Chapter Notes: Atomic Foundations of Matter

Introduction
This chapter builds on the concepts from Chapter 8 - Journey Inside the Atom, where subatomic particles (electrons, protons, neutrons) and atomic structure were studied. It was established that atoms with an octet of electrons in their valence shell are stable, and atoms can lose, gain or share electrons to achieve this stability.
- Many properties of elements are not retained when they form a compound
- Example: Hydrogen (combustible gas) + Oxygen (supports combustion) → Water (liquid that extinguishes fire)
- Water has completely different properties from both hydrogen and oxygen
- However, the mass of water formed = sum of masses of hydrogen and oxygen that combined
This chapter goes on to explore the following topics:
- Whether mass remains unchanged during physical and chemical changes
- Laws of Chemical Combination
- Dalton's Atomic Theory
- How atoms combine and types of chemical bonds
- How to write chemical formulae
9.1 Law of Conservation of Mass
The total mass remains the same before and after a chemical reaction. So, matter can neither be created nor destroyed in a chemical reaction. This is known as the Law of Conservation of Mass, proposed by Antoine Lavoisier in 1789.
1. Physical Change
In a physical change, no new substance is formed and the mass remains unchanged.
Example: When salt dissolves in water, the mass of the solution is equal to the sum of the masses of salt and water. This shows that dissolving does not change total mass.
2. Chemical Change
In a chemical change, new substances are formed, but the total mass still remains constant if the system is closed.
Example: When baking soda reacts with vinegar, carbon dioxide gas is produced.
Vinegar + Baking soda (Sodium hydrogencarbonate) Carbon dioxide + Other substances
- In an open system, the gas escapes, so the measured mass appears to decrease.
- In a closed system (like with a balloon), the gas is trapped, and the total mass before and after the reaction remains the same.
Meet a Scientist - Antoine LavoisierAntoine Lavoisier is known as the Father of Modern Chemistry. He proposed the Law of Conservation of Mass. This law applies to every chemical reaction. Lavoisier continued to study this and proposed that "...in every operation an equal quantity of matter exists both before and after the operation."
Verification of Law of Conservation of Mass
The law can be verified by a chemical reaction between sodium sulfate and barium chloride.
When these two solutions are mixed, a white precipitate of barium sulfate is formed along with sodium chloride:
Na₂SO₄ + BaCl₂ → BaSO₄ (precipitate) + 2NaCl
The total mass of both solutions before mixing is equal to the total mass of products after mixing.
Since no gas is formed in this reaction, it can be carried out in an open system and still shows that mass remains constant.
This confirms that in chemical reactions, total mass of reactants = total mass of products, verifying the law of conservation of mass.
Example: In a group activity, students place 4.0 g of calcium carbonate with 2.92 g of hydrochloric acid in a closed container. After the reaction is over, they measured 1.76 g of carbon dioxide, 0.72 g of water, and 4.44 g of calcium chloride. Verify whether the Law of Conservation of Mass is obeyed or not.
Solution:Mass of calcium carbonate = 4.0 g Mass of hydrochloric acid = 2.92 g Total mass of reactants = 4.0 g + 2.92 g = 6.92 g
Mass of carbon dioxide = 1.76 g Mass of water = 0.72 g Mass of calcium chloride = 4.44 g Total mass of products = 1.76 g + 0.72 g + 4.44 g = 6.92 g
Mass of reactants = Mass of products. Hence, the Law of Conservation of Mass is obeyed.
Example: 12 g of carbon combines with 32 g of oxygen to form 44 g of carbon dioxide as per the given equation: Carbon + Oxygen → Carbon dioxide
If 2.4 g of carbon reacts completely with oxygen, how much carbon dioxide will be produced?
Solution:Given that 12 g of carbon reacts with 32 g of oxygen to give 44 g of carbon dioxide.
So, 1 g of carbon will give = 44/12 g of carbon dioxide.
Thus, 2.4 g of carbon will give = (44/12) × 2.4 g = 8.8 g of carbon dioxide.
Hence, 8.8 g of carbon dioxide will be produced.
9.2 Law of Constant Proportions
Joseph Louis Proust proposed that in any chemical compound, the elements are always present in a fixed ratio by mass, irrespective of the source or method of preparation.
Water always contains hydrogen and oxygen in the mass ratio 1 : 8.
Thus, 9 g of water will always give 1 g hydrogen and 8 g oxygenon decomposition.
The composition of a compound remains constant. This law is also known as Proust's Law.
Meet a Scientist - Joseph Louis Proust
Joseph Louis Proust showed that chemical compounds always contain elements in fixed ratios by mass, leading to the Law of Definite Proportions. For example, he found that copper carbonate always has copper, carbon, and oxygen in the same proportion, regardless of its source. His work helped form the foundation of modern chemistry.
Threads of Curiosity
In ancient times, a red pigment called cinnabar (hingula) was widely used. It was found that heating cinnabar produces mercury and sulfur in fixed mass ratios (about 86.22% mercury and 13.78% sulfur). Similarly, combining mercury and sulfur in the same ratio forms cinnabar, showing that compounds are formed in fixed proportions by mass.
Example: Sodium chloride (NaCl) contains sodium and chlorine in the mass ratio of 23:35.5. If 46 g of sodium reacts completely, how much chlorine is needed to form NaCl?
Solution:Mass of chlorine required = (35.5 ÷ 23) × 46 = 71 g
9.3 Dalton's Atomic Theory
The two laws discussed (Law of Conservation of Mass and Law of Constant Proportions) formed the basis of Dalton's Atomic Theory. This theory attempts to logically explain why substances combine in fixed proportions and why there is no loss or gain of mass during a chemical reaction. Dalton later explained these by proposing that during a chemical reaction, atoms are indivisible and merely rearrange, rather than being created or destroyed.
Meet a Scientist - John Dalton
John Dalton was born in England. In 1793, Dalton moved to Manchester to teach mathematics, physics and chemistry at a college. He spent most of his life teaching and researching there. In 1808, he presented his atomic theory, which proved to be a turning point in the study of matter.
Postulates of Dalton's Atomic Theory
John Dalton postulated that:
- All matter is made up of very tiny particles called atoms, which participate in chemical reactions.
- Atoms are indivisible particles, which cannot be created or destroyed in a chemical reaction.
- Atoms of a given element are identical in mass and chemical properties.
- Atoms of different elements have different masses and chemical properties.
- Atoms combine in the ratio of simple whole numbers to form compounds.
- The relative number and kinds of atoms are constant in a given compound.
Dalton's postulates provide the basis for the modern understanding of atoms and their behaviour.
When elements combine to form compounds, their atoms are not destroyed but simply rearranged. For example, hydrogen and oxygen combine to form water without changing the atoms themselves. Similarly, when magnesium burns in air, it forms magnesium oxide, showing that magnesium atoms combine with oxygen atoms to form a new substance.
9.4 How Atoms Combine?
Atoms of the same element can combine to form molecules, such as a hydrogen molecule made of two hydrogen atoms. Atoms of different elements combine to form molecules of compounds, like hydrogen chloride formed from hydrogen and chlorine atoms.
A molecule is an electrically neutral group of atoms that can exist independently and shows the properties of the substance. Some elements, like helium, exist as single atoms because they are already stable.
Atoms are stable when their outermost shell is complete (8 electrons, or 2 for the K-shell). If not, they become stable by gaining, losing, or sharing electrons. In doing so, atoms combine to form compounds, mainly in two ways:
- Sharing of electrons - Share a few or all of their valence electrons with another atom.
- Transfer of electrons - Transfer one or more of the valence electrons to another atom, or accept one or more electrons from some other atom.
When atoms combine, the total energy of the system becomes lower than the sum of the energies of the individual atoms, making the resulting arrangement more stable. The force that holds atoms together is called a chemical bond.
9.4.1 Bonding by Sharing of Electrons - Covalent Bond
A. Molecules of Elements
Formation of Hydrogen Molecule (H₂):
- Hydrogen has 1 electron in its K-shell.
- It needs 1 more electron to become stable (duplet).
- Two hydrogen atoms share one electron each.
- The shared pair forms a covalent bond.
- This is a single bond, represented as H-H (H₂).
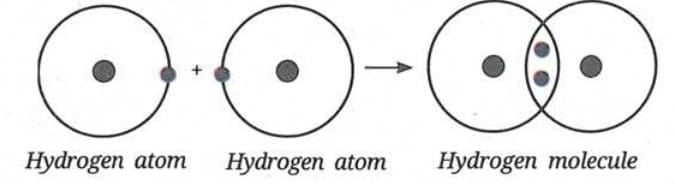
Formation of Chlorine Molecule (Cl₂):
- Chlorine atom has 7 electrons in its valence shell.
- Each chlorine atom needs 1 electron to become stable.
- Two chlorine atoms share one electron each.
- The shared pair forms a single covalent bond.
- Chlorine molecule is represented as Cl-Cl (Cl₂).
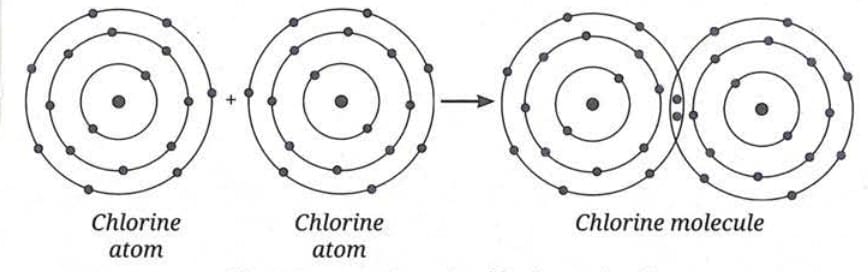
Formation of Oxygen Molecule (O₂):
- Oxygen has 6 electrons in its valence shell.
- It needs 2 more electrons to complete its octet.
- Two oxygen atoms share 2 electrons each.
- This forms a double covalent bond.
- Oxygen molecule is represented as O=O (O₂).
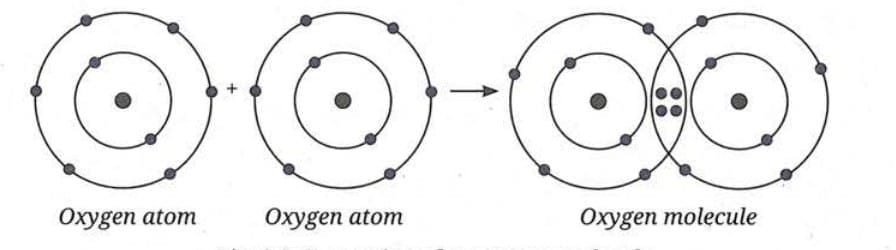
B. Molecules of Compounds
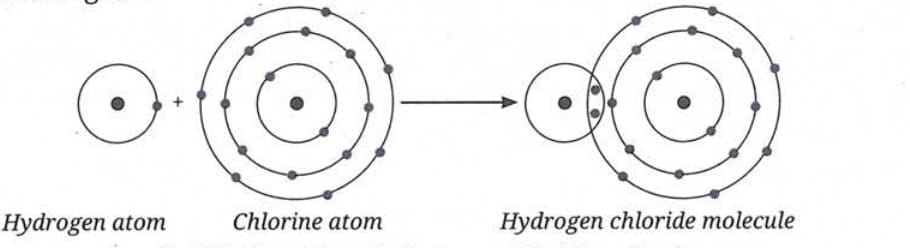
Formation of Hydrogen Chloride Molecule (HCl):
- Electronic configuration: Hydrogen (1) → 1; Chlorine (17) → 2, 8, 7.
- Chlorine needs 1 electron to complete its octet; hydrogen needs 1 electron to complete its duplet.
- Both share one electron each to form hydrogen chloride (HCl).
- This sharing forms a covalent bond.
- HCl has a single bond, represented as H-Cl.
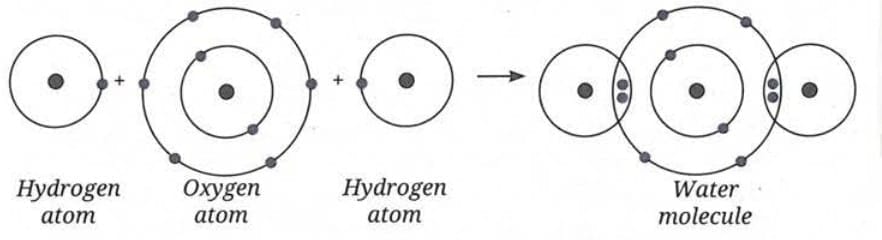
 Formation of Water Molecule (H₂O):
Formation of Water Molecule (H₂O):
- Hydrogen needs 1 electron; oxygen needs 2 electrons to become stable.
- Oxygen shares its two electrons with two hydrogen atoms.
- Each hydrogen shares one electron with oxygen.
- This forms a water molecule.
- Represented as H₂O (2 hydrogen + 1 oxygen).
Bridging Science and Society
Atoms can release large amounts of energy when their nuclei split or combine-this is called nuclear energy. It is used in electricity generation, medicine, research, and space. In nuclear power plants, this energy produces steam to run turbines. In India, Raja Ramanna played an important role in developing nuclear energy for peaceful uses.
C. Naming Covalent Compounds
- Covalent compounds are named using prefixes to show the number of atoms.
- The first element keeps its name; the second element ends with "-ide."
- Common prefixes: mono (1), di (2), tri (3), tetra (4), penta (5), hexa (6), etc.
- "Mono" is usually not used for the first element, but is used for the second.
- If a prefix ends in "o" or "a" and the next word starts with a vowel, drop the last vowel (e.g., monoxide, pentoxide).
- If the prefix ends in "i", it is kept (e.g., dioxide, trioxide).
Examples:
- CO is named as carbon monoxide (not monooxide).
- CO₂ is named as carbon dioxide (not monocarbon dioxide).
- CS₂ is named as carbon disulfide, showing two sulfur atoms.
- PCl₃ is named as phosphorus trichloride, showing three chlorine atoms.
- SF₆ is named as sulfur hexafluoride, showing six fluorine atoms.
- N₂O₄ is named as dinitrogen tetroxide (not tetraoxide).
- N₂O₅ is named as dinitrogen pentoxide.
When hydrogen is the first element in a compound, no prefix is used before it, regardless of the number of hydrogen atoms. For example, H₂S is named hydrogen sulfide, not dihydrogen sulfide. Some compounds are also commonly known by their traditional names rather than systematic names; for instance, H₂O is called water instead of hydrogen monoxide, and NH₃ is called ammonia instead of nitrogen trihydride.
9.4.2 Bonding by Electron Transfer - Ionic Bond
Atoms always try to achieve a stable electronic configuration. The number of valence electrons determines how they do this:
- Atoms with less than 4 valence electrons → tend to donate electrons
- Atoms with more than 4 valence electrons → tend to gain or share electrons
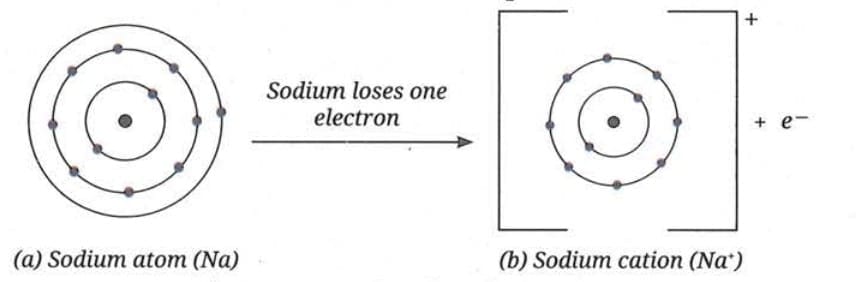
Formation of Sodium Chloride (NaCl):
Sodium has an atomic number of 11, meaning its valence shell contains only 1 electron. Since this is less than 4, sodium achieves stability by losing this electron.
- After losing the electron, sodium becomes a positively charged ion called a cation
- Represented as Na⁺
- Na⁺ contains → 11 protons and 10 electrons
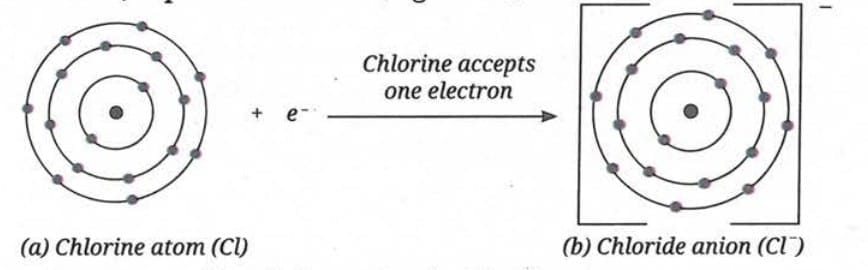
Chlorine has an atomic number of 17, meaning its valence shell contains 7 electrons. Since this is more than 4, chlorine achieves stability by gaining one electron from another atom.
- After gaining one electron, it acquires a negative charge and becomes an anion
- Represented as Cl⁻
- Cl⁻ contains → 17 protons and 18 electrons
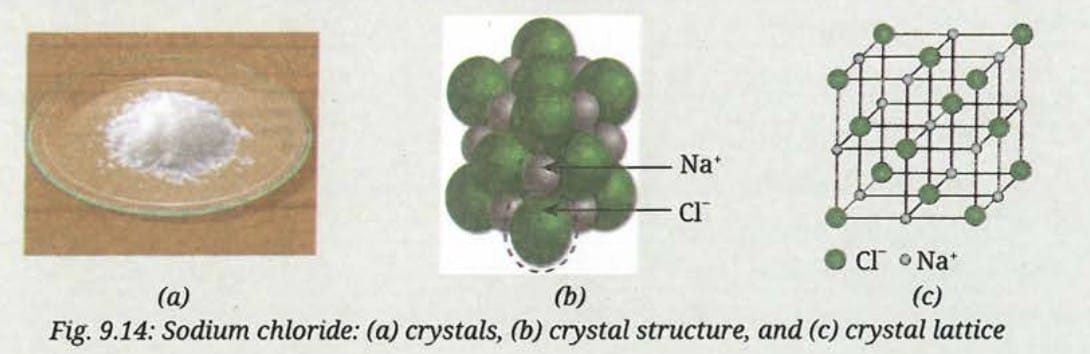
Cations and anions are collectively called ions. Once Na⁺ and Cl⁻ ions are formed, they are attracted to each other due to their opposite charges. This force that holds them together is known as an ionic bond.
- Ionic bond = electrostatic force of attraction between oppositely charged ions
- Na⁺ (positive) and Cl⁻ (negative) → attract each other → form NaCl (Sodium Chloride)
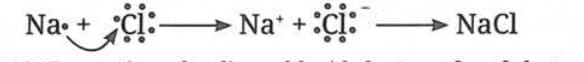
Threads of Curiosity:Ionic compounds do not exist as single units but form three-dimensional crystals with ions arranged in a repeating pattern. In sodium chloride (NaCl), each sodium ion is surrounded by six chloride ions and each chloride ion by six sodium ions. This regular arrangement of oppositely charged ions forms a crystal structure called a crystal lattice.
Some elements like sulfur have six electrons in their outer shell and need two more to complete their octet. When a sulfur atom gains two electrons, it forms a negatively charged ion with a charge of 2⁻, written as S²⁻.
A. Naming Ionic Compounds
Ionic compounds are named by writing the cation first, followed by the anion. Simple anions end with "-ide" (e.g., chloride, sulfide), while polyatomic anions generally do not end with -ide.
- Metals → generally form cations
- Non-metals → generally form anions
- Ionic compounds are typically formed when metals combine with non-metals
- Examples: Sodium Chloride, Calcium Oxide, Magnesium Sulfide
Some ions are made up of two or more elements combined together - these are called polyatomic ions.
- Polyatomic ions contain multiple atoms acting as a single ion
- Their names generally do not end with -ide
- Examples: SO₄²⁻ (Sulphate), NO₃⁻ (Nitrate), OH⁻ (Hydroxide)
Table: Some common monoatomic ions
| Name of ion | Formula | Valency |
|---|---|---|
| Sodium | Na⁺ | 1 |
| Lithium | Li⁺ | 1 |
| Potassium | K⁺ | 1 |
| Silver | Ag⁺ | 1 |
| Calcium | Ca²⁺ | 2 |
| Barium | Ba²⁺ | 2 |
| Iron (Ferrous) | Fe²⁺ | 2 |
| Iron (Ferric) | Fe³⁺ | 3 |
| Copper (Cuprous) | Cu⁺ | 1 |
| Copper (Cupric) | Cu²⁺ | 2 |
| Magnesium | Mg²⁺ | 2 |
| Zinc | Zn²⁺ | 2 |
| Aluminium | Al³⁺ | 3 |
| Fluoride | F⁻ | 1 |
| Chloride | Cl⁻ | 1 |
| Bromide | Br⁻ | 1 |
| Iodide | I⁻ | 1 |
| Oxide | O²⁻ | 2 |
| Sulfide | S²⁻ | 2 |
Table: Some common polyatomic ions
| Name of ion | Formula | Valency |
|---|---|---|
| Hydroxide | OH⁻ | 1 |
| Nitrate | NO₃⁻ | 1 |
| Hydrogencarbonate | HCO₃⁻ | 1 |
| Carbonate | CO₃²⁻ | 2 |
| Sulfate | SO₄²⁻ | 2 |
| Ammonium | NH₄⁺ | 1 |
9.5 Writing Chemical Formulae
9.5.1 Writing Chemical Formulae of Covalent Compounds
(i) Write the symbols of the constituent elements of the compound.
(ii) Write the valencies of these elements>
(iii) Crossover the valencies of the combining atoms and write them as subscripts after the symbols of elements.
Examples:
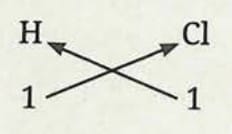
Formula of hydrogen chloride:
- Symbol of element: H and Cl
- Valency: 1 and 1
- The formula of the compound would be HCl.
- If the valency is one after criss-crossing, it is not written.
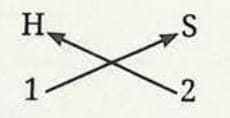
 Formula of hydrogen sulfide:
Formula of hydrogen sulfide:
- Symbol of element: H and S
- Valency: 1 and 2
- The formula of the compound would be H₂S.
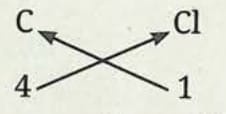
Formula of carbon tetrachloride:
- Symbol of element: C and Cl
- Valency: 4 and 1
- The formula of the compound would be CCl₄.
9.5.2 Writing Chemical Formulae of Ionic Compounds
To write the chemical formula of an ionic compound, follow these steps:
- Step 1 → Write the symbol of the cation first, followed by the symbol of the anion
- Step 2 → Write the charges under the symbols (not as superscripts)
- Step 3 → Criss-cross the charges (only the numbers) to get the subscripts
- Step 4 → Simplify the subscripts by dividing them by a common factor if any
The chemical formula gives the simplest ratio of elements in a compound. So after criss-crossing, always check if the subscripts can be reduced.
- Example: If you get subscripts 2 and 4 → divide both by 2 → get 1 and 2
- These simplified numbers are then used as the final subscripts in the formula
Note: The charges on the ions are not indicated in the formula of the compound.
Examples:
Formula of calcium chloride:
- Symbol: Ca and Cl
- Charge: 2+ and 1-
- Formula: CaCl₂
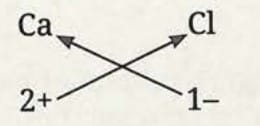
- The positive and negative charges must balance each other, and the overall structure must be neutral.
Formula of aluminium oxide:
- Symbol: Al and O
- Charge: 3+ and 2-
- Formula: Al₂O₃
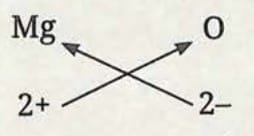
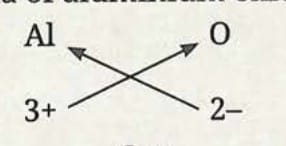 Formula of magnesium oxide:
Formula of magnesium oxide:
- Symbol: Mg and O
- Charge: 2+ and 2-
- Here, the valencies of the two elements are the same. We arrive at the formula Mg₂O₂ but it is simply written as MgO.
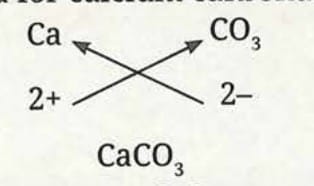
This method can also be used to write formulae of compounds of metals with other polyatomic ions, such as calcium carbonate.
Formula for calcium carbonate:
- Symbol: Ca and CO₃
- Charge: 2+ and 2-
- Here, the valencies of the two ions are the same. The formula Ca₂(CO₃)₂ is simply written as CaCO₃.
When a formula contains two or more polyatomic ions of the same type, we use brackets () around the polyatomic ion, followed by the subscript.
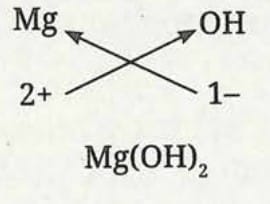
Formula of magnesium hydroxide:
- Symbol: Mg and OH
- Charge: 2+ and 1-
- Formula: Mg(OH)₂
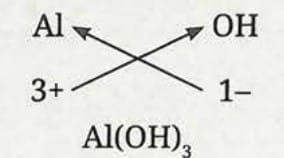
Formula of aluminium hydroxide:
- Symbol: Al and OH
- Charge: 3+ and 1-
- Formula: Al(OH)₃
Formula of aluminium sulfate:
- Symbol: Al and SO₄
- Charge: 3+ and 2-
- Formula: Al₂(SO₄)₃
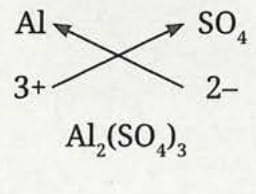
9.6 Properties of the Ionic and the Covalent Compounds
- Solubility:
- Ionic compounds (e.g., sodium chloride, copper sulfate) are soluble in water but insoluble in kerosene and petrol.
- Covalent compounds (e.g., camphor, naphthalene) are insoluble in water but soluble in kerosene and petrol.
- Electrical Conductivity:
- Ionic compounds do not conduct electricity in solid state (ions are fixed).
- They conduct electricity in aqueous or molten state (ions are free to move).
- Covalent compounds generally do not conduct electricity (no free ions), even in solution (e.g., sugar).
- Melting and Boiling Points:
- Ionic compounds have high melting and boiling points due to strong electrostatic forces.
- Covalent compounds usually have lower melting and boiling points.
9.7 Molecular Mass of Covalent Compounds
The masses of molecules can be found by simply adding up the masses of the atoms present in them.
Example:
1. Molecular mass of water (H₂O): Atomic mass - H = 1 u; O = 16 u Molecular mass of H₂O = (1 u × 2) + (16 u × 1) = 18 u
2. Molecular mass of carbon dioxide (CO₂): Atomic mass - C = 12 u; O = 16 u Molecular mass of CO₂ = (12 u × 1) + (16 u × 2) = 44 u
Note: Ionic compounds do not form molecules.
9.8 Formula Unit Mass of Ionic Compounds
In ionic compounds, the collection of the simplest whole number ratio of ions is termed as a formula unit. The mass of a formula unit is called the formula unit mass.
Example:
1. Formula unit mass of sodium oxide (Na₂O): Atomic mass - Na = 23 u; O = 16 u Formula unit mass of Na₂O = (23 u × 2) + (16 u × 1) = 62 u
2. Formula unit mass of calcium nitrate, Ca(NO₃)₂: Atomic mass - Ca = 40 u; N = 14 u; O = 16 u Formula unit mass of Ca(NO₃)₂ = (40 u × 1) + {(14 u × 1) + (16 u × 3)} × 2 = 164 u
Summary
- Laws of Chemical Reactions:
- Law of Conservation of Mass → Mass can neither be created nor destroyed in a chemical reaction
- Law of Definite Proportions → A compound always contains the same elements in a fixed ratio by mass, regardless of how or where it is obtained
Molecules & Atoms:
- A molecule is an electrically neutral entity of more than one atom that exists independently and shows all chemical properties
- Atoms combine to form molecules of elements or compounds to become stable, held together by a chemical bond
Types of Chemical Bonds:
- Covalent Bond → formed by sharing of electrons between atoms
- Ionic Bond → formed by transfer of electrons, where one atom loses (forms cation) and another gains electrons (forms anion)
Chemical Formulas:
- Covalent compound → formula represents the elements and number of atoms of each element
- Ionic compound → formula represents the simplest whole number ratio of atoms of different elements
Mass Calculations:
- Molecular Mass → sum of atomic masses of all atoms in a molecule
- Formula Unit Mass → sum of atomic masses of all atoms in the simplest ratio of ions in an ionic compound
FAQs on Chapter Notes: Atomic Foundations of Matter
| 1. What is the Law of Conservation of Mass? |  |
| 2. How does a physical change differ from a chemical change? |  |
| 3. What are the key postulates of Dalton's Atomic Theory? |  |
| 4. What is a covalent bond and how does it form? |  |
| 5. Can you explain the concept of ionic bonding? |  |