NCERT Solutions: Journey Inside the Atom
Think It Over - Page 14o
Q1: Are atoms the smallest indivisible particles?
Ans: No, atoms are not the smallest indivisible particles. They are made up of smaller subatomic particles such as electrons, protons, and neutrons.
Q2: Why do electrons not fall into the nucleus even though they are attracted to protons in it?
Ans: Electrons revolve around the nucleus in fixed energy levels (shells). In these stable orbits, they do not lose energy, so they do not fall into the nucleus.
Q3: Why did scientists keep modifying atomic models?
Ans: Scientists kept modifying atomic models because new experiments and discoveries revealed limitations in earlier models. To better explain the structure of the atom and experimental observations, improved models were proposed.
Pause and Ponder - Page 143
Q 1. Suppose you made up your own 'atom', as Thomson described, using clay for the positive charge and small beads for the electrons spread through it. What will happen if:
(i) the positive charge on the clay is lesser than the total negative charge of the beads?
Ans: The atom would not be electrically neutral. It would carry a net negative charge. This would not represent a real neutral atom since in a real atom, the total positive charge always equals the total negative charge, making the atom electrically neutral.
(ii) by mistake, the clay itself carries a bit of negative charge? Would your model still represent a neutral atom?
Ans: No, the model would not represent a neutral atom. If the clay (which is supposed to represent the positive sphere) carries a negative charge, then both the clay and the beads (electrons) would be negatively charged. The total charge of the model would be entirely negative. There would be no positive charge to balance the negative charge of the electrons, so the model would not represent a neutral atom.
Q 2. Could an orange or a lemon, which also contain seeds inside soft pulp, be a good comparison? In what ways does it match Thomson's idea and where does it fall short?
Ans: An orange or lemon can be used as a rough comparison to Thomson's model in the following ways:
Matches Thomson's idea:
- The soft pulp (like the positive sphere in Thomson's model) fills the interior.
- The seeds (like electrons) are embedded within the pulp.
- The object has a roughly spherical shape, similar to an atom.
Falls short:
- In Thomson's model, electrons are uniformly and evenly distributed throughout the positive sphere. But in an orange or lemon, the seeds are not uniformly distributed - they are clustered together in certain sections (compartments).
- The pulp of an orange or lemon is not uniformly positive - it is just neutral fruit pulp, not a positively charged sphere.
- So while it gives a visual idea, it is not a perfect or accurate representation.
Q 3. Why did Thomson conclude that electrons are present in all atoms?
Ans: Thomson found that cathode rays (streams of electrons) were produced regardless of the material used for the cathode or the type of gas filled in the cathode ray tube. Since the same negatively charged particles (electrons) were emitted from any material under any condition, Thomson concluded that electrons are a fundamental and common component present in all atoms of all elements.
Pause and Ponder - Page 144
Q 4. What do you think would happen if alpha-particles were replaced with negatively charged particles in Rutherford's gold foil experiment?
Ans: If negatively charged particles were used instead of positively charged alpha particles, the result would be very different. The nucleus of gold atoms is positively charged. A negatively charged particle approaching the nucleus would be attracted towards the nucleus (unlike alpha particles which are repelled). So instead of bouncing back or being deflected at large angles (which happens due to repulsion between like charges), the negatively charged particles would be pulled inward towards the nucleus. Most of them would curve towards the nucleus rather than pass straight through or bounce back. The scattering pattern would be completely different.
Q 5. Rutherford found that a few alpha-particles bounced back sharply. How does this single surprising result completely rule out Thomson's 'plum pudding model' of the atom?
Ans: In Thomson's plum pudding model, the positive charge is spread uniformly throughout the entire volume of the atom like a diffuse sphere. If this were true, the positive charge at any point would be extremely weak and spread out. An alpha particle passing through such an atom would never encounter a concentrated or strong enough repulsive force to bounce it back. Even if slightly deflected, the particle could not be deflected at large angles, and certainly could not bounce straight back.
The fact that a few alpha particles did bounce back sharply clearly means there must be a very small, extremely dense, and very strongly positively charged region inside the atom - the nucleus. Only a concentrated mass of positive charge could repel the fast-moving alpha particles so strongly. This single surprising observation completely rules out the plum pudding model and proves that positive charge cannot be spread out uniformly throughout the atom.
Q 6. If you could ask Rutherford one question about his work, what would it be?
Ans:
A thoughtful question could be: "If the nucleus is so small and yet contains all the positive charge and most of the mass of the atom, what force is responsible for holding the positively charged protons together inside the nucleus without them flying apart due to their mutual repulsion?"
This question relates directly to the limitation of Rutherford's model and the concept of nuclear force, which is relevant to the chapter.
Pause and Ponder - Page 145
Q 7. Assertion (A): Rutherford concluded that most of the mass of an atom is concentrated in a small region at the centre called the nucleus.
Reason (R): According to Thomson's model, electrons are embedded in a uniformly distributed positive charge sphere. Choose the correct option:
(i) Both A and R are true, and R is the correct explanation of A.
(ii) Both A and R are true, but R is not the correct explanation of A.
(iii) A is true, but R is false.
(iv) A is false, but R is true.
Ans: Option (iii): A is true, but R is false.
Explanation: The Assertion (A) is true - Rutherford did conclude from the gold foil experiment that most of the mass of an atom is concentrated in a small, dense region called the nucleus. This was one of his key conclusions. However, the Reason (R) is false - it describes Thomson's model (where electrons are embedded in a uniformly distributed positive charge sphere), which is actually the opposite of what Rutherford proposed. Thomson's model was the model that Rutherford disproved. So the reason does not explain the assertion, and in fact describes the model that the assertion's evidence argues against.
What if - Page 147
Q: What if an atom had no empty space? How would this have affected the size of various objects?
Ans: If atoms had no empty space, their size would be much smaller. Since most of the volume of matter is due to empty space in atoms, removing it would make objects extremely small and compact. All substances would shrink drastically in size.
Pause and Ponder - Page 149
Q 8. Imagine you are a scientist who has discovered a new element. Name this element after yourself and justify that the symbol you have chosen follows the IUPAC rules.
Ans:
For example: Suppose the student's name is "Aryan". The element could be named "Aryanium" with the symbol Ay.
Justification:
- The first letter 'A' is written as a capital (uppercase) letter - follows IUPAC rule.
- The second letter 'y' is written as a small (lowercase) letter - follows IUPAC rule.
- The symbol uses the first two letters of the element's name - follows IUPAC rule.
- The symbol is a unique combination that does not conflict with any existing element symbols.
Q 9. What problems could arise if every scientist used different symbols for the same element?
Ans: If every scientist used different symbols for the same element, the following problems would arise:
- There would be great confusion in scientific communication worldwide. Scientists from different countries could not understand each other's research papers or findings.
- Chemical equations and formulas would be inconsistent and completely unreadable to others.
- Errors would occur in chemical experiments, industrial applications, and medicine, which could be dangerous.
- International scientific collaboration and the sharing of knowledge would become impossible.
- The same element could be represented by hundreds of different symbols, making it impossible to maintain a universal language of science.
- It would slow down scientific progress and create chaos in science education globally.
Pause and Ponder - Page 150
Q 10. An atom with an atomic number of 26 has 56 nucleons. Find out its number of electrons, protons and neutrons.
Ans:
- Atomic number (Z) = 26, so number of protons = 26.
- Since the atom is electrically neutral, number of electrons = number of protons = 26.
- Number of nucleons (mass number A) = 56.
- Number of neutrons = A - Z = 56 - 26 = 30.
- Answer: Protons = 26, Electrons = 26, Neutrons = 30. (This element is Iron, Fe.)
Q 11. The nucleus of an atom contains 20 protons. If its mass number is 41, find the number of neutrons in it.
Ans:
- Number of protons = 20, Mass number (A) = 41.
- Number of neutrons = A - number of protons = 41 - 20 = 21.
- Answer: The atom has 21 neutrons.
Q 12. An atom has 18 neutrons and an atomic number of 17. What is its mass number?
Ans:
- Number of neutrons = 18, Atomic number (protons) = 17.
- Mass number (A) = number of protons + number of neutrons = 17 + 18 = 35.
- Answer: The mass number is 35. (This element is Chlorine, Cl.)
Q 13. An atom 23A has 11 electrons. Find the number of neutrons in it.
Ans:
- Mass number (A) = 23, number of electrons = 11.
- Since the atom is neutral, number of protons = number of electrons = 11.
- Number of neutrons = mass number - number of protons = 23 - 11 = 12.
- Answer: The atom has 12 neutrons. (This element is Sodium, Na.)
Pause and Ponder - Page 152
Q 14. Identify the number of electrons in the outermost shell of the following elements:
(i)¹²₆C
Ans: Carbon has atomic number = 6. Electronic configuration = 2, 4. The outermost shell is the L-shell (n = 2) and it contains 4 electrons.
(ii) ¹⁹₉F
Ans: Fluorine has atomic number = 9. Electronic configuration = 2, 7. The outermost shell is the L-shell and it contains 7 electrons.
(iii) ²⁸₁₄Si
Ans: Silicon has atomic number = 14. Electronic configuration = 2, 8, 4. The outermost shell is the M-shell and it contains 4 electrons.
Q 15. Write the electronic configuration of the elements having atomic numbers 12, 16 and 18.
Ans:
- Atomic number 12 (Magnesium, Mg): Electronic configuration = 2, 8, 2. (K = 2, L = 8, M = 2)
- Atomic number 16 (Sulfur, S): Electronic configuration = 2, 8, 6. (K = 2, L = 8, M = 6)
- Atomic number 18 (Argon, Ar): Electronic configuration = 2, 8, 8. (K = 2, L = 8, M = 8)
Q 16. Solve this riddle: I am an atom with a mass number of 23 and 11 protons. I am a soft metal and react vigorously with water. Who am I and how many neutrons do I have? You can also create one such riddle.
Ans:
- Mass number = 23, number of protons (atomic number) = 11.
- Number of neutrons = 23 - 11 = 12.
- The element with atomic number 11 is Sodium (Na).
- Sodium is indeed a soft metal that reacts vigorously with water.
- Answer: I am Sodium (Na) and I have 12 neutrons.
Pause and Ponder - Page 156
Q 17. Two different atoms have 11 protons each, but one has 12 neutrons, and the other has 13 neutrons. How do their atomic numbers and mass numbers compare? Are they the same element or different elements?
Ans:
- Both atoms have 11 protons, so both have atomic number = 11.
- Since the atomic number is the same, both atoms belong to the same element - Sodium (Na).
- Mass number of first atom = 11 + 12 = 23.
- Mass number of second atom = 11 + 13 = 24.
- So their atomic numbers are the same but their mass numbers are different.
- These two atoms are isotopes of sodium - they are the same element but with different mass numbers.
Q 18. If a bromine atom is available in the form of, say two isotopes, ⁷⁹₃₅Br (49.7%) and ⁸¹₃₅Br (50.3%), calculate the average atomic mass of the bromine atom.
Ans: Weighted average atomic mass = (mass of first isotope x its % abundance/100) + (mass of second isotope x its % abundance/100)
= (79 x 49.7/100) + (81 x 50.3/100) = (79 x 0.497) + (81 x 0.503) = 39.263 + 40.743 = 80.006 u ≈ 80 u
Answer: The average atomic mass of bromine is approximately 80 u.
Revise, Reflect, Refine
Q 1. Choose the correct options and explain the reason for the correct and incorrect options in the context of Ernest Rutherford's gold foil experiment:
(i) The experiment clearly showed the existence of neutrons in the nucleus.
(ii) The results disproved the plum pudding model and led to the idea of a nucleus at the centre of the atom.
(iii) The large deflection of a few alpha particles indicated that most of the mass of the atom and positive charge are packed into a tiny centre.
(iv) The way alpha particles were deflected showed that electrons move around the nucleus.
Ans:
(i) INCORRECT. The gold foil experiment did not show the existence of neutrons. Neutrons were discovered by James Chadwick in 1932, much later, through a completely different experiment. The gold foil experiment only revealed the existence and nature of the nucleus (positively charged and dense).
(ii) CORRECT. The gold foil experiment's results - that most alpha particles passed through undeflected but some were sharply deflected and a few bounced back - could not be explained by Thomson's plum pudding model (where positive charge is spread out). These results disproved the plum pudding model and led Rutherford to propose the idea of a small, dense, positively charged nucleus at the centre of the atom.
(iii) CORRECT. A few alpha particles bounced back sharply, which is only possible if they encountered a very small but extremely dense and strongly positive region. If the positive charge were spread out (as in Thomson's model), no alpha particle would bounce back. The large deflection of a few alpha particles confirmed that most of the mass and all the positive charge are packed into a tiny centre called the nucleus.
(iv) INCORRECT. The gold foil experiment did not show or study the movement of electrons around the nucleus. It only revealed information about the nucleus - its size, charge, and density. The movement of electrons in orbits around the nucleus was proposed by Rutherford's model as a logical conclusion, but the experiment itself did not directly demonstrate this.
Q 2. Which of the following statements are correct or incorrect according to the Bohr's atomic model? Give a reason for each statement.
(i) Electrons lose energy while moving in fixed orbits and slowly fall into the nucleus.
(ii) Electrons can exist anywhere around the nucleus with no fixed energy.
(iii) Electrons revolve around the nucleus in orbits of fixed energy without losing energy.
(iv) Electrons can be found between energy levels as they move around the nucleus.
Ans:
(i) INCORRECT. According to Bohr's model, electrons do NOT lose energy while moving in fixed orbits. The key feature of Bohr's model is the concept of stationary states - when an electron is in a fixed orbit (shell), its energy remains constant and it does not lose energy. Therefore, electrons do not spiral inward or fall into the nucleus.
(ii) INCORRECT. According to Bohr's model, electrons cannot exist anywhere around the nucleus freely. They must follow fixed circular paths called orbits or shells (K, L, M, N, ...). Each shell has a definite, fixed amount of energy. Electrons must be in one of these fixed energy levels; they cannot float freely with no fixed energy.
(iii) CORRECT. This statement correctly describes Bohr's model. Electrons revolve around the nucleus in fixed circular orbits (stationary states). While revolving in these fixed orbits, electrons do not lose energy. Their energy remains constant in a given orbit. This is the reason Bohr's model could explain atomic stability, unlike Rutherford's model.
(iv) INCORRECT. According to Bohr's model, electrons cannot be found between energy levels. They can only exist in the fixed shells (K, L, M, N, ...). An electron can move from one shell to another by absorbing or releasing a fixed amount of energy, but it cannot stay in the space between two energy levels. The moment energy is absorbed or released, the electron jumps directly from one level to another.
Q 3. The composition of the nuclei of three atomic species X, Y, and Z are given as follows: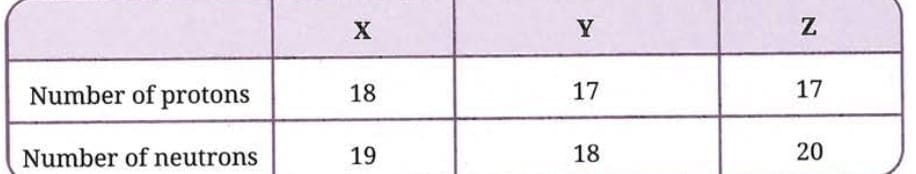
Explain the relation between the following:(i) Y and Z(ii) Z and X
Ans:
First, let us find the mass numbers:
- X: protons = 18, neutrons = 19, mass number = 18 + 19 = 37
- Y: protons = 17, neutrons = 18, mass number = 17 + 18 = 35
- Z: protons = 17, neutrons = 20, mass number = 17 + 20 = 37
(i) Y and Z: Both Y and Z have the same number of protons (atomic number = 17), which means they belong to the same element - Chlorine (Cl). However, they have different mass numbers (Y = 35, Z = 37). Therefore, Y and Z are isotopes of each other. They are atoms of the same element with the same atomic number but different mass numbers.
(ii) Z and X: Z has atomic number = 17 (Chlorine) and X has atomic number = 18 (Argon). They are different elements. However, both Z and X have the same mass number = 37. Atoms of different elements that have the same mass number but different atomic numbers are called isobars. Therefore, Z and X are isobars.
Q 4. What conclusion did Rutherford draw about the position and characteristics of the atom's positively charged part based on the few alpha particles that bounced back or were deflected at large angles in the gold foil experiment?
Ans: Based on the observation that a few alpha particles bounced back or were deflected at large angles, Rutherford drew the following conclusions about the positively charged part of the atom:
- The positive charge of the atom is not spread throughout the volume of the atom (as Thomson suggested) but is concentrated in an extremely small region at the centre.
- This small, central, positively charged region is called the nucleus.
- The nucleus is extremely dense - it contains most of the mass of the atom packed into a very tiny volume.
- The nucleus is very small compared to the overall size of the atom. The diameter of the atom is approximately 10^-10 m, while the diameter of the nucleus is approximately 10^-15 m - the nucleus is about 10^5 (one lakh) times smaller than the atom.
- Since most of the alpha particles passed straight through without deflection, most of an atom is empty space.
- Only when an alpha particle came very close to the nucleus was it repelled strongly enough to bounce back or deflect at a large angle.
Q 5. Explain and arrange the following statements in the correct chronological order to show how atomic models have evolved over time.
(i) Bohr's model proposed that electrons move in fixed orbits around the nucleus, each with a definite energy.
(ii) Thomson's model depicted the atom as a 'plum pudding' with electrons embedded in a sphere of positive charge.
(iii) Rutherford's model proposed that atoms have a dense central nucleus.
(iv) Dalton's model described atoms as indivisible particles.
Ans:
Correct chronological order and explanations:
- (iv) Dalton's model (1808): Dalton described atoms as indivisible particles - the fundamental building blocks of matter that cannot be broken down further. This was the first scientific atomic theory, based on experiments of that time.
- (ii) Thomson's model (1897 onwards): After the discovery of the electron by J. J. Thomson, he proposed that the atom is a sphere of positive charge with electrons embedded throughout it (plum pudding model). This was the first model to acknowledge that atoms contain smaller charged particles.
- (iii) Rutherford's model (1911): After the gold foil experiment, Rutherford proposed the planetary model - the atom has a small, dense, positively charged nucleus at its centre, with electrons revolving around it like planets around the Sun. This disproved Thomson's model.
- (i) Bohr's model (1913): To explain why electrons do not lose energy and fall into the nucleus (a limitation of Rutherford's model), Niels Bohr proposed that electrons move in fixed circular orbits (shells) with definite energies, called stationary states. Electrons do not lose energy while in these fixed orbits.
So the chronological order is: (iv) → (ii) → (iii) → (i)
Q 6. Electrons move around the nucleus in orbits. Why do they not fly away from the atom? Explain what keeps them attracted to the nucleus.
Ans: Electrons do not fly away from the atom because they are held in place by the electrostatic force of attraction between the negatively charged electrons and the positively charged protons in the nucleus. This attractive force acts like the force that keeps a planet in orbit around the Sun - it continuously pulls the electron towards the nucleus, preventing it from flying away in a straight line.
According to Bohr's model, electrons revolve in fixed circular orbits (stationary states). In these fixed orbits, the centripetal force needed to keep the electron moving in a circle is provided by the electrostatic attraction between the electron and the nucleus. As long as the electron remains in its allowed orbit, it does not lose energy and does not fly away or fall inward.
Thus, the attractive electrostatic force between the oppositely charged electron and nucleus is what keeps the electron orbiting and prevents it from flying away.
Q 7. Assertion (A): The discovery of subatomic particles helped in understanding the atomic structure.
Reason (R): The number of electrons is equal to the number of protons in an atom. Choose the correct option:
(i) Both A and R are true, and R is the correct explanation of A.
(ii) Both A and R are true, but R is not the correct explanation of A.
(iii) A is true, but R is false.
(iv) A is false, but R is true.
Ans: Option (ii): Both A and R are true, but R is not the correct explanation of A.
Explanation:
- The Assertion (A) is true. The discovery of subatomic particles (electrons by Thomson, protons and nucleus by Rutherford, neutrons by Chadwick) was crucial in helping scientists understand the structure of the atom. Each discovery led to a better and more accurate model of the atom.
- The Reason (R) is also true. In a neutral atom, the number of electrons is always equal to the number of protons. This is a fundamental fact that ensures atoms are electrically neutral.
- However, R is not the correct explanation of A. The equal number of electrons and protons is a consequence (result) of atomic neutrality - it tells us something about the balance of charge in an atom. It does not explain how or why the discovery of subatomic particles helped understand atomic structure. The discovery of subatomic particles helped understand atomic structure by revealing what atoms are made of, how their mass is distributed, and how they are arranged - not merely because electrons equal protons.
Q 8. Magnesium is essential for many biological processes, including muscle contraction. For an atom of magnesium with a mass number of 24 and atomic number 12, determine the number of (i) protons, (ii) neutrons, (iii) electrons, and also illustrate the arrangement of electrons in a magnesium atom
Ans:
- Mass number (A) = 24, Atomic number (Z) = 12.
(i) Number of protons = Atomic number = 12.
(ii) Number of neutrons = Mass number - Atomic number = 24 - 12 = 12.
(iii) Number of electrons = Number of protons (atom is neutral) = 12.
Arrangement of electrons in Magnesium (Electronic Configuration):
- K-shell (n = 1): 2 electrons (maximum = 2, filled first)
- L-shell (n = 2): 8 electrons (maximum = 8, filled next)
- M-shell (n = 3): 2 electrons (remaining electrons)
- Electronic configuration: 2, 8, 2
Q 9. Find the following information for the elements shown in Fig. 8.17:
(i) Name of the element
(ii) Symbol
(iii) Total number of electrons
(iv) Number of valence electrons
(v) Valency of the element
(vi) Number of protons
(vii) Atomic number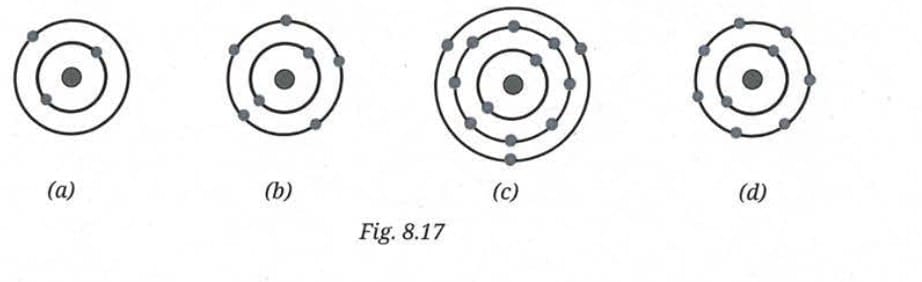
Ans:
- (a): 1 shell with 2 electrons
- (b): 2 shells - K=2, L=8
- (c): 3 shells - K=2, L=8, M=5 (with an extra electron shown, total = 15)
- (d): 2 shells - K=2, L=7
(a) Atom with 2 electrons (K=2):
- (i) Name: Helium
- (ii) Symbol: He
- (iii) Total number of electrons: 2
- (iv) Number of valence electrons: 2 (only shell is K-shell)
- (v) Valency: 0 (outermost shell is completely filled with 2 electrons, which is the maximum for K-shell - stable)
- (vi) Number of protons: 2
- (vii) Atomic number: 2
(b) Atom with K=2, L=8 - total 10 electrons:
- (i) Name: Neon
- (ii) Symbol: Ne
- (iii) Total number of electrons: 10
- (iv) Number of valence electrons: 8 (L-shell is the outermost shell)
- (v) Valency: 0 (outermost shell has 8 electrons - complete octet, stable)
- (vi) Number of protons: 10
- (vii) Atomic number: 10
(c) Atom with K=2, L=8, M=5 - total 15 electrons:
- (i) Name: Phosphorus
- (ii) Symbol: P
- (iii) Total number of electrons: 15
- (iv) Number of valence electrons: 5 (M-shell is the outermost shell)
- (v) Valency: 3 (needs to gain 3 electrons to complete octet; since 5 > 4, it tends to gain electrons: 8 - 5 = 3)
- (vi) Number of protons: 15
- (vii) Atomic number: 15
(d) Atom with K=2, L=7 - total 9 electrons:
- (i) Name: Fluorine
- (ii) Symbol: F
- (iii) Total number of electrons: 9
- (iv) Number of valence electrons: 7 (L-shell is the outermost shell)
- (v) Valency: 1 (needs to gain 1 electron to complete octet: 8 - 7 = 1)
- (vi) Number of protons: 9
- (vii) Atomic number: 9
Q 10. Both Rutherford's and Bohr's models have electrons orbiting the nucleus. Why did Rutherford's model fail to explain atomic stability, while Bohr's model succeeded?
Ans: The key difference lies in how each model describes the electron's motion:
Rutherford's model and why it failed:
- In Rutherford's model, electrons orbit the nucleus freely, similar to planets around the Sun.
- However, according to the laws of physics (as understood from Chapter 4 - describing circular motion), a particle moving in a circular path is constantly changing direction, which means it is accelerating.
- A negatively charged particle that is accelerating should continuously emit energy (in the form of radiation).
- If electrons kept losing energy, they would slow down, spiral inward toward the nucleus, and eventually fall into it.
- If this happened, atoms would collapse in a very short time and would not exist in a stable form.
- Rutherford's model had no explanation for why this does not happen. It could not explain atomic stability.
Bohr's model and why it succeeded:
- Bohr introduced the concept of stationary states (fixed orbits/shells).
- He proposed that when an electron is in a fixed allowed orbit, its energy remains constant - it does not emit or lose energy while in that orbit.
- This was a postulate (an assumed rule) that Bohr introduced specifically to address Rutherford's limitation.
- By placing electrons in fixed energy levels where energy is constant, Bohr's model successfully explained why electrons do not spiral inward and why atoms are stable.
- Bohr's model could also explain many experimental observations, like the spectrum of hydrogen.
Q 11. An atom 70X has 31 electrons. How many neutrons are there in its nucleus?
Ans:
- Mass number (A) = 70 (given as superscript, so the atom is 70X).
- Number of electrons = 31.
- Since the atom is neutral, number of protons = number of electrons = 31. So atomic number (Z) = 31.
- Number of neutrons = Mass number - Atomic number = 70 - 31 = 39.
- Answer: There are 39 neutrons in its nucleus. (This element is Gallium, Ga.)
Q 12. An atom has 79 protons and a mass number of 197. Calculate (i) the number of neutrons, and (ii) the number of electrons.
Ans:
- Number of protons (atomic number Z) = 79.
- Mass number (A) = 197.
(i) Number of neutrons = Mass number - Number of protons = 197 - 79 = 118.
(ii) Since the atom is electrically neutral, number of electrons = number of protons = 79.
Answer: (i) Number of neutrons = 118. (ii) Number of electrons = 79. (This element is Gold, Au.)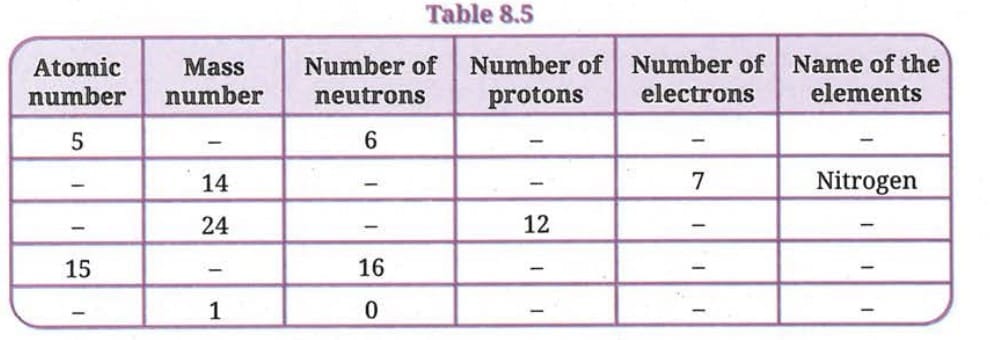
Q 13. Complete the Table 8.5:
Given data and solutions:
Row 1: Atomic number = 5, neutrons = 6
- Number of protons = Atomic number = 5
- Mass number = 5 + 6 = 11
- Number of electrons = 5 (neutral atom)
- Name = Boron (B)
Row 2: Mass number = 14, electrons = 7, Name = Nitrogen
- Since it is Nitrogen, atomic number = 7, so protons = 7
- Number of neutrons = 14 - 7 = 7
- Number of electrons = 7
Row 3: Mass number = 24, protons = 12
- Atomic number = 12
- Number of neutrons = 24 - 12 = 12
- Number of electrons = 12 (neutral atom)
- Name = Magnesium (Mg)
Row 4: Atomic number = 15, neutrons = 16
- Number of protons = 15
- Mass number = 15 + 16 = 31
- Number of electrons = 15 (neutral atom)
- Name = Phosphorus (P)
Row 5: Mass number = 1, neutrons = 0
- Number of protons = 1 - 0 = 1
- Atomic number = 1
- Number of electrons = 1
- Name = Hydrogen (H)
Completed Table 8.5:
| Atomic Number | Mass Number | Number of Neutrons | Number of Protons | Number of Electrons | Name of Element |
|---|---|---|---|---|---|
| 5 | 11 | 6 | 5 | 5 | Boron (B) |
| 7 | 14 | 7 | 7 | 7 | Nitrogen (N) |
| 12 | 24 | 12 | 12 | 12 | Magnesium (Mg) |
| 15 | 31 | 16 | 15 | 15 | Phosphorus (P) |
| 1 | 1 | 0 | 1 | 1 | Hydrogen (H) |
Q 14. Aman was discussing the structure of atom with his classmates. During the discussion, he learnt that an element X has a mass number of 35 and contains 18 neutrons. Based on this information, answer the following questions:
(i) How many electrons and protons does element X have?
Ans:
- Mass number = 35, number of neutrons = 18.
- Number of protons = Mass number - Number of neutrons = 35 - 18 = 17.
- Since the atom is neutral, number of electrons = number of protons = 17.
- Answer: Element X has 17 protons and 17 electrons.
(ii) What is its atomic number?
Ans: Atomic number = Number of protons = 17.
(iii) Identify the element X.
Ans: The element with atomic number 17 is Chlorine (Cl).
(iv) Write its electronic configuration.
Ans:
- Atomic number = 17, so 17 electrons.
- K-shell: 2 electrons, L-shell: 8 electrons, M-shell: 7 electrons.
- Electronic configuration = 2, 8, 7.
(v) How many valence electrons does it have?
Ans: The outermost (valence) shell is the M-shell which has 7 electrons. So element X has 7 valence electrons.
(vi) What will be the mass number if two neutrons are added to its nucleus?
Ans:
- Original neutrons = 18, adding 2 more = 20 neutrons.
- New mass number = number of protons + new number of neutrons = 17 + 20 = 37.
- Answer: The new mass number will be 37.
(vii) What will be the relation of X with the new atom?
Ans: The original element X has mass number 35 and atomic number 17. The new atom has mass number 37 and atomic number 17 (same, because protons have not changed). Both atoms have the same atomic number (same element - Chlorine) but different mass numbers. Therefore, X and the new atom are isotopes of each other.
Q 15. In an atom, there are 12 protons and 12 neutrons in the nucleus. Now, imagine that all the electrons are replaced with some hypothetical particles that have the same charge as electrons but are 500 times heavier. What effect will this replacement have on the atom's:
(i) Atomic number
Ans: The atomic number is determined by the number of protons in the nucleus. Since the protons in the nucleus remain unchanged (we only replaced electrons, not protons), the atomic number will remain the same = 12.
(ii) Atomic mass
Ans: Increases, because normally electrons have negligible mass, but replacing them with particles 500 times heavier adds significant extra mass to the atom.
(iii) Mass number
Ans: Mass number = number of protons + number of neutrons. Both protons and neutrons remain unchanged (12 + 12 = 24). Electrons (or the hypothetical particles) are not included in the mass number calculation. Therefore, the mass number will remain unchanged = 24.
(iv) Overall charge
Ans: The hypothetical particles have the same charge as electrons (charge = -1 each). Since the number of hypothetical particles equals the original number of electrons (12), and they carry the same charge, the total negative charge remains -12. The total positive charge from the 12 protons remains +12. Therefore, the overall charge of the atom remains zero - the atom remains electrically neutral. The overall charge will not change.
FAQs on NCERT Solutions: Journey Inside the Atom
| 1. What is the structure of an atom? |  |
| 2. Who proposed the atomic theory and what are its main points? |  |
| 3. What is the significance of the discovery of the electron? |  |
| 4. How do isotopes differ from one another? |  |
| 5. What role do neutrons play in the stability of an atom? |  |






