NCERT Solutions: Atomic Foundations of Matter
Think It Over (Page 164)
Q1: Water can be obtained from various sources. Are all these samples of water chemically identical?
Ans: Yes, all samples of pure water are chemically identical because they have the same composition (H₂O), even though their physical impurities may differ.
Q2: Oxygen is sometimes represented as O and sometimes as O₂. What is the difference between these symbols?
Ans: O represents a single oxygen atom, while O₂ represents a molecule of oxygen consisting of two oxygen atoms bonded together.
Q3: Why does dissolved salt in water conduct electricity, but sugar does not?
Ans: Salt dissociates into ions in water, which carry electric current. Sugar does not form ions when dissolved, so it does not conduct electricity.
Pause and Ponder (Page 166)
Q1. A student burns 10 g of ethanol in an open beaker. After the reaction, no residue is left in the beaker. Does this mean the Law of Conservation of Mass is violated? Explain.
Answer: No, the Law of Conservation of Mass is NOT violated. When ethanol burns, it reacts with oxygen from the air and produces carbon dioxide and water vapour, both of which escape into the atmosphere as gases. If we account for the mass of oxygen consumed from the air and the mass of carbon dioxide and water vapour released, the total mass of the products will be equal to the total mass of the reactants (ethanol + oxygen). The fact that no residue is left in the beaker does not mean mass is lost - the products have escaped as gases. The Law of Conservation of Mass is obeyed.
Q2. When 20 g of hydrogen reacts completely with 160 g of oxygen, how much water is formed according to the Law of Conservation of Mass?
Answer: According to the Law of Conservation of Mass, the total mass of products = total mass of reactants. Total mass of reactants = 20 g (hydrogen) + 160 g (oxygen) = 180 g. Therefore, mass of water formed = 180 g.
Pause and Ponder (Page 167)
Q3. A compound consists of 40% sulfur and 60% oxygen by mass. In a sample of the same compound containing 20 g of sulfur, what mass of oxygen must be present to satisfy the Law of Constant Proportions?
Answer: The compound has sulfur and oxygen in the mass ratio of 40:60 = 2:3. If 20 g of sulfur is present, then: Mass of oxygen = (60/40) × 20 = 30 g. Therefore, 30 g of oxygen must be present.
Q4. Carbon monoxide (CO) contains carbon and oxygen in the mass ratio of 3:4. How much oxygen will combine with 9 g of carbon to form carbon monoxide?
Answer: Carbon and oxygen are in the mass ratio 3:4. If 3 g of carbon combines with 4 g of oxygen, then 9 g of carbon will combine with = (4/3) × 9 = 12 g of oxygen. Therefore, 12 g of oxygen will combine with 9 g of carbon to form carbon monoxide.
Q5. The Law of Definite Proportions holds true for compounds but not for mixtures. Give reason.
Answer: In a compound, elements are chemically combined in a fixed ratio by mass, irrespective of the source or method of preparation. This fixed ratio is a characteristic property of the compound. For example, water always contains hydrogen and oxygen in the ratio 1:8 by mass.
In a mixture, however, the components are not chemically combined. They can be present in any ratio and the composition of a mixture can vary. For example, a mixture of sugar and water can be prepared in any proportion. Hence, the Law of Definite Proportions does not apply to mixtures.
Q6. Students X and Y, both prepared an oxide of copper by combining copper and oxygen in the ratios of 4:1 and 8:2, respectively. Do their results justify the Law of Constant Proportions? Explain.
Answer: Yes, their results justify the Law of Constant Proportions.
Student X: ratio of copper to oxygen = 4:1 Student Y: ratio of copper to oxygen = 8:2 = 4:1
Both ratios simplify to 4:1. Since both students obtained the same compound (the same oxide of copper) in the same fixed mass ratio of copper to oxygen, the Law of Constant Proportions is satisfied.
Pause and Ponder (Page 168)
Q7. Assertion (A): 2 g of hydrogen combines with 16 g of oxygen to form 18 g of water.
Reason (R): According to Dalton's Atomic Theory, atoms combine in a simple whole number ratio by mass to form compounds.Choose the correct option:
(i) Both A and R are true, and R is the correct explanation of A.
(ii) Both A and R are true, but R is not the correct explanation of A.
(iii) A is true, but R is false.
(iv) A is false, but R is true.
Answer: (ii) Both A and R are true, but R is not the correct explanation of A.
Assertion A is true: 2 g of hydrogen + 16 g of oxygen = 18 g of water (consistent with the Law of Conservation of Mass and the formula H₂O).
Reason R is also true: Dalton's Atomic Theory does state that atoms combine in simple whole number ratios. However, R is not the correct explanation of A. Assertion A is explained by the Law of Conservation of Mass (total mass of reactants = total mass of products), not directly by the statement that atoms combine in simple whole number ratios by mass. The combining ratio in A is by number of atoms (2 hydrogen atoms to 1 oxygen atom), not by mass.
Pause and Ponder (Page 170)
Q8. Nitrogen has five valence electrons. Draw the structure of the nitrogen molecule (N₂).
Answer: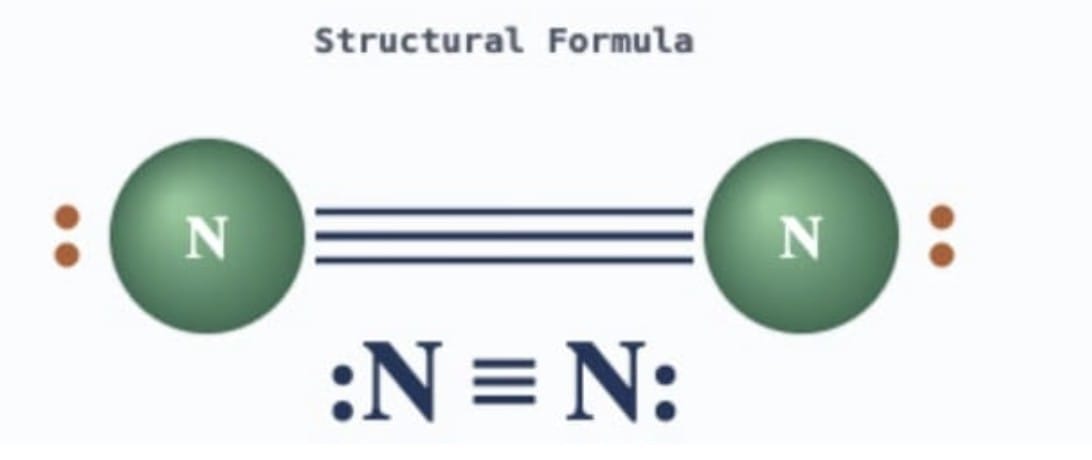 Each nitrogen atom has 5 valence electrons and needs 3 more electrons to complete its octet. So, two nitrogen atoms share three electrons each, forming a nitrogen molecule with a triple bond.
Each nitrogen atom has 5 valence electrons and needs 3 more electrons to complete its octet. So, two nitrogen atoms share three electrons each, forming a nitrogen molecule with a triple bond.
Structure: N≡N (triple bond between two nitrogen atoms)
In terms of electron sharing: each nitrogen atom contributes 3 electrons to form 3 shared pairs. Each nitrogen atom also has 1 lone pair of electrons (the remaining 2 valence electrons that are not shared). The nitrogen molecule can be represented as :N≡N: where each nitrogen has one lone pair and shares three electron pairs.
Q9. The atomic number of fluorine is 9. Explain the formation of the fluorine molecule (F₂).
Answer: The atomic number of fluorine is 9. Its electronic configuration is 2, 7 - meaning fluorine has 7 electrons in its valence shell (second shell). It needs 1 more electron to complete its octet.
Each fluorine atom shares one electron with another fluorine atom to form a fluorine molecule (F₂). The two atoms are joined by a single covalent bond (one shared pair of electrons). The molecule can be represented as F-F.
Pause and Ponder (Page 171)
Q10. Show the formation of the following molecules:(i) Carbon dioxide (CO₂)(ii) Hydrogen sulfide (H₂S)(iii) Ammonia (NH₃)
Answer:
(i) Carbon dioxide (CO₂):Carbon (atomic number 6) has 4 valence electrons and needs 4 more to complete its octet. Oxygen (atomic number 8) has 6 valence electrons and needs 2 more. Each oxygen atom shares 2 electrons with the carbon atom. Carbon shares 2 electrons with each of the two oxygen atoms. Thus, two double bonds are formed: O=C=O. Carbon dioxide has two double covalent bonds.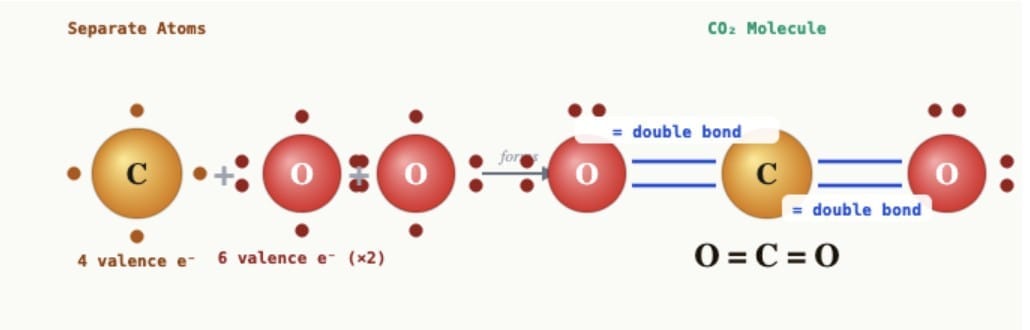
(ii) Hydrogen sulfide (H₂S):Sulfur (atomic number 16) has 6 valence electrons and needs 2 more electrons to complete its octet. Hydrogen needs 1 electron to complete its duplet. Sulfur shares one electron each with two hydrogen atoms. Each hydrogen atom shares one electron with sulfur. Two single bonds are formed: H-S-H (each bond is a single covalent bond).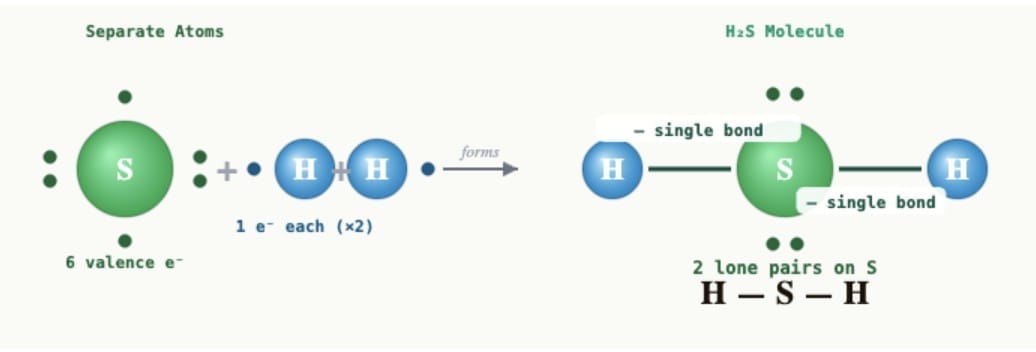
(iii) Ammonia (NH₃):Nitrogen (atomic number 7) has 5 valence electrons and needs 3 more electrons to complete its octet. Hydrogen needs 1 electron to complete its duplet. Nitrogen shares one electron each with three hydrogen atoms. Each hydrogen atom shares one electron with nitrogen. Three single bonds are formed: H-N-H with one H below (or depicted as N at centre bonded to three H atoms). Ammonia has three single covalent bonds and one lone pair on nitrogen.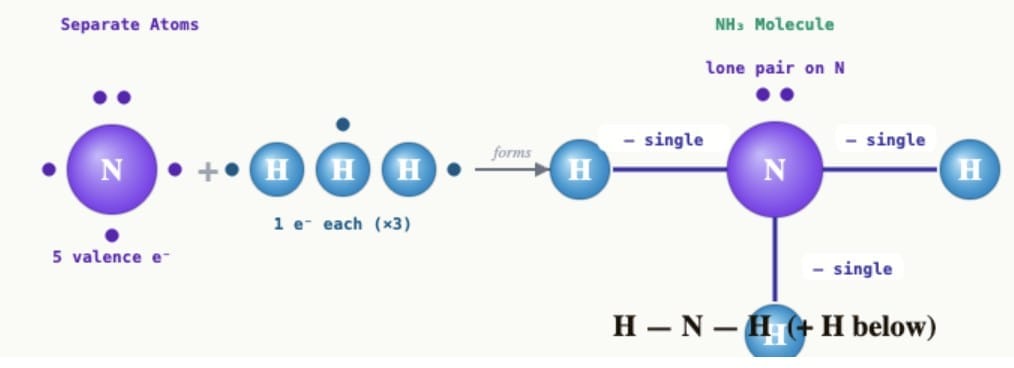
Q11. Neon (atomic number 10) neither transfers nor shares its valence electrons. Explain.
Answer: The atomic number of neon is 10. Its electronic configuration is 2, 8 - meaning neon has 8 electrons in its outermost (valence) shell. This is a complete octet, which is the most stable electronic configuration.
Since neon's valence shell is already full (octet complete), it does not need to gain, lose, or share any electrons to become stable. Therefore, neon neither transfers nor shares its valence electrons and does not form chemical bonds. It exists as a monoatomic gas.
Pause and Ponder (Page 173)
Q12. What kind of ion will oxygen (O) form?
Answer: Oxygen (atomic number 8) has electronic configuration 2, 6. It has 6 electrons in its valence shell and needs 2 more electrons to complete its octet. Since oxygen has more than 4 valence electrons, it gains electrons rather than losing them. Oxygen gains 2 electrons to form an oxide anion with a charge of 2-, represented as O²⁻.
Q13. Fill in the blanks.Among magnesium and chlorine, magnesium atom can give two electrons to become Mg²⁺. However, chlorine can take only one electron to become ___________. Now, ___________ ion of magnesium and ___________ ions of chlorine combine to give magnesium chloride.
Answer: Among magnesium and chlorine, magnesium atom can give two electrons to become Mg²⁺. However, chlorine can take only one electron to become Cl⁻. Now, one ion of magnesium and two ions of chlorine combine to give magnesium chloride (MgCl₂).
Q14. Show the formation of cations of potassium (K) and calcium (Ca) atoms, and the formation of their corresponding chlorides using diagrams.
Answer:
Formation of Potassium cation (K⁺):Potassium (atomic number 19) has electronic configuration 2, 8, 8, 1. It has 1 electron in its valence shell. To attain a stable configuration, it loses this 1 electron: K → K⁺ + e⁻ Potassium becomes K⁺ ( K⁺ has 19 protons and 18 electrons).
Formation of Potassium chloride (KCl):K⁺ (valency 1) + Cl⁻ (valency 1) → KCl One potassium ion combines with one chloride ion to form potassium chloride.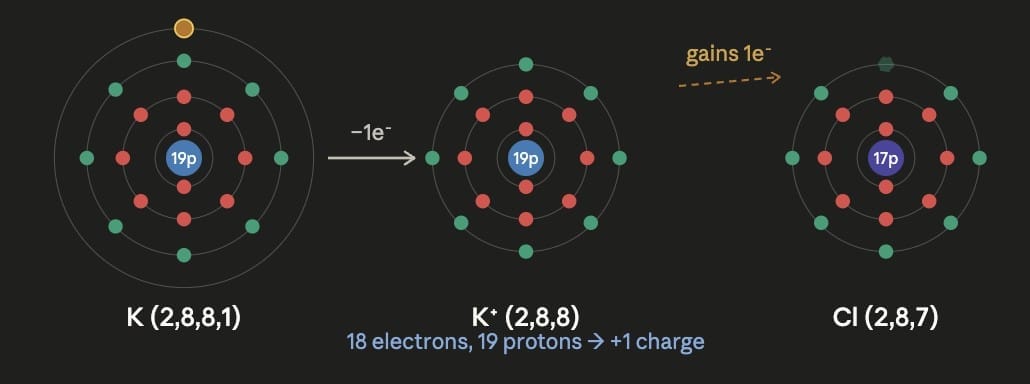
Formation of Calcium cation (Ca²⁺):Calcium (atomic number 20) has electronic configuration 2, 8, 8, 2. It has 2 electrons in its valence shell. To attain a stable configuration, it loses both electrons: Ca → Ca²⁺ + 2e⁻ Calcium becomes Ca²⁺ (20 protons and 18 electrons).
Formation of Calcium chloride (CaCl₂):Ca²⁺ (valency 2) + 2 Cl⁻ (valency 1 each) → CaCl₂ One calcium ion combines with two chloride ions to form calcium chloride.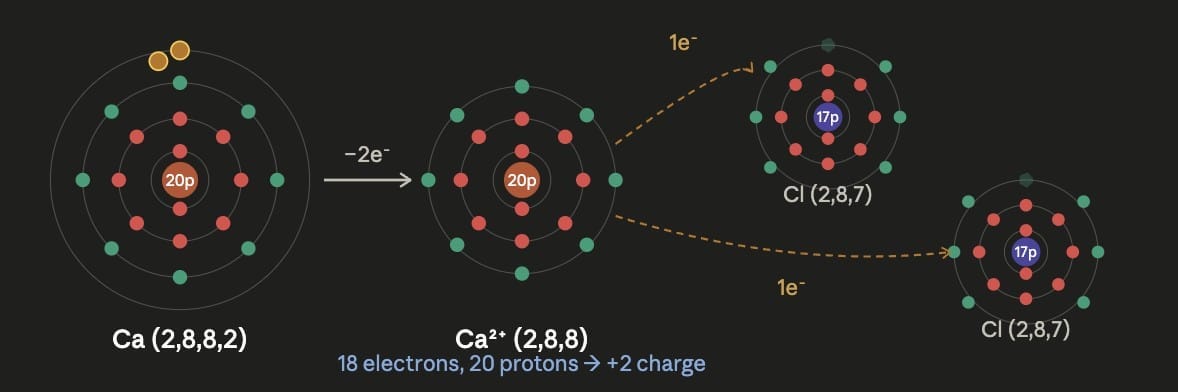
Q15. Illustrate how sodium sulfide (Na₂S) is formed.
Answer: 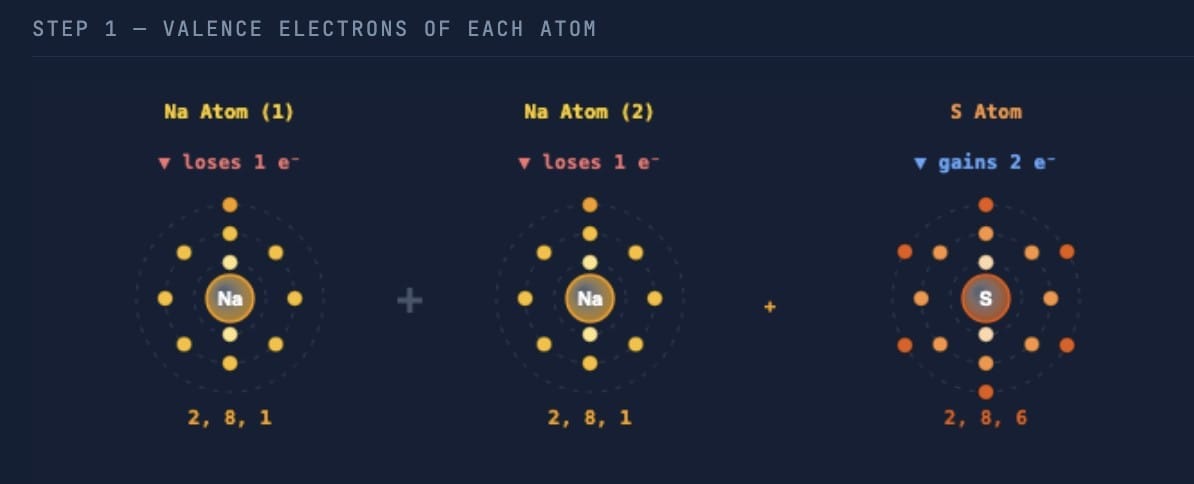 Sodium (atomic number 11) has electronic configuration 2, 8, 1. It has 1 valence electron, which it loses to form Na⁺: Na → Na⁺ + e⁻
Sodium (atomic number 11) has electronic configuration 2, 8, 1. It has 1 valence electron, which it loses to form Na⁺: Na → Na⁺ + e⁻
Sulfur (atomic number 16) has electronic configuration 2, 8, 6. It has 6 valence electrons and needs 2 more electrons to complete its octet. It gains 2 electrons to form S²⁻: S + 2e⁻ → S²⁻
Two sodium atoms each lose one electron (total 2 electrons) which are gained by one sulfur atom: 2Na → 2Na⁺ + 2e⁻ S + 2e⁻ → S²⁻
The two Na⁺ ions and one S²⁻ ion are held together by electrostatic force of attraction (ionic bond) to form sodium sulfide: 2Na⁺ + S²⁻ → Na₂S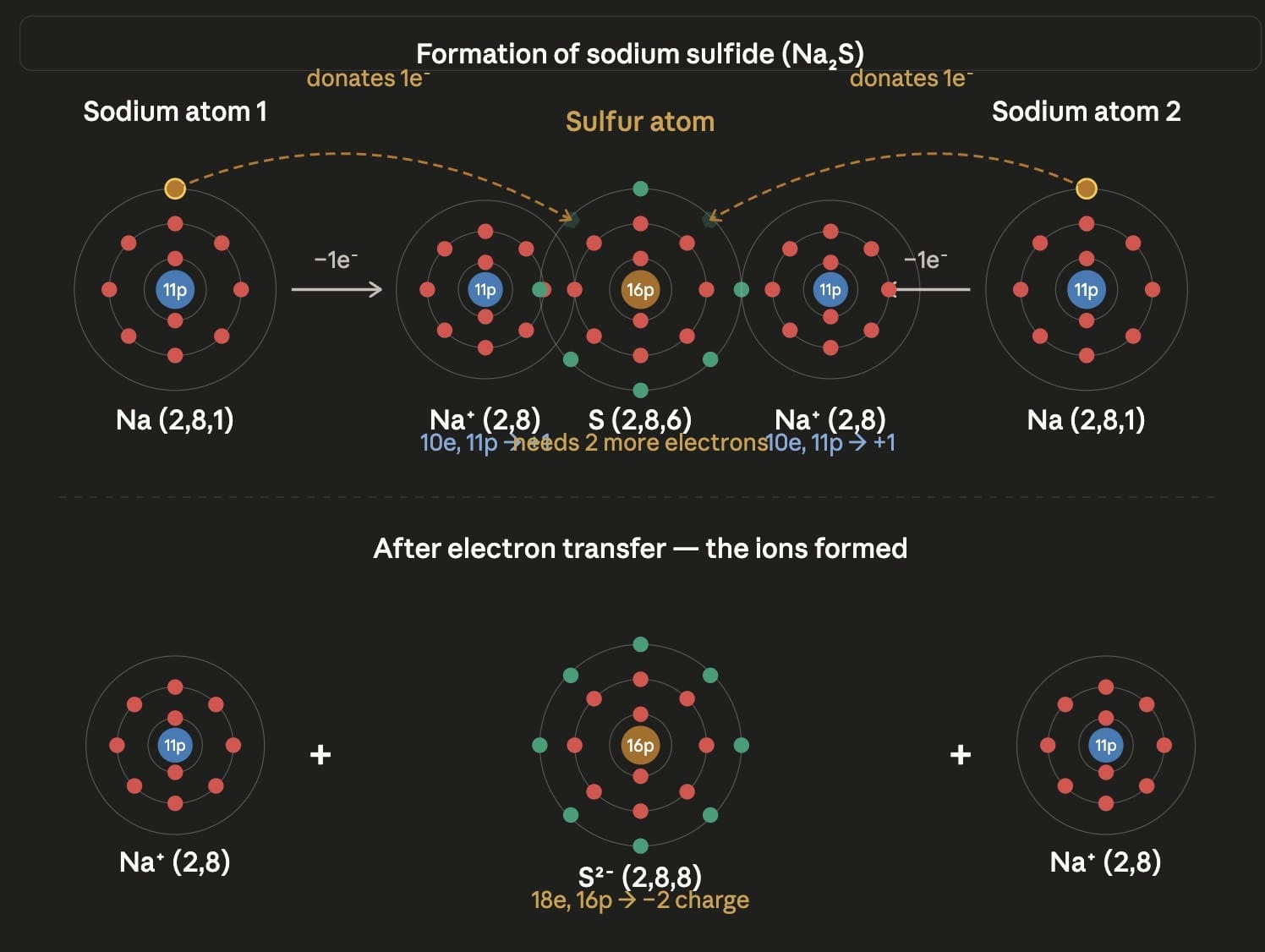
Pause and Ponder (Page 177)
Q16. Name the following:(i) CO₂(ii) NO₂(iii) SF₆(iv) PCl₃
Answer: (i) CO₂ - Carbon dioxide (ii) NO₂ - Nitrogen dioxide (iii) SF₆ - Sulfur hexafluoride (iv) PCl₃ - Phosphorus trichloride
Q17. Write the formula for the following:(i) Sodium hydrogencarbonate(ii) Sulfur dioxide(iii) Ferric chloride(iv) Cuprous oxide
Answer:
(i) Sodium hydrogencarbonate → NaHCO₃
(ii) Sulfur dioxide → SO₂
(iii) Ferric chloride → FeCl₃
(iv) Cuprous oxide → Cu₂O
Q18. Write the formulae for the compounds formed from the following pairs of ions:(i) Fe³⁺ and OH⁻(ii) K⁺ and CO₃²⁻
Answer: (i) Fe³⁺ and OH⁻ → Fe(OH)₃
(ii) K⁺ and CO₃²⁻ → K₂CO₃
Pause and Ponder (Page 179)
Q19. What type of chemical bond is present in a solid compound that does not conduct electricity in the solid state but conducts electricity when dissolved in water?
Answer: The compound has an ionic bond. Ionic compounds do not conduct electricity in the solid state because their ions are held in fixed positions by strong electrostatic forces. However, when dissolved in water, the ions become free to move, which allows the compound to conduct electricity. This behaviour is characteristic of ionic compounds such as sodium chloride and copper sulfate.
Q20. Metal M, with two electrons in its valence shell (M shell), reacts with oxygen to form a compound that is slightly soluble in water. Predict its:
(i) formula
(ii) type of bond
(iii) electrical conductivity of its aqueous solution.
Answer: The metal M has two electrons in its valence shell (M shell, which is the third shell). This means M has electronic configuration 2, 8, 2. This is magnesium (Mg), which forms Mg²⁺ by losing 2 electrons.
(i) Formula:Mg²⁺ combines with O²⁻ (oxide ion, valency 2). Formula: MgO (magnesium oxide)
(ii) Type of bond:Since magnesium (a metal) loses electrons and oxygen (a non-metal) gains electrons, an ionic bond is formed. Type of bond: Ionic bond
(iii) Electrical conductivity of its aqueous solution:MgO is slightly soluble in water and forms an aqueous solution. In this solution, Mg²⁺ and O²⁻ (or OH⁻) ions are present and are free to move. Therefore, its aqueous solution will conduct electricity.
Q21. Find the molecular mass of nitric acid (HNO₃).Atomic mass - H = 1 u; N = 14 u; O = 16 u.
Answer: HNO₃ contains: 1 hydrogen atom, 1 nitrogen atom, 3 oxygen atoms. Molecular mass of HNO₃ = (1 u × 1) + (14 u × 1) + (16 u × 3) = 1 + 14 + 48 = 63 u
Q22. Find the molecular mass of methane (CH₄).Atomic mass - C = 12 u; H = 1 u.
Answer: CH₄ contains: 1 carbon atom, 4 hydrogen atoms. Molecular mass of CH₄ = (12 u × 1) + (1 u × 4) = 12 + 4 = 16 u
Pause and Ponder (Page 180 )
Q23. Find the formula unit mass of potassium chloride (KCl).Atomic mass - K = 39 u; Cl = 35.5 u.
Answer: KCl contains: 1 potassium ion and 1 chloride ion. Formula unit mass of KCl = (39 u × 1) + (35.5 u × 1) = 39 + 35.5 = 74.5 u
Q24. Find the formula unit mass of magnesium hydroxide, Mg(OH)₂.Atomic mass - Mg = 24 u; O = 16 u; H = 1 u.
Answer: Mg(OH)₂ contains: 1 magnesium ion and 2 hydroxide ions. Mg: 1 atom, O: 2 atoms, H: 2 atoms. Formula unit mass of Mg(OH)₂ = (24 u × 1) + (16 u × 2) + (1 u × 2) = 24 + 32 + 2 = 58 u
Revise, Reflect, Refine
Q1. A particular element (A) has one electron in its third shell. There is another element (B) with six electrons in its second shell.
(i) How many electrons does A tend to give or take to become stable?
(ii) What kind of ion would it form?
(iii) How many electrons does B tend to give or take to become stable?
(iv) What kind of ion would it form?
(v) If A and B were to combine, what kind of bond would be formed?
(vi) What would be the formula for the compound thus formed?
Answer: Element A has 1 electron in its third shell → Electronic configuration: 2, 8, 1. This is sodium (Na). Element B has 6 electrons in its second shell → Electronic configuration: 2, 6. This is oxygen (O).
(i) Element A (Na) has 1 valence electron. It tends to give 1 electron to become stable (achieving the configuration of neon: 2, 8).
(ii) By losing 1 electron, A forms a cation with a charge of +1. It would form Na⁺ (a positively charged ion / cation).
(iii) Element B (O) has 6 valence electrons. It needs 2 more to complete its octet. It tends to take 2 electrons to become stable.
(iv) By gaining 2 electrons, B forms an anion with a charge of 2-. It would form O²⁻ (a negatively charged ion / anion).
(v) If A (Na) loses an electron and B (O) gains electrons, the bond formed is an ionic bond (electrostatic force of attraction between the oppositely charged ions Na⁺ and O²⁻).
(vi) A = Na⁺ (valency 1), B = O²⁻ (valency 2). Formula: Na₂O (sodium oxide)
Q2. An element X has six electrons in its outer shell and forms a diatomic molecule.
(i) Why would that be so?
(ii) What kind of bond would it form?
(iii) Draw the structure of the molecule it would form.
(iv) A certain other element Y has two electrons in its second shell. Draw the structure of the molecule that X would form with Y.
Answer: Element X has 6 valence electrons and needs 2 more to complete its octet. This is oxygen (O), atomic number 8.
(i) X has 6 valence electrons and needs 2 more to complete its octet, so two X atoms share electrons and form a diatomic molecule.
(ii) It forms a covalent bond by sharing electrons.
(iii) Structure: X₂ molecule with a double covalent bond between the two atoms (X = X).
×× •• ××
O == O
×× •• ××
(iv) Element Y has 2 valence electrons, so it forms an ionic compound with X. Y donates 2 electrons and X gains 2 electrons, forming YX (like MgO) with ionic bonding.
•• •• ••
O == C == O
•• •• ••
Q3. You want to design a new ionic compound, where the total positive charge is 6+ and the total negative charge is 6-. Which of the following combinations gives the correct number of ions?(i) 2 Al³⁺ and 3 Cl⁻(ii) 3 Mg²⁺ and 1 PO₄³⁻(iii) 2 Fe³⁺ and 3 O²⁻(iv) 3 Ca²⁺ and 2 SO₄²⁻
Answer: Check total positive and negative charges:
(i) 2 Al³⁺: total positive = 2 × 3 = 6+; 3 Cl⁻: total negative = 3 × 1 = 3-. Not balanced (6+ ≠ 3-).
(ii) 3 Mg²⁺: total positive = 3 × 2 = 6+; 1 PO₄³⁻: total negative = 1 × 3 = 3-. Not balanced.
(iii) 2 Fe³⁺: total positive = 2 × 3 = 6+; 3 O²⁻: total negative = 3 × 2 = 6-. Balanced! Total positive = 6+, total negative = 6-.
(iv) 3 Ca²⁺: total positive = 3 × 2 = 6+; 2 SO₄²⁻: total negative = 2 × 2 = 4-. Not balanced.
Answer: (iii) 2 Fe³⁺ and 3 O²⁻
Q4. Choose the correct statement(s) and correct the false statement(s).
(i) Elements are made up of molecules and compounds are made up of atoms.
(ii) The molecule of a compound is always made up of two or more atoms of the same kind.
(iii) One molecule of nitrogen gas contains three nitrogen atoms.
(iv) Water is made of two hydrogen atoms, covalently bonded with one oxygen atom.
Answer:
(i) FALSE. Correct statement: Elements are made up of atoms and compounds are made up of molecules (which contain atoms of two or more different elements). For example, iron is an element made up of iron atoms; water is a compound made up of molecules each containing 2 hydrogen atoms and 1 oxygen atom.
(ii) FALSE. Correct statement: The molecule of a compound is made up of two or more atoms of different kinds (different elements). For example, HCl is made of hydrogen and chlorine atoms (different kinds). It is the molecule of an element (like H₂ or O₂) that is made up of atoms of the same kind.
(iii) FALSE. Correct statement: One molecule of nitrogen gas (N₂) contains two nitrogen atoms, not three.
(iv) TRUE. Water (H₂O) is indeed made of two hydrogen atoms covalently bonded with one oxygen atom.
Q5. Write the chemical formulae for the following compounds.(i) Aluminium nitrate(ii) Calcium oxide(iii) Ferric oxide
Answer:
(i) Aluminium nitrate:Al³⁺ (valency 3) and NO₃⁻ (valency 1). Formula: Al(NO₃)₃
(ii) Calcium oxide:Ca²⁺ (valency 2) and O²⁻ (valency 2). Since valencies are the same, simplifies to: Formula: CaO
(iii) Ferric oxide:Fe³⁺ (ferric, valency 3) and O²⁻ (valency 2). Formula: Fe₂O₃
Q6. Write the formulae of the compounds formed from the following pairs of ions.(i) Ca²⁺ and Br⁻(ii) Al³⁺ and CO₃²⁻(iii) K⁺ and SO₄²⁻(iv) NH₄⁺ and Cl⁻
Answer:
(i) Ca²⁺ and Br⁻ → CaBr₂ (Charges: 2+ and 1-; calcium bromide)
(ii) Al³⁺ and CO₃²⁻ → Al₂(CO₃)₃ (Charges: 3+ and 2-; aluminium carbonate)
(iii) K⁺ and SO₄²⁻ → K₂SO₄ (Charges: 1+ and 2-; potassium sulfate)
(iv) NH₄⁺ and Cl⁻ → NH₄Cl (Charges: 1+ and 1-; ammonium chloride)
Q7. Which of the following, in Fig. 9.18, correctly represents Cl⁻ ion (Atomic number of chlorine = 17).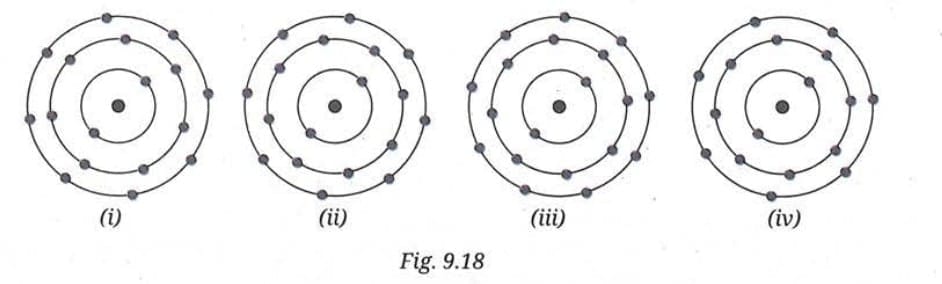
Answer: Chlorine (atomic number 17) has the electronic configuration 2, 8, 7. It has 7 electrons in its valence shell (third shell).
When chlorine gains one electron to form the chloride ion (Cl⁻), it has 18 electrons total. The electronic configuration of Cl⁻ becomes 2, 8, 8.
So the correct diagram should show:
- 17 protons in the nucleus
- 18 electrons total
- First shell: 2 electrons
- Second shell: 8 electrons
- Third shell: 8 electrons
The correct option is (ii) - which shows an atom with three shells having 2, 8, and 8 electrons (total 18 electrons), representing the Cl⁻ ion with a complete outermost shell.
Q8. Determine the formula unit mass of the following substances.
(i) Ammonium nitrate (NH₄NO₃), used as a nitrogen fertiliser, which is essential for plant growth.
(ii) Phosphoric acid (H₃PO₄), used to make phosphate fertiliser and detergents.
(iii) Sodium hydrogencarbonate (NaHCO₃), used to relieve acidity and helps in digestion.
Answer:
(i) Ammonium nitrate (NH₄NO₃):NH₄NO₃ contains: N = 2, H = 4, O = 3 Formula unit mass = (14 × 2) + (1 × 4) + (16 × 3) = 28 + 4 + 48 = 80 u
(ii) Phosphoric acid (H₃PO₄):H₃PO₄ contains: H = 3, P = 1, O = 4 Formula unit mass = (1 × 3) + (31 × 1) + (16 × 4) = 3 + 31 + 64 = 98 u
(iii) Sodium hydrogencarbonate (NaHCO₃):NaHCO₃ contains: Na = 1, H = 1, C = 12 u, O = 3 Formula unit mass = (23 × 1) + (1 × 1) + (12 × 1) + (16 × 3) = 23 + 1 + 12 + 48 = 84 u
Q9. Write the formulae for the compounds formed by the reaction of:(i) Magnesium and nitrogen(ii) Lithium and nitrogen(iii) Sodium and sulfur(iv) Aluminium and oxygen
Answer:
(i) Magnesium and nitrogen:Mg forms Mg²⁺ (valency 2); N forms N³⁻ (valency 3).Formula: Mg₃N₂ (magnesium nitride)
(ii) Lithium and nitrogen:Li forms Li⁺ (valency 1); N forms N³⁻ (valency 3). Formula: Li₃N (lithium nitride)
(iii) Sodium and sulfur:Na forms Na⁺ (valency 1); S forms S²⁻ (valency 2). Formula: Na₂S (sodium sulfide)
(iv) Aluminium and oxygen:Al forms Al³⁺ (valency 3); O forms O²⁻ (valency 2). Formula: Al₂O₃ (aluminium oxide)
Q10. Complete the Table 9.3 by writing the formulae of the compounds formed by the cations on the left and the anions at the top. LiNO₃ is given as an example.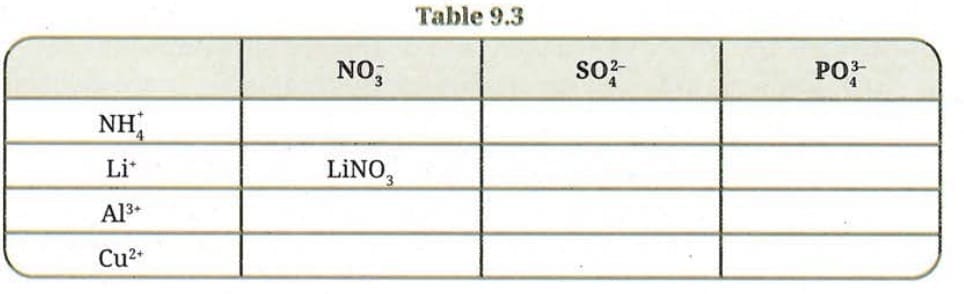
Answer:
| NO₃⁻ | SO₄²⁻ | PO₄³⁻ | |
|---|---|---|---|
| NH₄⁺ | NH₄NO₃ | (NH₄)₂SO₄ | (NH₄)₃PO₄ |
| Li⁺ | LiNO₃ | Li₂SO₄ | Li₃PO₄ |
| Al³⁺ | Al(NO₃)₃ | Al₂(SO₄)₃ | AlPO₄ |
| Cu²⁺ | Cu(NO₃)₂ | CuSO₄ | Cu₃(PO₄)₂ |
Working:
NH₄⁺ (valency 1) with NO₃⁻ (valency 1): NH₄NO₃ NH₄⁺ (valency 1) with SO₄²⁻ (valency 2): (NH₄)₂SO₄ NH₄⁺ (valency 1) with PO₄³⁻ (valency 3): (NH₄)₃PO₄
Li⁺ (valency 1) with NO₃⁻ (valency 1): LiNO₃ (given) Li⁺ (valency 1) with SO₄²⁻ (valency 2): Li₂SO₄ Li⁺ (valency 1) with PO₄³⁻ (valency 3): Li₃PO₄
Al³⁺ (valency 3) with NO₃⁻ (valency 1): Al(NO₃)₃ Al³⁺ (valency 3) with SO₄²⁻ (valency 2): Al₂(SO₄)₃ Al³⁺ (valency 3) with PO₄³⁻ (valency 3): AlPO₄ (both valency 3, subscripts both 3, divide by 3 = 1 each, so AlPO₄)
Cu²⁺ (valency 2) with NO₃⁻ (valency 1): Cu(NO₃)₂ Cu²⁺ (valency 2) with SO₄²⁻ (valency 2): CuSO₄ (both valency 2, simplifies to 1:1) Cu²⁺ (valency 2) with PO₄³⁻ (valency 3): Cu₃(PO₄)₂
Q11. 5.3 g of sodium carbonate and 6.0 g of acetic acid react to produce 2.2 g of carbon dioxide, 0.9 g of water, and 8.2 g of sodium acetate. Verify whether the law of conservation of mass is valid.
Answer: Total mass of reactants = 5.3 g (sodium carbonate) + 6.0 g (acetic acid) = 11.3 g
Total mass of products = 2.2 g (carbon dioxide) + 0.9 g (water) + 8.2 g (sodium acetate) = 2.2 + 0.9 + 8.2 = 11.3 g
Mass of reactants = Mass of products = 11.3 g
Since the total mass before and after the reaction is equal, the Law of Conservation of Mass is valid.
Q12. If a species has 11 protons, 12 neutrons and 10 electrons then:(i) what is its atomic number and mass number?(ii) is it neutral, a cation or an anion? Explain.(iii) write its electronic configuration.(iv) name the species.
Answer:
(i) Atomic number and mass number:Atomic number = number of protons = 11Mass number = number of protons + number of neutrons = 11 + 12 = 23
(ii) Is it neutral, a cation or an anion?It is a cation. A neutral atom has equal numbers of protons and electrons. This species has 11 protons but only 10 electrons. Since the number of protons (positive charges) is greater than the number of electrons (negative charges), the species carries a net positive charge of +1. A positively charged species is called a cation. It is represented as Na⁺.
(iii) Electronic configuration:The species has 10 electrons. Electronic configuration: 2, 8 (K-shell = 2, L-shell = 8)
(iv) Name of the species:Atomic number 11 corresponds to sodium. The species is a sodium cation, Na⁺ (sodium ion).
Q13. Two elements, A and B, have the following configurations:A: 2, 8, 5 B: 2, 8, 7(i) Which element is more reactive?(ii) Will A and B form ionic or covalent bonds when they combine? Explain using electron transfer or sharing.(iii) Predict the formula of the compound they would form.
Answer:
Element A has electronic configuration 2, 8, 5 → 5 valence electrons → needs 3 more electrons to complete its octet. Atomic number = 2+8+5 = 15 → This is Phosphorus (P).
Element B has electronic configuration 2, 8, 7 → 7 valence electrons → needs 1 more electron to complete its octet. Atomic number = 2+8+7 = 17 → This is Chlorine (Cl).
(i) Which is more reactive?Element B (Cl) is more reactive. Chlorine needs only 1 electron to complete its octet, whereas phosphorus needs 3 electrons. Elements that need fewer electrons to complete their octet tend to be more reactive non-metals (they have a higher tendency to gain electrons). Therefore, B (chlorine) is more reactive.
(ii) Type of bond:Both A (P) and B (Cl) are non-metals. A has 5 valence electrons and needs 3 more. B has 7 valence electrons and needs 1 more.
When non-metals combine with each other, they generally form covalent bonds by sharing electrons. A (P) shares one electron each with three B (Cl) atoms. Each Cl atom completes its octet by sharing one electron from P, and P completes its octet by sharing one electron from each of the three Cl atoms. This is electron sharing, which forms covalent bonds.
(iii) Formula of the compound:Phosphorus (A) has a valency of 3 (needs 3 electrons). Chlorine (B) has a valency of 1 (needs 1 electron). Formula: PCl₃ (phosphorus trichloride)
Q14. Assertion (A): Copper sulfate conducts electricity in the molten state but not in the solid state.
Reason (R): Copper and sulfate ions are fixed in the lattice in molten state, while in solid state they can move freely.Choose the correct option:
(i) Both A and R are true, and R is the correct explanation of A.
(ii) Both A and R are true, but R is not the correct explanation of A.
(iii) A is true, but R is false.
(iv) A is false, but R is true.
Answer: (iii) A is true, but R is false.
Assertion A is TRUE: Copper sulfate (an ionic compound) conducts electricity in the molten state but not in the solid state.
Reason R is FALSE: The reason stated in R is the opposite of what actually happens. In the solid state, copper and sulfate ions are fixed in the lattice (they cannot move freely), which is why the solid does not conduct electricity. In the molten state, the ions are free to move, which is why the molten state conducts electricity. The reason has incorrectly swapped the conditions for the solid and molten states.
Q15. The species ²⁷Al, ⁸⁰Br⁻ and ²⁰¹Hg²⁺ have 13, 35 and 80 protons, respectively. How many electrons and neutrons do they have?
Answer:
For ²⁷Al (aluminium atom, neutral):Protons = 13 (given) Electrons = 13 (neutral atom, so electrons = protons) Mass number = 27 Neutrons = Mass number - Protons = 27 - 13 = 14 neutronsElectrons = 13
For ⁸⁰Br⁻ (bromide ion):Protons = 35 (given) Br⁻ has gained 1 electron → Electrons = 35 + 1 = 36 electronsMass number = 80 Neutrons = Mass number - Protons = 80 - 35 = 45 neutrons
For ²⁰¹Hg²⁺ (mercury(II) ion):Protons = 80 (given) Hg²⁺ has lost 2 electrons → Electrons = 80 - 2 = 78 electronsMass number = 201 Neutrons = Mass number - Protons = 201 - 80 = 121 neutrons
FAQs on NCERT Solutions: Atomic Foundations of Matter
| 1. What are the basic components of an atom? |  |
| 2. How did the concept of the atom evolve over time? |  |
| 3. What is the significance of atomic mass and atomic number? |  |
| 4. What role do electrons play in chemical bonding? |  |
| 5. How do isotopes differ from one another? |  |






