Chapter Notes: Journey Inside the Atom

Introduction
Everything that you see, observe, or feel around is matter. Matter consists of tiny particles called atoms. Both living beings (like us) and non-living things (like a house) are ultimately composed of atoms. These atoms are so tiny that they cannot be seen with the naked eye.
The central question of this chapter is: Is an atom truly the smallest unit of matter, or can it be divided even further? Scientists have been exploring whether atoms are divisible, what their constituents are, and how these constituents are arranged.
1. Rediscovering the Roots of Atomic Theory
Ancient Ideas About Matter
The idea of the atom is more than 2,000 years old. Ancient thinkers in both India and Greece independently pondered the question: What is everything made up of?
Acharya Kanada (India):
- Suggested that if matter (dravya) is divided repeatedly, you will reach a stage where you encounter the smallest particles that can no longer be divided.
- He called these particles parmanus.
- His ideas are recorded in the Sanskrit text Vaisesika Sutras.
- A parmanu is infinitely small and cannot be perceived by the senses.
- Combinations of parmanus form dyads (groups of two parmanus) and triads (groups of three parmanus).
- Out of these combinations, the whole of the material universe including the bodies of living beings is created.
- However, this description does not specify the proportions in which parmanus combine to form different substances.
Greek Philosophers - Leucippus and Democritus:
- Proposed a similar idea independently.
- They called these indivisible particles atomos (in Greek, atomos means indivisible).
Important Note: The concept of 'atom' originated as an imaginary idea rather than from experimental observations.
Dalton's Atomic Theory (1808)
Many centuries later, in 1808, John Dalton proposed his atomic theory based on scientific experiments of that time. He proposed that:
- All matter is composed of indivisible particles called atoms.
- Atoms are the fundamental building blocks of matter that cannot be broken down into smaller parts.
- Dalton's atomic theory was the first scientific description of how matter is made.
- It became the starting point for the current understanding of atomic structure.
Following Dalton's theory, scientists were curious to know:
- What are atoms made up of?
- What would atoms look like if we could see them?
- What makes the atoms of one element different from the atoms of another element?
2. A Short Historical Journey Through Atomic Models
More than a hundred years ago, scientists tried to imagine what atoms might look like by proposing simple models. As new experiments were performed and new evidence came to light, these models were changed and improved. Although we now know that the early models were not fully correct, they are still important because they show how science moves forward - one step at a time, driven by curiosity, questioning, and experimentation.
Until the late 19th century, atoms were thought to be the smallest, indivisible units of matter. However, scientists discovered that certain elements emit invisible energy and particles called radiation, a phenomenon known as radioactivity. This showed that atoms must be composed of smaller particles, proving that they were not indivisible as previously believed.
Discovery of the Electron (J. J. Thomson, 1897)
In 1897, J. J. Thomson studied the conduction of electric current through gases at a very low pressure. He used a glass tube with two electrodes and applied a high voltage. He observed rays moving from the cathode (negative electrode) to the anode (positive electrode). These were called cathode rays.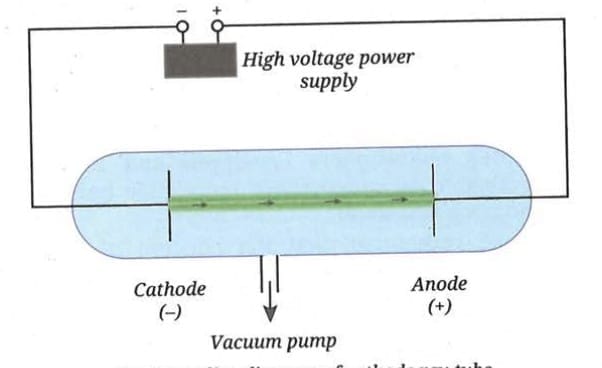
By studying these cathode rays in electric and magnetic fields, he concluded that:
- They are streams of negatively charged particles.
- These particles have a much smaller mass than atoms.
- These particles, later called electrons, were emitted from atoms.
- This indicated that atoms are composed of smaller subatomic components.
The charge of an electron is -1.602 x 10-19 C, taken as -1 as a matter of convention.
It was found that the nature of cathode rays was independent of the material of the cathode and the gas filled in the cathode ray tube. This showed that electrons are a fundamental component of all atoms, present in every element.
J. J. Thomson discovered the electron, the first identified subatomic particle present in all atoms. He received the Nobel Prize in Physics in 1906 for his work on the electrical conductivity of gases. As head of the Cavendish Laboratory, he guided many scientists, including Ernest Rutherford.
2.1 Thomson's Model of an Atom
When J. J. Thomson discovered electrons, he faced a puzzle - atoms are neutral, so where is the positive charge present?
To solve this, Thomson proposed:
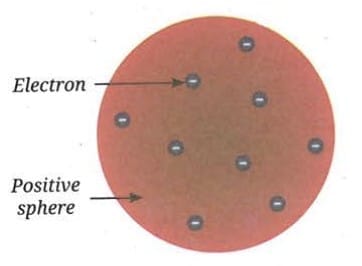
- The atom is a sphere of positive charge with electrons distributed throughout it.
- This model was compared to a pudding with plums embedded in it - called the plum pudding model.
- A more familiar comparison is a watermelon, where the red pulp represents the positively charged matter and the seeds represent electrons distributed throughout the atom.
- This was the first genuine attempt to describe how the atom's positive and negative charges stay balanced.
2.2 Testing Thomson's Model: The Gold Foil Experiment
In 1911, Geiger and Marsden, working under Ernest Rutherford, tested Thomson's model of the atom through the famous gold foil experiment.
Setup and Procedure:
- They aimed a narrow beam of alpha (symbol α) particles at an extremely thin sheet of gold foil.
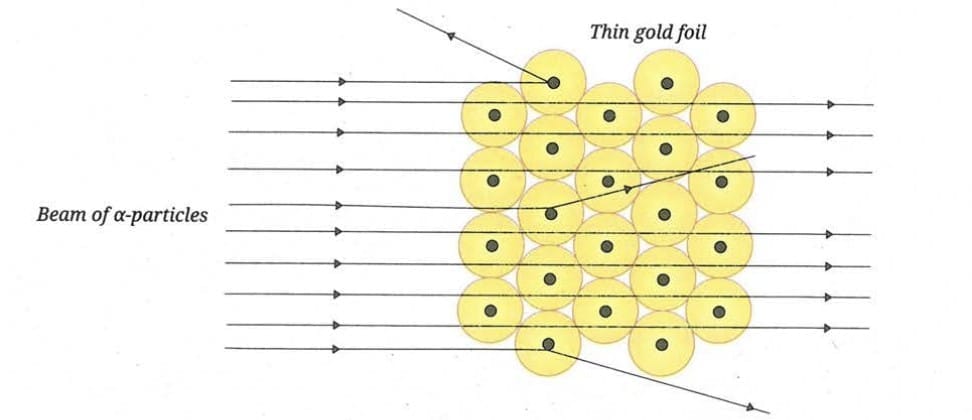
- Alpha particles are tiny, positively charged particles emitted from certain radioactive elements.
- An alpha particle is actually a nucleus of a helium atom containing two protons and two neutrons.
Expected Result (according to Thomson's model):
- Since positive charge in Thomson's model was spread out evenly, alpha particles were expected to pass straight through or be deflected only slightly.
Actual Observations:
- Most particles passed through undeflected (as expected).
- Some were sharply deflected at large angles (unexpected).
- A few even bounced back (very unexpected).
This deflection from the straight path is called scattering. Hence, the gold foil experiment is also called an alpha-ray scattering experiment.
Conclusion:Thomson's model failed to explain the results of the gold foil experiment, particularly the deflection of some alpha-particles through large angles and that most of the alpha-particles passed undeflected.
A. Rutherford's Model of an Atom
From the gold foil experiment, Rutherford concluded that the positive charge of an atom is not spread throughout but remains concentrated in an extremely small region called the nucleus.
He proposed that:
- Most of an atom is empty space, as most alpha-particles passed through the gold foil without any deflection.
- The nucleus is dense, contains all the positive charge and most of the mass of an atom.
- The electrons revolve around the nucleus, somewhat like planets orbiting the Sun.
- Hence, this model is called the planetary model of the atom.
 Important measurements:
Important measurements:
- The diameter of an atom is approximately 10-10 m.
- The diameter of the nucleus is approximately 10-15 m.
- The nucleus is about 105 (one lakh) times smaller than the atom.
- If an atom were of the size of a cricket ground (about 100 m across), the nucleus would be just a tiny black pepper grain (a few mm) at the centre.
Meet a Scienrist
Ernest Rutherford was born in New Zealand and later worked with J. J. Thomson in Cambridge. He is known as the Father of Nuclear Physics and discovered the atomic nucleus. He won the 1908 Nobel Prize in Chemistry and proposed the nuclear model of the atom in 1911.
B. Limitations of Rutherford's Model
While the idea of a central nucleus was a major step forward, Rutherford's model could not explain why atoms are stable.
- A particle moving in a circular path is constantly changing direction, which means it is accelerating.
- If a negatively charged electron keeps accelerating around the nucleus, it should lose energy.
- Losing energy would make it spiral inward and eventually fall into the positively charged nucleus.
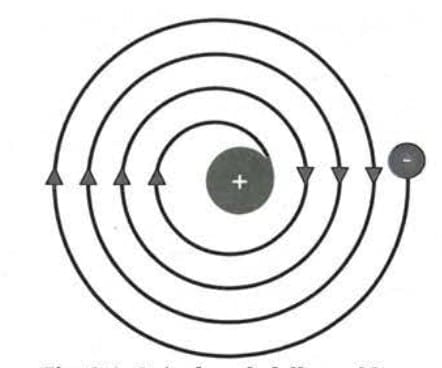
- If that really happened, atoms would collapse and would not exist!
- But in reality, atoms are stable - that is why matter around us stays intact.
- This meant that Rutherford's model was not completely correct, and a new explanation was needed to describe how electrons remain in motion without collapsing into the nucleus.
C. Discovery of the Proton
Rutherford showed that the nucleus carries positive charge which comes from the particles called protons.
- Protons are much heavier than electrons.
- Protons possess a charge equal and opposite to that of electrons (charge = +1).
- For an atom to be electrically neutral, the number of protons must be equal to the number of electrons.
- Example: A helium atom has 2 protons and 2 electrons. A sodium atom has 11 protons and 11 electrons. The total positive charge equals total negative charge, so they are electrically neutral.
- Similarly, all atoms are electrically neutral.
2.3 Bohr's Model of the Atom
To explain why atoms are stable, Niels Bohr proposed a new model of the atom in 1913.
According to Bohr:
- Electrons do not move randomly around the nucleus but follow fixed circular paths called stationary states, orbits, or shells. In each shell, an electron has a definite amount of energy, so these shells are also called energy levels.
- These shells are represented by the letters K, L, M, N, ... or by the numbers n = 1, 2, 3, 4, ... .
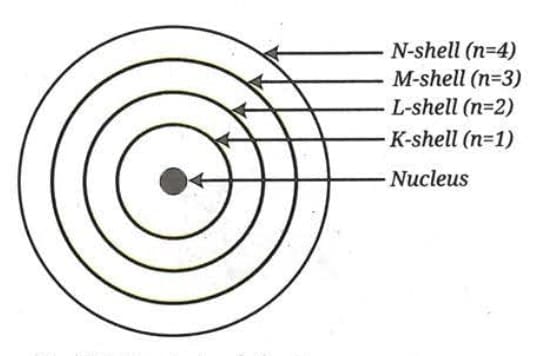
- Electrons can revolve only in these allowed shells, and not in between them. While moving in a fixed shell, an electron does not lose energy.
- The first energy level K (n = 1) is the one closest to the nucleus and has the least energy.
- The energy of these levels increases as we move away from the nucleus. That is, the energy of an electron in the L-shell (n = 2) is more than that of an electron in the K-shell (n = 1). The farther away a shell is from the nucleus, the higher is its energy.
- An electron can move to another shell by absorbing or releasing a fixed amount of energy equal to the difference between the energies of the two levels.
- Each shell can hold only a certain number of electrons.
How Bohr's model explains stability:
- In a stationary state, the energy of an electron remains constant, even though it is in motion around the nucleus.
- This concept of stationary states (introduced as a postulate) explains why electrons do not lose energy while revolving - they are in fixed states where energy is constant.
- Bohr's model could explain many experimental observations and marked a major step in understanding the atomic structure.
Threads of Curiosity - Why are Bohr's shells called K, L, M, N and not A, B, C, D?The naming came from early X-ray experiments by the physicist Charles Barkla, who called the first observed X-ray line K. He didn't start naming from A to leave room for possible discovery of a series earlier than the K series, although none were ever found. Bohr adopted the same notation for atomic shells.
Note: Later, even Bohr's model was found to have limitations, and another model, the quantum mechanical model, was proposed. You will learn about it in higher grades.
Meet a Scientist
Niels Bohr was a professor at Copenhagen University who studied atomic structure. He explained how electrons remain stable around the nucleus, improving earlier models of the atom. He received the Nobel Prize in 1922 for his work on atomic structure.
3. What Components Contribute to the Mass of an Atom?
Rutherford's model showed that most of the mass of an atom is concentrated in its nucleus. Electrons that revolve around the nucleus are so light that their mass can be ignored.
However, something puzzling appeared early in the 20th century:
- A hydrogen atom has one proton.
- A helium atom has two protons.
- Yet the mass of a helium atom is about four times that of a hydrogen atom, not double.
This led scientists to wonder whether besides protons, there is something else in the nucleus adding mass without affecting its charge.
Meet a Scientist
The discovery of the neutron marked a new era in atomic physics. Since neutrons have no charge, they can easily enter atomic nuclei, leading to advances like artificial radioactivity and the splitting of uranium atoms. This led to the atomic age, enabling the development of nuclear power as well as nuclear weapons.
3.1 Discovery of the Neutron
In 1932, this problem was solved by James Chadwick (a student of Ernest Rutherford).
- He discovered a new subatomic particle with a mass nearly equal to that of a proton but no electrical charge.
- This neutral particle was named neutron and is usually represented by the symbol 'n'.
- Neutrons are found in the nucleus of all atoms except hydrogen.
- Thus, the mass of an atom comes mainly from its protons and neutrons packed tightly in the nucleus.
- This also explains why atoms are heavier than the mass of their total number of protons.
Table : Symbols and Relative Charges of Subatomic Particles
| S.No. | Subatomic Particle | Symbol | Relative Charge |
|---|---|---|---|
| 1 | Electron | e- | -1 |
| 2 | Proton | p+ | +1 |
| 3 | Neutron | n0 | 0 |
Why don't protons in the nucleus push each other away?
- Every proton inside the nucleus repels every other proton because they all carry positive charge.
- Neutrons being neutral help reduce this repulsion by intervening and increasing the distance between protons.
- Neutrons also strengthen the nuclear force - the force that binds all particles together.
- So, heavier atoms need many more neutrons to hold everything in the nucleus tightly bound.
- For example: Iron has 26 protons and 30 neutrons; Uranium has 92 protons and 146 neutrons.
James Chadwick won the Nobel Prize in Physics in 1935 for his discovery of the neutron.
India's Scientific Contributions
The Bhabha Atomic Research Centre (BARC), Mumbai, conducts advanced neutron-scattering experiments using reactors like Dhruva reactor. These studies help understand materials such as superconductors, batteries, and drug molecules, leading to improvements in medicines, energy storage, and industrial materials in India.
Note: By 1869, scientists knew about 69 elements, most of which were naturally occurring. Today, 118 chemical elements are known, including some that are artificially created, and the search for new elements continues.
4. Symbols of Elements
John Dalton realised the need for a standard way to represent elements and compounds to make the study of chemistry easier. In 1803, he introduced the first pictorial symbols to represent the known elements.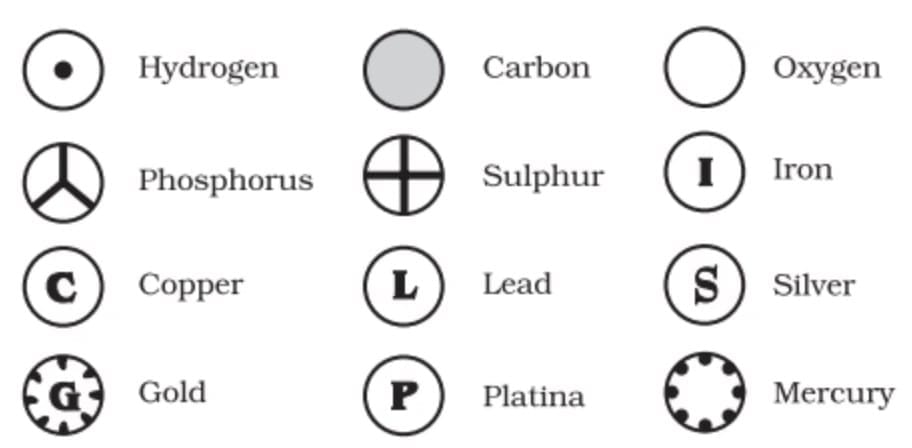
In 1813, Berzelius suggested that the symbols of elements should be derived from their Latin names. Thus came alphabetic chemical symbols.
Nowadays, the International Union of Pure and Applied Chemistry (IUPAC), an international scientific organisation, approves the names and symbols of elements.
Rules for writing chemical symbols (IUPAC norms):
- Many symbols are the first letter or the first two letters of the name of the element.
- The first letter of a symbol is always written as a capital letter (uppercase) while the second letter (if there is one) is written as a small letter (lowercase). For example, hydrogen - H; aluminium - Al (not AL); cobalt - Co (not CO).
- Symbols of some elements are formed from the first letter of the name and a letter other than the second letter in the name. For example, chlorine - Cl; zinc - Zn.
- Symbols of some elements come from their Latin, Greek, or German names rather than English. For example, iron is Fe (from Latin ferrum), mercury is Hg (from Greek hydrargyros), and tungsten is W (from German wolfram).
Scientists use these symbols instead of full names because they are internationally recognised and allow scientists worldwide to communicate clearly, regardless of language barriers.
Table: Names of Some Common Elements and Their Symbols
| Element | Symbol | Element | Symbol | Element | Symbol |
|---|---|---|---|---|---|
| Aluminium | Al | Copper (Cuprum) | Cu | Nitrogen | N |
| Argon | Ar | Fluorine | F | Oxygen | O |
| Barium | Ba | Gold (Aurum) | Au | Potassium (Kalium) | K |
| Boron | B | Hydrogen | H | Silicon | Si |
| Bromine | Br | Iodine | I | Silver (Argentum) | Ag |
| Calcium | Ca | Iron (Ferrum) | Fe | Sodium (Natrium) | Na |
| Carbon | C | Lead (Plumbum) | Pb | Sulfur | S |
| Chlorine | Cl | Magnesium | Mg | Uranium | U |
| Cobalt | Co | Neon | Ne | Zinc | Zn |
5. Atomic Number
The atoms of an element are all alike but different from the atoms of other elements. They differ in the number of electrons and protons in them.
- The number of protons in the nucleus of an atom of an element is known as its atomic number.
- It is designated by the symbol Z.
- This number determines the identity of an element and its chemical behaviour.
- Since the atom as a whole is neutral, the number of protons equals the number of electrons orbiting the nucleus.
Examples:
- Hydrogen: atomic number = 1 (1 proton, 1 electron)
- Helium: atomic number = 2 (2 protons, 2 electrons)
Elements with different atomic numbers are distinct from each other, and the atomic number uniquely identifies an element.
6. Mass Number
The total number of protons and neutrons present in the nucleus of an atom is called its mass number, and is denoted by A. The protons and neutrons present in the nucleus are called nucleons.
Mass number (A) = Number of protons (Z) + Number of neutrons (n)
Since the mass of a neutron is roughly equal to that of a proton, the mass of a helium atom can be accounted for by two protons and two neutrons.
The electron has almost negligible mass and hence can be ignored in calculations.
Table: Mass Number of Different Elements
| Element | Protons (p+) | Neutrons (n0) | Mass Number (A) |
|---|---|---|---|
| Hydrogen | 1 | 0 | 1 |
| Helium | 2 | 2 | 4 |
| Lithium | 3 | 4 | 7 |
Standard notation for an atom:The symbol, atomic number (Z) and mass number (A) are written as - Mass Number on top left, Atomic Number on bottom left, followed by the Symbol of the element.
For example, for carbon (C), atomic number = 6, mass number = 12, it is written as ¹²₆C
7. How Are Electrons Distributed in Different Energy Levels?
Bohr and Bury suggested the following rules for the distribution of electrons in shells:
- The maximum number of electrons present in a shell is given by the formula 2n², where 'n' is the number of the shell.
- K-shell (n = 1): 2 x 1² = 2 electrons maximum
- L-shell (n = 2): 2 x 2² = 8 electrons maximum
- M-shell (n = 3): 2 x 3² = 18 electrons maximum
- The maximum number of electrons that can be accommodated in the outermost shell is 8 (the first shell can accommodate a maximum of two electrons).
- Electrons are filled in these shells in a stepwise manner, starting from the one closest to the nucleus and moving outward, i.e., in the order K, L, M, N, ... The L-shell will be filled only after the K-shell is complete, and so on.
The distribution of electrons among various shells is known as the electronic configuration of the atom.
7.1 Building Up Atoms
Two-dimensional atomic structures for elements are created by adding one electron to the appropriate energy level each time atomic number is increased by 1. Schematic atomic structure of first 18 elements
Schematic atomic structure of first 18 elements
Table : Electronic Configuration of the First 18 Elements
| Element | Symbol | Atomic No. | Protons | Neutrons | Electrons | K | L | M |
|---|---|---|---|---|---|---|---|---|
| Hydrogen | H | 1 | 1 | - | 1 | 1 | - | - |
| Helium | He | 2 | 2 | 2 | 2 | 2 | - | - |
| Lithium | Li | 3 | 3 | 4 | 3 | 2 | 1 | - |
| Beryllium | Be | 4 | 4 | 5 | 4 | 2 | 2 | - |
| Boron | B | 5 | 5 | 6 | 5 | 2 | 3 | - |
| Carbon | C | 6 | 6 | 6 | 6 | 2 | 4 | - |
| Nitrogen | N | 7 | 7 | 7 | 7 | 2 | 5 | - |
| Oxygen | O | 8 | 8 | 8 | 8 | 2 | 6 | - |
| Fluorine | F | 9 | 9 | 10 | 9 | 2 | 7 | - |
| Neon | Ne | 10 | 10 | 10 | 10 | 2 | 8 | - |
| Sodium | Na | 11 | 11 | 12 | 11 | 2 | 8 | 1 |
| Magnesium | Mg | 12 | 12 | 12 | 12 | 2 | 8 | 2 |
| Aluminium | Al | 13 | 13 | 14 | 13 | 2 | 8 | 3 |
| Silicon | Si | 14 | 14 | 14 | 14 | 2 | 8 | 4 |
| Phosphorus | P | 15 | 15 | 16 | 15 | 2 | 8 | 5 |
| Sulfur | S | 16 | 16 | 16 | 16 | 2 | 8 | 6 |
| Chlorine | Cl | 17 | 17 | 18 | 17 | 2 | 8 | 7 |
| Argon | Ar | 18 | 18 | 22 | 18 | 2 | 8 | 8 |
8. Combining Capacity of an Atom: Valency
Atoms of the same or different elements can combine to form molecules. The number of atoms of hydrogen or chlorine with which one atom of an element can combine to form a compound is called its combining capacity. It is expressed in terms of hydrogen and chlorine because both possess a combining capacity of one.
Example: In H2O (water), oxygen combines with two hydrogen atoms, so the combining capacity of oxygen is two.
The combining capacity of an atom is decided by its electronic configuration.
Valence Shell and Valence Electrons:
- The outermost shell containing electrons of an atom is known as its valence shell.
- The electrons present in the valence shell are known as valence electrons.
Octet:
- If the outermost shell of an atom has 8 electrons, it is called an octet.
- Elements with complete octet of electrons (8 electrons), or 2 electrons in the case of helium, in their valence shell are largely unreactive and more stable.
- Atoms with incomplete valence shells are usually more reactive. Such elements lose, gain, or share electrons to complete their octet.
Valency:
- The number of electrons gained, lost, or shared to complete the octet is called the valency of the element.
- If the element has fewer than four electrons in its valence shell, it tends to lose electrons to complete its octet and become stable.
- If the number of valence electrons is more than four, it tends to gain electrons to complete its octet.
Examples:
- Sodium: electronic configuration = 2, 8, 1. It can get an octet by losing one electron. Therefore, its valency = 1.
- Oxygen: electronic configuration = 2, 6. It has six valence electrons. It can gain two electrons to attain an octet. Therefore, its valency = 2.
- Carbon: electronic configuration = 2, 4. Carbon has four valence electrons and cannot easily gain or lose them. Therefore, it can share four electrons with other atoms to complete its octet. Thus, the valency of carbon = 4.
Note: Atoms that already have eight electrons in their outermost shell (like neon, argon) or two electrons (like helium) do not need to lose or gain electrons. Their valency is 0 (zero). They are called noble gases and are largely unreactive.
9. A Deeper Look into Atomic Structure
9.1 Isotopes
Dalton proposed that all atoms of an element are identical and have the same mass. But scientists later discovered that atoms of the same element can have the same number of protons (atomic number, Z) yet can have different numbers of neutrons, and thus different mass numbers (A = p+ + n0).
These 'twin atoms' with the same atomic number but different mass numbers are called isotopes.
Example - Isotopes of Hydrogen:Naturally occurring hydrogen is a mixture of three different isotopes:
- Protium ¹₁H : ~99.98% - 1 proton, 0 neutrons, 1 electron
- Deuterium ²₁H: ~0.015% - 1 proton, 1 neutron, 1 electron
- Tritium ³₁H : in traces - 1 proton, 2 neutrons, 1 electron
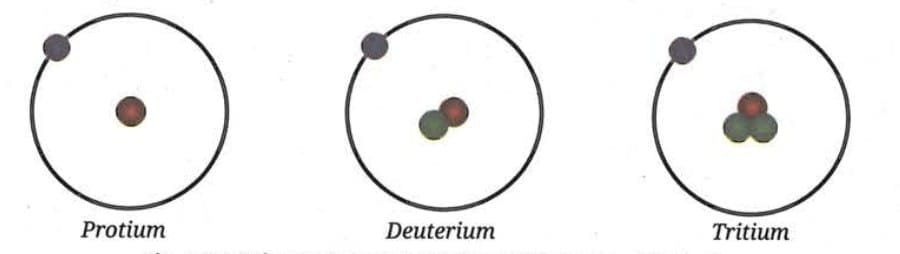 All three isotopes of hydrogen have one proton each.
All three isotopes of hydrogen have one proton each.
Example - Isotopes of Carbon:
Carbon has three isotopes: ¹²₆C, ¹³₆C, and ¹⁴₆C
- Each of these has six protons and six electrons.
- ¹²₆C is the most abundant isotope in nature.
- These three isotopes differ in terms of the number of neutrons.
Chemical Properties of Isotopes:
- The chemical properties of isotopes are similar because they have the same number of electrons and the same electronic configuration.
- Chemical properties depend mainly on the number of valence electrons.
- All isotopes of an element will have the same chemical properties although they differ in their physical properties (for example, boiling and melting points).
Applications of Isotopes:
- ²³⁵₉₂U (an isotope of uranium) is used as fuel in a nuclear reactor to generate electricity in a nuclear power plant.

- ⁶⁰₂₇Co (a radioactive isotope of cobalt) is used in radiation treatment for cancer.
- ¹³¹₅₃I (an isotope of iodine) is used to treat goitre and thyroid cancer.
- ¹⁴₆C (an isotope of carbon) is used in archaeology and geology to determine the age of ancient fossils and artefacts.
Ready to Go Beyond
Atoms are extremely small, so their masses cannot be measured in grams or kilograms. Scientists use a special unit called the unified atomic mass unit (u) to measure atomic mass. Earlier, this unit was called atomic mass unit (amu).
A. Average Atomic Mass
Chlorine occurs in nature in two isotopic forms. One isotope has a mass of 35 u, and the other has a mass of 37 u. They occur in the ratio 3:1.
Simple Average: Average atomic mass = (35 + 37) / 2 = 36 u
However, this does not accurately reflect nature, since isotopes do not occur in equal ratios. The more common isotope is 35Cl, which constitutes about 75%, while 37Cl makes up about 25%.
Weighted Average Atomic Mass:The accurate average atomic mass can be calculated by considering their natural abundances. This is called the weighted average atomic mass, calculated by multiplying the mass of each isotope by its percent relative abundance and then adding the values.
= (35 x 75/100) + (37 x 25/100) = (105/4) + (37/4) = 142/4 = 35.5 u
This means that if you take 1 million chlorine atoms, they will contain 7.5 lakh 35Cl and 2.5 lakh 37Cl atoms with a weighted average atomic mass of 35.5 u.
The weighted average accurately reflects the mass of the element as it occurs in nature, whereas the simple average ignores abundance.
Meet a Scientist
Homi Jehangir Bhabha was an Indian physicist known as the father of the Indian nuclear programme. He made major contributions to atomic energy in India and established institutions like Tata Institute of Fundamental Research and Bhabha Atomic Research Centre. His work promoted the peaceful use of atomic energy in electricity, agriculture, and medicine.
Ready to Go Beyond
Electron microscopes can show materials at the atomic level. Scanning Tunnelling Microscopes (STMs) are used to study surface details, while Transmission Electron Microscopes (TEMs) reveal the arrangement of atoms inside thin samples.
9.2 Isobars
Consider three elements - calcium (atomic number 20), potassium (atomic number 19), and argon (atomic number 18). These elements have different numbers of protons, yet each has a mass number of 40. This shows that the total number of nucleons in their atoms is the same, even though they are different elements.
When atoms of different elements have the same mass number, but different atomic numbers, they are called isobars.
Example: ⁴⁰₂₀Ca, ⁴⁰₁₉K, and ⁴⁰₁₈Ar are all isobars (same mass number 40, but different atomic numbers).
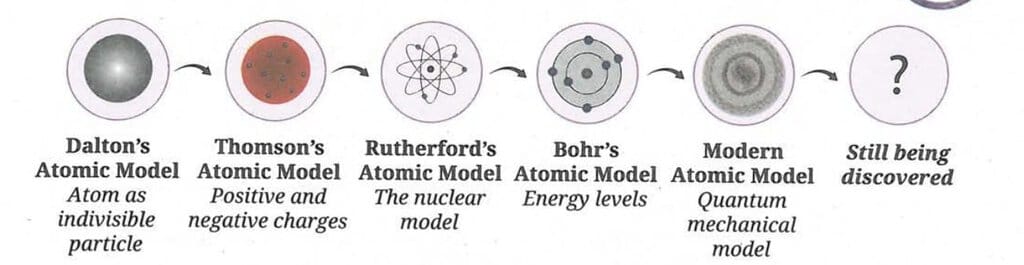
Development of Atomic Models - Summary
The journey of atomic models evolved as follows
- Dalton's Atomic Model: Atom as indivisible particle.
- Thomson's Atomic Model: Positive and negative charges (plum pudding model).
- Rutherford's Atomic Model: The nuclear model (dense nucleus at centre).
- Bohr's Atomic Model: Energy levels (electrons in fixed orbits/shells).
- Modern Atomic Model: Quantum mechanical model (electrons as electron clouds).
- Still being discovered.
Key Terms to Remember
| Term | Meaning |
|---|---|
| Atom | The smallest unit of an element that retains its chemical properties |
| Parmanu | Ancient Indian concept for the smallest indivisible particle of matter (by Acharya Kanada) |
| Atomos | Greek word meaning indivisible; the origin of the word 'atom' |
| Radioactivity | Emission of invisible energy and particles from certain elements |
| Electron | Negatively charged subatomic particle; charge = -1; negligible mass |
| Cathode rays | Streams of negatively charged particles (electrons) moving from cathode to anode |
| Proton | Positively charged subatomic particle; charge = +1; found in nucleus |
| Neutron | Neutral subatomic particle; no charge; mass nearly equal to proton; found in nucleus |
| Nucleus | Dense, positively charged central core of an atom containing protons and neutrons |
| Nucleons | Protons and neutrons collectively |
| Plum Pudding Model | Thomson's model - atom as sphere of positive charge with electrons embedded in it |
| Planetary Model | Rutherford's model - electrons revolve around a central nucleus like planets around the Sun |
| Alpha (α) particles | Tiny, positively charged particles emitted from radioactive elements; nucleus of helium atom |
| Scattering | Deflection of alpha particles from their straight path in the gold foil experiment |
| Shell / Orbit | Fixed circular paths followed by electrons around the nucleus (Bohr's model) |
| Energy levels | Different shells representing different definite energies for electrons |
| Stationary states | Fixed shells/orbits in which electrons revolve without losing energy (Bohr's concept) |
| Atomic number (Z) | Number of protons in the nucleus of an atom |
| Mass number (A) | Total number of protons and neutrons (nucleons) in the nucleus |
| Electronic configuration | Distribution of electrons among various shells of an atom |
| Valence shell | The outermost shell of an atom |
| Valence electrons | Electrons present in the valence (outermost) shell |
| Octet | A condition where an atom has 8 electrons in its outermost shell |
| Valency | The combining capacity of an atom - number of electrons gained, lost, or shared to complete the octet |
| Isotopes | Atoms of the same element with same atomic number but different mass numbers |
| Isobars | Atoms of different elements with same mass number but different atomic numbers |
| Weighted average atomic mass | Average mass of an element calculated by considering the relative abundances of its isotopes |
| IUPAC | International Union of Pure and Applied Chemistry - body that approves names and symbols of elements |
| Unified atomic mass unit (u) | Unit used to measure the mass of atoms |
FAQs on Chapter Notes: Journey Inside the Atom
| 1. What is atomic theory and why is it important? |  |
| 2. Can you explain the historical development of atomic models? |  |
| 3. What are the components that contribute to the mass of an atom? |  |
| 4. How is the atomic number defined and what does it signify? |  |
| 5. What is valency and how does it relate to an atom's combining capacity? |  |







