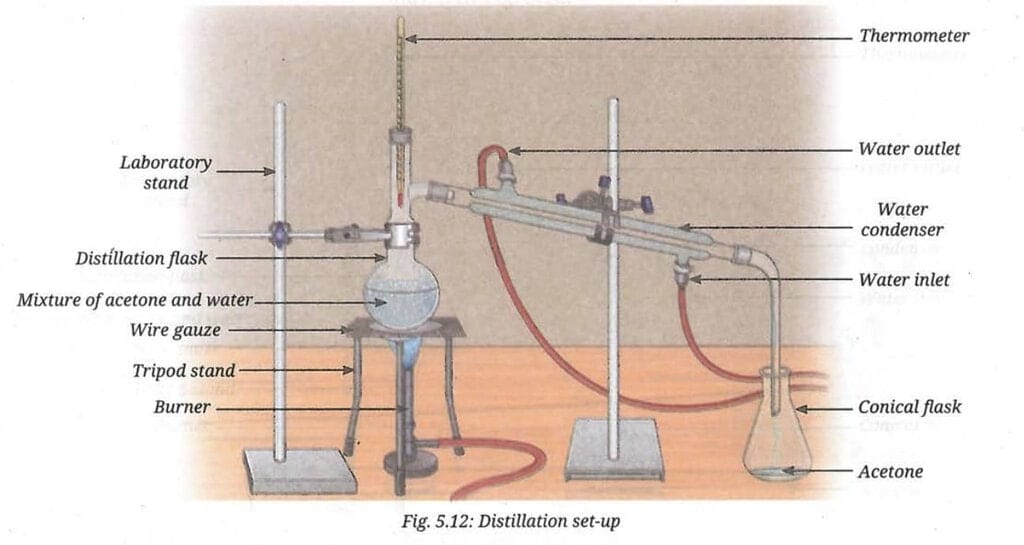
Ans: Distillation is effective for separating water and acetone because:
(i) Water and acetone are miscible liquids (they mix completely), so simple filtration or a separating funnel cannot separate them.
(ii) They have significantly different boiling points - acetone boils at 56°C and water boils at 100°C. The difference is 44°C, which is well above the required minimum of 25°C for distillation to work effectively.
(iii) When the mixture is heated, acetone vaporises first at 56°C, the vapour is condensed and collected as pure acetone. Water remains behind in the distillation flask.
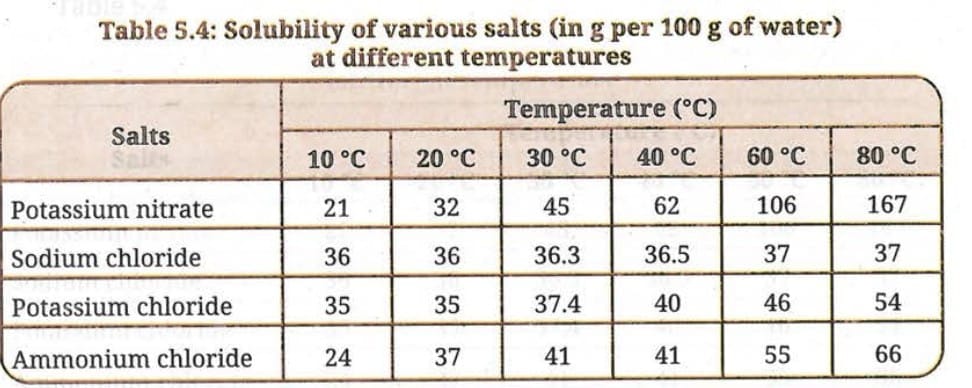
Q 13 (i). What mass of potassium nitrate would be needed to prepare its saturated solution in 50 g of water at 40°C?
Ans: From the table, the solubility of potassium nitrate at 40°C = 62 g per 100 g of water. For 50 g of water: Mass of potassium nitrate = (62/100) x 50 = 31 g. So 31 g of potassium nitrate is needed to make a saturated solution in 50 g of water at 40°C.
Q 13 (ii). A student makes a saturated solution of potassium chloride in water at 80°C and leaves the solution to cool at room temperature (25°C). What would she observe as the solution cools? Explain.
Ans: At 80°C, the solubility of potassium chloride is 54 g per 100 g water. At 25°C, solubility is approximately 35 g per 100 g water (interpolating from the table between 20°C and 30°C values).
As the solution cools from 80°C to 25°C, the solubility drops from 54 g to about 35 g. The extra potassium chloride (54 - 35 = 19 g per 100 g water) that can no longer remain dissolved will crystallise out. The student would observe white crystals of potassium chloride forming and settling at the bottom of the beaker.
Q 13 (iii). What is the effect of a change in temperature on the solubility of salts? Also, compare the changes in the solubility of the four given salts with increasing temperature from 10°C to 80°C.
Ans: Effect of temperature: The solubility of all four salts increases with an increase in temperature (for solid solutes in liquid solvents).
Comparison:
- Potassium nitrate: Most dramatic increase - from 21 g at 10°C to 167 g at 80°C. Solubility increases very rapidly with temperature.
- Sodium chloride: Least change - from 36 g at 10°C to 37 g at 80°C. Solubility is nearly constant, barely affected by temperature.
- Potassium chloride: Moderate increase - from 35 g at 10°C to 54 g at 80°C. Steady increase with temperature.
- Ammonium chloride: Significant increase - from 24 g at 10°C to 66 g at 80°C. Considerable increase with temperature.
Order of increase in solubility with temperature (least to most): Sodium chloride < Potassium chloride < Ammonium chloride < Potassium nitrate.
Student A dissolves 20 g of sugar in 80 g of water. Student B dissolves 20 g of sugar in 100 g of water. - Student C dissolves 30 g of sugar in 80 g of water.
Q 14 (i). Calculate the mass percentage (% m/m) concentration of sugar in each student's solution.
Ans:
- Student A: Total mass = 20 + 80 = 100 g. % m/m = (20/100) x 100 = 20% m/m
- Student B: Total mass = 20 + 100 = 120 g. % m/m = (20/120) x 100 = 16.67% m/m
- Student C: Total mass = 30 + 80 = 110 g. % m/m = (30/110) x 100 = 27.27% m/m
Q 14 (ii). Whose solution is the most concentrated? Explain why.
Ans: Student C's solution is the most concentrated (27.27% m/m). This is because Student C dissolved the largest amount of sugar (30 g) in the smallest amount of solvent (80 g water), giving the highest mass percentage. Concentration depends on both the amount of solute and the total mass of the solution - more solute in less solvent means higher concentration.
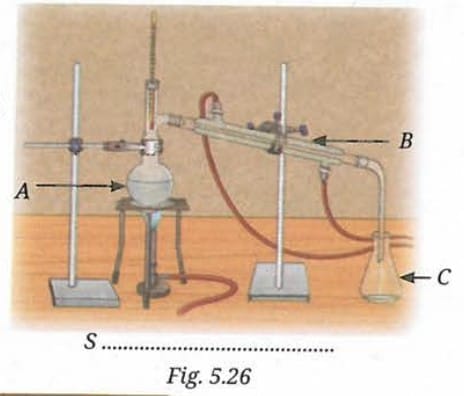
Q 15 (i). Identify the separation technique marked as 'S'.
Ans: The separation technique marked as 'S' is Distillation. This is indicated by the distillation flask, thermometer, condenser, and collection flask visible in the figure.
Q 15 (ii). Label the apparatus A, B and C.
Ans:
- A = Distillation flask (where the mixture is heated).
- B = Water condenser (where vapour is cooled back to liquid).
- C = Conical flask (where the distillate - the purified liquid - is collected).
Q 15 (iii). Which of the following mixtures can be separated by the technique identified above? Use the data given in Table 5.5.
Mixtures:
(a) water-acetone
(b) water-salt
(c) acetone-alcohol
(d) sand-salt
(e) alcohol-chloroform
(f) alcohol-benzene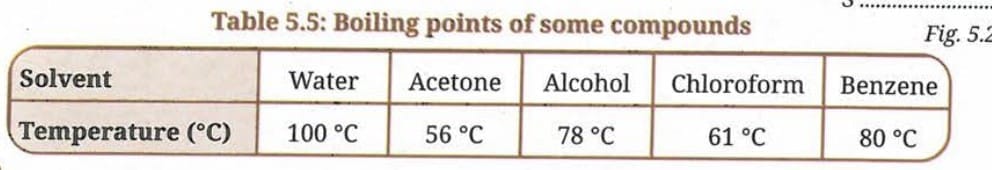
Ans: Distillation works when liquids are miscible and differ in boiling points by at least about 25°C.
- (a) Water (100°C) and acetone (56°C): Difference = 44°C > 25°C. CAN be separated by distillation.
- (b) Water and salt: Salt is a solid. Water can be distilled away from salt solution. CAN be separated.
- (c) Acetone (56°C) and alcohol (78°C): Difference = 22°C < 25°C. CANNOT be separated by simple distillation.
- (d) Sand and salt: Both are solids. Distillation is NOT applicable.
- (e) Alcohol (78°C) and chloroform (61°C): Difference = 17°C < 25°C. CANNOT be separated by simple distillation.
- (f) Alcohol (78°C) and benzene (80°C): Difference = 2°C, far less than 25°C. CANNOT be separated by simple distillation.
Mixtures that CAN be separated by distillation: (a) water-acetone and (b) water-salt.
FAQs on NCERT Solutions: Exploring Mixtures and their Separation
| 1. What are mixtures and how are they classified? |  |
| 2. What are some common methods for separating mixtures? |  |
| 3. How does filtration work in the separation of mixtures? |  |
| 4. What is the principle behind distillation? |  |
| 5. Why is the separation of mixtures important in daily life? |  |






