NCERT Based Activity: Exploring Mixtures and their Separation
Activity 5.1: Let us experiment - Group activity
Divide the class into three groups - A, B and C. Each group prepares a mixture in a colourless, transparent glass tumbler or a 100 mL beaker by following these steps:
- Group A: Add one spatula of common salt to 50 mL of water in a beaker and stir it well. Label it A.
- Group B: Add one spatula of chalk powder to 50 mL of water in a beaker and stir it well. Label it B.
- Group C: Add a few drops of milk to 50 mL of water in a beaker and stir it well. Label it C.
- Are the particles visible in each mixture? Record your observations.
- Direct the light from a laser pointer through the beakers containing the mixtures (Fig. 5.3) and observe it from the side of the beaker in a direction perpendicular to the laser beam. Record your observations.
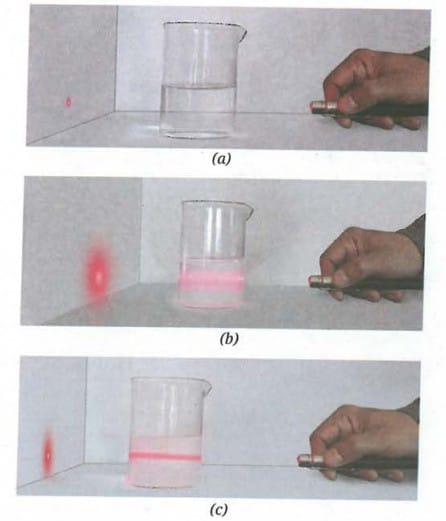 Fig. 5.3: Passing laser light through various mixtures (a) salt and water, (b) chalk powder and water, and (c) milk and water
Fig. 5.3: Passing laser light through various mixtures (a) salt and water, (b) chalk powder and water, and (c) milk and water
Safety first: Do not look directly into the laser beam. It can cause irreversible eye damage.
- Predict what you would observe in each of the beakers if you leave them undisturbed for a few minutes.
- Set up a filtration apparatus and filter each mixture separately. Is there any residue left on the filter paper?
- Based on your observations, do you think these are the same types of mixtures or are they different?
Observation:
Beaker A (Salt and water): No particles are visible. The laser beam path is not visible when passed through the mixture. On leaving undisturbed, no settling occurs - the mixture remains clear and uniform. No residue is left on the filter paper. This is a solution (homogeneous mixture).
Beaker B (Chalk powder and water): Particles are clearly visible. The laser beam path is clearly visible (Tyndall effect observed). On leaving undisturbed, chalk particles settle to the bottom. Residue is left on the filter paper. This is a suspension (heterogeneous mixture).
Beaker C (Milk and water): Particles are not visible to the naked eye. The laser beam path is visible (Tyndall effect observed), even though the mixture appears uniform. On leaving undisturbed, no settling occurs. No residue is left on the filter paper. This is a colloid (heterogeneous mixture that appears homogeneous).
Explanation:
The three beakers represent the three main types of mixtures. A solution (Beaker A) has very small solute particles (less than 1 nm diameter) that are completely dissolved, do not scatter light, and cannot be separated by filtration. A suspension (Beaker B) has large particles (more than 1000 nm diameter) that are visible, settle on standing, scatter light, and can be separated by filtration. A colloid (Beaker C) has intermediate-sized particles (1-1000 nm) that do not settle, scatter light (showing the Tyndall effect), and cannot be separated by ordinary filtration.
This activity demonstrates that mixtures can be classified as solutions, suspensions and colloids based on properties such as particle size, visibility, settling, Tyndall effect and filterability.
Activity 5.2: Let us represent solubility graphically
Consider water as the solvent, and compounds 'A' and 'B' as the solutes. Each substance has a different solubility. A graph of solubility versus temperature is called a solubility curve. The solubility curves for 'A' and 'B' as solutes are shown in Fig. 5.6. The x-axis shows temperature (°C), whereas the y-axis indicates the solubility of the solute in grams per 100 g of water.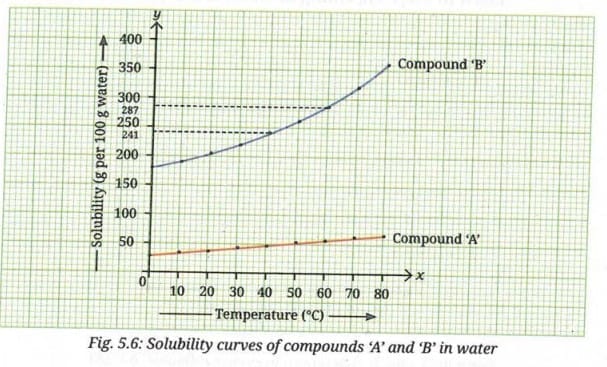
Based on the information from the above graph, predict which of the two compounds, 'A' or 'B', will dissolve more in a given amount of water at a given temperature?
Observe Fig. 5.6 and fill in the blanks of the following statements:
(i) The solubility of compound 'A' in water at 20 °C is less than (less than/more than/similar to) its solubility at 60 °C.
(ii) The solubility of compound 'B' at 20 °C is less than (less than/more than/similar to) its solubility at 60 °C.
(iii) The solubility of compound 'B' increases more than that of compound 'A' with an increase in the temperature.
What do you think will happen if you make a saturated solution at a higher temperature and cool it slowly? Let us find out!
Observation:
- At any given temperature shown on the graph, Compound 'B' has a much higher solubility than Compound 'A'. Therefore, Compound 'B' will dissolve more in a given amount of water at a given temperature.
- The solubility of both Compound 'A' and Compound 'B' increases with an increase in temperature - this is typical behaviour for solid solutes in liquid solvents.
- Compound 'B' shows a much steeper rise in solubility with temperature compared to Compound 'A', whose solubility curve is relatively flat.
- At 20 °C, the solubility of Compound 'B' is approximately 50 g per 100 g of water, while at 60 °C it rises to approximately 241 g per 100 g of water - a very large increase.
- Compound 'A' shows only a modest increase in solubility across the same temperature range.
Explanation:
The solubility of a substance is the maximum amount of solute that can dissolve in a fixed quantity of solvent at a given temperature. A solution that cannot dissolve any more solute at that temperature is called a saturated solution. The solubility of most solid solutes in liquid solvents increases with temperature. Compound 'B' shows a significantly greater dependence of solubility on temperature than Compound 'A'. This difference in solubility-temperature behaviour forms the basis of crystallization - if a saturated solution of Compound 'B' is made at a high temperature and then cooled slowly, the excess solute that can no longer remain dissolved will separate out as pure crystals.
Activity 5.3: Let us prepare
- Collect a sample of copper sulfate (blue vitriol). If copper sulfate is unavailable, you can use common salt as an alternative.
Safety first: Copper sulfate is toxic. Perform the experiment under adult supervision and do not touch it with your bare hands.
- Take 1 g of copper sulfate and place it in a 100 mL beaker. Add 25 mL of water to the beaker and add a drop of dilute sulfuric acid. Gently heat the mixture in a water bath while stirring constantly. Sulfuric acid helps in making pure crystals by preventing unwanted reactions.
Safety first: It is advised that the teacher should add a drop of sulfuric acid. Handle sulfuric acid very carefully!
- Gradually, add more copper sulfate until the solution becomes saturated (Fig. 5.8a).
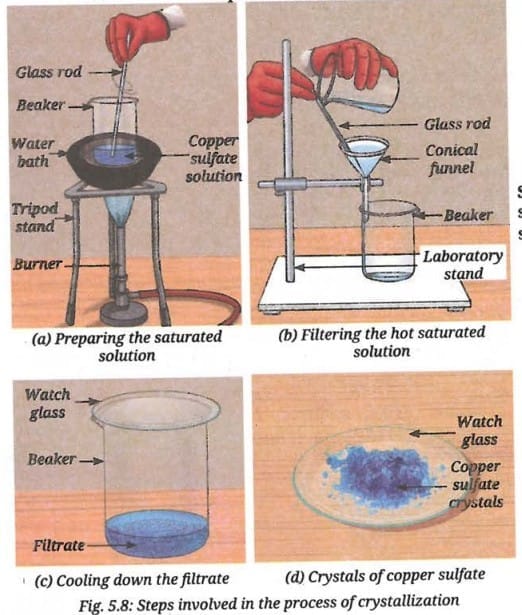
- Filter the hot solution to remove insoluble impurities (Fig. 5.8b). Collect it in a clean beaker and cover it with a watch glass.
- Allow the solution to cool slowly without disturbing it (Fig. 5.8c). It gives enough time to the particles in the solution to come together, resulting in the formation of larger, shiny, well-shaped and blue-coloured crystals.
- Filter the crystals, rinse them with cold water and allow them to dry on a watch glass (Fig. 5.8d).
You may also try:
- Place 1-2 mL of the saturated copper sulfate solution on a small glass plate or a lamination sheet.
- Leave it for some time. What do you observe?
Did you get crystals? If yes, is this a good way to experiment? Explain.
Observation:
- On heating, the copper sulfate dissolves readily in water to form a blue solution.
- On gradual addition of more copper sulfate, a point is reached where no more copper sulfate dissolves - the solution becomes saturated.
- On filtering the hot saturated solution, a clear, deep blue filtrate is obtained with any undissolved impurities remaining on the filter paper.
- On cooling slowly, large, well-shaped, shiny, blue crystals of copper sulfate form at the bottom and sides of the beaker.
- When the saturated solution is placed on a glass plate and allowed to evaporate at room temperature, smaller and less well-shaped crystals form compared to slow cooling. This shows that slow cooling gives larger, purer, better-shaped crystals.
Explanation:
Crystallization is the process of forming crystals from a saturated solution. When a hot saturated solution is cooled slowly, the solubility of the solute decreases with falling temperature. The excess solute that can no longer remain dissolved separates out as pure, well-shaped crystals. Slow cooling allows particles more time to arrange themselves into an orderly, regular geometric structure - producing larger, purer crystals. Rapid cooling or evaporation at room temperature produces smaller, less ordered crystals. Crystallization is a common technique used to purify solids - impurities present in small quantities remain in solution while the pure substance crystallizes out.
Think as a Scientist
If a hot, saturated solution of copper sulfate is cooled rapidly in ice-cold water, smaller and less well-formed crystals will form than if it is cooled slowly at room temperature. How would you design and perform an experiment to test this hypothesis?
Hint: Prepare a hot saturated solution of copper sulfate and divide it into two equal parts.
Experimental Design to Test the Hypothesis:
Hypothesis:
Rapid cooling of a hot saturated copper sulfate solution produces smaller, less well-formed crystals compared to slow cooling at room temperature.
Materials Required:
- Copper sulfate powder
- Distilled water
- Beaker (2), heating apparatus
- Ice-cold water bath
- Thermometer, stirring rod
- Filter paper, microscope/magnifying glass
Procedure:
Step 1 - Prepare the solution:Heat distilled water and dissolve as much copper sulfate as possible to make a hot saturated solution.
Step 2 - Divide into two equal parts:Pour the hot saturated solution equally into two identical beakers (A and B).
Step 3 - Cool differently:
| Beaker A | Beaker B |
|---|---|
| Place in ice-cold water (rapid cooling) | Leave at room temperature (slow cooling) |
Step 4 - Observe crystal formation:Once crystals form in both beakers, filter and collect them. Observe under a magnifying glass or microscope.
Observations & Expected Results:
| Parameter | Beaker A (Rapid) | Beaker B (Slow) |
|---|---|---|
| Crystal size | Smaller | Larger |
| Crystal shape | Irregular, poorly formed | Well-defined, regular |
| Colour | Pale blue | Deep blue |
Conclusion:
If Beaker A gives smaller, irregular crystals and Beaker B gives larger, well-formed crystals, the hypothesis is supported. Slow cooling allows molecules more time to arrange themselves into proper crystal lattices, while rapid cooling "freezes" them into many small, disorganised clusters.
Variable Control:
- Independent variable: Rate of cooling
- Dependent variable: Crystal size and shape
- Controlled variables: Same solution, same volume, same concentration
Activity 5.4: Let us describe a process
Observe Fig. 5.9, it shows how salt crystals are obtained from seawater. Can you describe the process in your own words?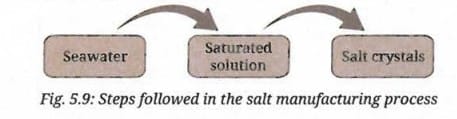
When we allow a solvent to evaporate to get a solute, such as salt from a salt solution, the solvent disappears. However, we may also want to recover the solvent. What should we do then? While experimenting in the laboratory, a student accidentally mixed acetone and water, two miscible liquids. How can we separate the two miscible liquids? Is it possible to separate the mixture of two miscible liquids by evaporation and obtain both the liquids?
Observation:
- Seawater is collected in large, shallow open pans or fields near the coast.
- The water is allowed to evaporate slowly under sunlight and wind over many days, gradually increasing the concentration of dissolved salts.
- As the water evaporates, the solution becomes more and more concentrated until it reaches saturation.
- Once the solution is saturated, further evaporation causes the salt to crystallize and separate out as solid crystals.
- The salt crystals are then collected, washed and dried.
Explanation:
The process of obtaining salt from seawater involves evaporation followed by crystallization. Seawater contains dissolved salts (primarily sodium chloride). When seawater is exposed to sunlight and wind in large shallow basins, the solvent (water) evaporates gradually. As the water content decreases, the concentration of dissolved salt increases until the solution becomes saturated. At this point, further evaporation causes the dissolved salt to come out of solution as solid crystals - this is crystallization. The process depends on the principle that solubility of a solid solute decreases when the solvent is removed by evaporation, causing the excess solute to separate as a pure crystalline solid.
Activity 5.5: Let us investigate
- Take a 3 cm wide strip of chromatographic paper and draw a straight, horizontal line, 2 cm from the bottom of the paper with a pencil (Fig. 5.15a). Alternatively, you can use a strip of filter paper.
- Mark a spot with a black sketch pen at the centre of the line (Fig. 5.15b).
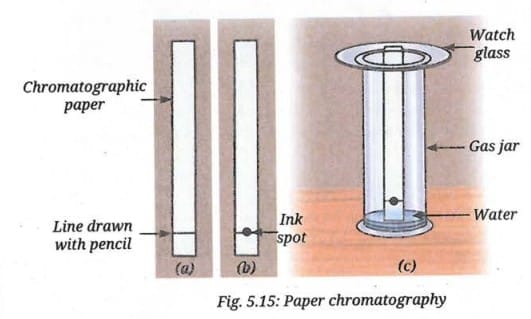
- Take enough water to make a thin layer at the bottom of a gas jar, a measuring cylinder, or a beaker.
- Place the paper strip with the ink spot vertically into the container, so that its lower end dips into the water. The water level should be below the spot, as shown in Fig. 5.15c.
- Observe the paper as the water rises through the paper. What do you notice?
- As the water rises, the ink starts to separate into different colour spots. What can you infer from this?
Observation:
- As water rises through the paper, it carries the ink components along with it.
- The black ink separates into distinct bands of different colours - typically blue, violet, red or yellow - at different heights on the paper.
- The different colour components travel at different speeds and distances from the original spot, indicating that black ink is a mixture of several pigments/dyes.
- The component that interacts least with the paper and most with the solvent (water) travels the highest, while the one that interacts most with the paper travels the least distance.
- This separation occurs because each component has a different solubility in the solvent and a different affinity for the paper.
Explanation:
This method of separating the components of a mixture is known as paper chromatography. It uses differences in the interactions of the components with the solvent and the paper to separate them. The liquid (mobile phase) carries the substances up the paper (stationary phase), separating them based on how fast they move. Substances that are more soluble in the solvent and interact less with the paper move faster and travel a greater distance. Substances with greater affinity for the paper move more slowly. Paper chromatography can be used to separate pigments, dyes, inks and other coloured substances, and can also be used to identify unknown components by comparing the distances they travel with known standards.
Activity 5.6: Let us separate
- Pour a mixture of 5 mL mustard oil and 20 mL water into a 50 mL separating funnel.
- Let it stand undisturbed. What do you observe?
- You will see the formation of two separate layers of mustard oil and water (Fig. 5.16). The yellow-coloured mustard oil forms the upper layer and water forms the lower layer. Can you explain why?
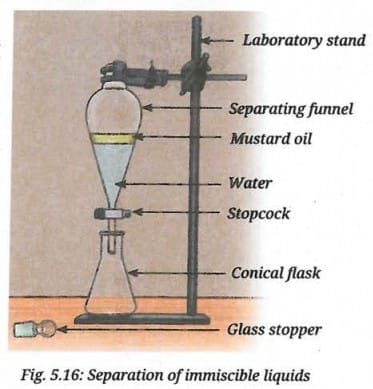
- Open the stopcock of the separating funnel slowly to collect the lower layer of water carefully into a container.
- Close the stopcock when the water is almost fully drained.
- Collect the next small portion that may contain both liquids and discard it.
- Collect the layer of oil separately by opening the stopcock again.
You now have two separate liquids.
Can you think of any heterogeneous mixtures with a gas as one of the components?
Observation:
- On standing undisturbed, the mixture separates into two distinct layers - the yellow mustard oil layer on top and the colourless water layer at the bottom.
- The two layers are clearly visible and do not mix with each other - they are immiscible liquids.
- Mustard oil forms the upper layer because it is less dense than water.
- Water forms the lower layer because it is denser than mustard oil.
- On opening the stopcock of the separating funnel, the lower layer (water) drains out first.
- After discarding the intermediate mixed portion, the upper layer (oil) is collected separately.
- At the end, two separate, pure liquids - water and mustard oil - are obtained.
Explanation:
A separating funnel is used to separate two immiscible liquids - liquids that do not mix with each other and form separate layers. The principle behind this technique is the difference in densities of the two liquids. The denser liquid (water) sinks to the bottom while the less dense liquid (mustard oil) remains on top. By carefully opening the stopcock, the lower layer is drained off first, followed by the upper layer, effectively separating the two liquids. This technique is widely used in laboratories and industries to separate organic compounds from aqueous solutions, to separate petroleum products, and in the extraction of essential oils from plants.
Activity 5.7: Let us explore
- Take a spatula full of the mixture containing crushed camphor and sand. Put it into a clean and dry china dish. Place it on the tripod stand using a wire gauze.
- Take a clean and dry glass funnel. Plug its nozzle with cotton.
- Keep this funnel inverted on the china dish and set up the apparatus as shown in Fig. 5.17.
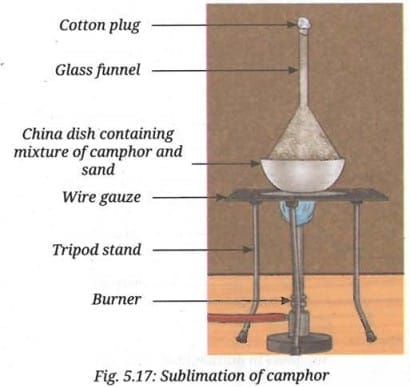
- Light the burner and place it under the wire gauze.
- Heat the china dish gently for a few minutes.
- Observe the inner wall of the funnel carefully. Do you notice any solid deposits?
- You may find white, solid camphor deposits on the inner wall of the funnel while sand remains in the china dish.
Observation:
- On gentle heating, the mixture of camphor and sand in the china dish is warmed.
- The camphor does not melt - instead, it directly converts from solid to vapour without passing through the liquid state.
- White, solid camphor deposits appear on the inner wall of the inverted glass funnel, as the vapours cool down and solidify on the cooler surface of the funnel.
- The sand remains behind in the china dish, as it does not undergo sublimation.
- The cotton plug in the nozzle of the funnel prevents the camphor vapours from escaping.
- Complete separation of camphor from sand is achieved by this process.
Explanation:
Sublimation is the process by which a solid directly changes into the vapour state without passing through the liquid state, on heating (below its melting point). The reverse process - when the vapour cools and directly changes back into a solid without becoming a liquid - is called deposition. Camphor sublimes readily on gentle heating and can therefore be separated from non-sublimable substances like sand by this method. The camphor vapours rise, cool on the inner surface of the funnel, and deposit as pure solid camphor. Sand, which does not sublime, remains in the china dish. Other examples of sublimable substances include naphthalene, iodine and solid carbon dioxide (dry ice). Sublimation is used in industry for the purification of substances like iodine and naphthalene.
Activity 5.8: Let us make a model
Make your own centrifuge with a cardboard disc and thick thread. You will be able to see how the heavier particles move outwards. It is a fun and hands-on way to understand the science behind separation. Which mixture would you like to separate using this mini centrifuge?
Observation:
- When the cardboard disc is spun slowly, particles placed on it tend to remain near the centre.
- As the speed of spinning increases, the particles are pushed outwards towards the edge of the disc, demonstrating the centrifugal force in action.
- The faster the disc spins, the stronger the outward push felt on the particles.
- In a real centrifuge, when a mixture (such as muddy water or blood) is spun at high speed in tubes, the heavier particles move outward and settle at the bottom of the tubes, while the lighter liquid remains at the top.
- This principle is demonstrated even with the simple cardboard model.
Explanation:
Centrifugation is a method of separating the components of a mixture by spinning it at a high speed. During centrifugation, the tubes containing the mixture become horizontal and the centrifugal force (outward force acting on a body moving in circular motion) causes the heavier particles to move outward and settle at the bottom of the tube, while the lighter liquid remains at the top. The simple cardboard model demonstrates this principle in a hands-on way. Centrifugation is widely used in laboratories to separate the components of blood (plasma, platelets, red blood cells, white blood cells), in the dairy industry to separate cream from milk, and in many chemical industries.
Activity 5.9:
Complete Table 5.1 and review what you have learnt about solutions, suspensions and colloids.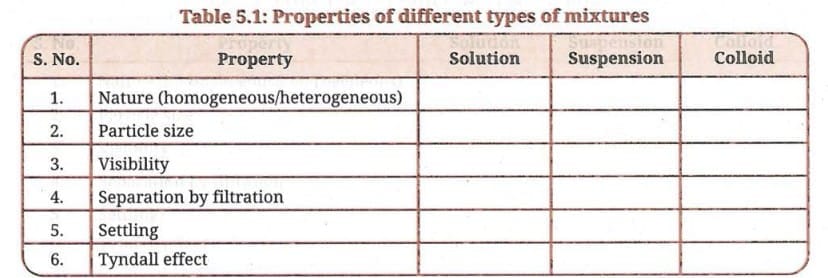
Observation:
Table 5.1: Properties of different types of mixtures
| S. No. | Property | Solution | Suspension | Colloid |
|---|---|---|---|---|
| 1. | Nature (homogeneous/heterogeneous) | Homogeneous | Heterogeneous | Appears homogeneous but is heterogeneous |
| 2. | Particle size | Less than 1 nm diameter | More than 1000 nm diameter | 1-1000 nm diameter |
| 3. | Visibility | Particles not visible even under microscope | Particles visible to naked eye | Particles not visible to naked eye |
| 4. | Separation by filtration | Cannot be separated by filtration | Can be separated by filtration | Cannot be separated by ordinary filtration |
| 5. | Settling | Does not settle on standing | Settles on standing when left undisturbed | Does not settle on standing |
| 6. | Tyndall effect | Does not show Tyndall effect | Shows Tyndall effect | Shows Tyndall effect |
- Solutions are perfectly homogeneous with the smallest particle size and do not show the Tyndall effect, settle, or get filtered.
- Suspensions are clearly heterogeneous with the largest particle size, are visible to the naked eye, settle on standing, can be filtered, and show the Tyndall effect.
- Colloids occupy the intermediate position - they appear homogeneous but are actually heterogeneous, their particles are intermediate in size, they do not settle, cannot be filtered by ordinary filter paper, but clearly show the Tyndall effect.
Explanation:
The three types of mixtures - solutions, suspensions and colloids - differ mainly in the size of the dispersed particles. In a solution, the solute particles (less than 1 nm) are completely dissolved and uniformly distributed. In a suspension, the particles (more than 1000 nm) are so large that they remain suspended temporarily but eventually settle due to gravity. In a colloid, the dispersed particles (1-1000 nm) are large enough to scatter light (causing the Tyndall effect) but small enough to remain uniformly dispersed without settling. The Tyndall effect - the scattering of a beam of light by particles - is the key test that distinguishes a colloid or suspension from a true solution. Examples: Solution - salt in water; Suspension - chalk in water; Colloid - milk, blood, fog.
FAQs on NCERT Based Activity: Exploring Mixtures and their Separation
| 1. What is solubility and how is it represented graphically? |  |
| 2. What methods can be used to separate mixtures? |  |
| 3. How can one investigate the solubility of a substance? |  |
| 4. What is the significance of preparing mixtures in a laboratory setting? |  |
| 5. What role do models play in understanding mixtures and their separation? |  |






