NCERT Based Activity: Journey Inside the Atom
Think as a Scientist
Observe Fig. 8.4 of the gold foil experiment. Predict the observations you would expect if the gold foil in the experiment were made thicker. Also, draw a simple diagram to show the observations you expect.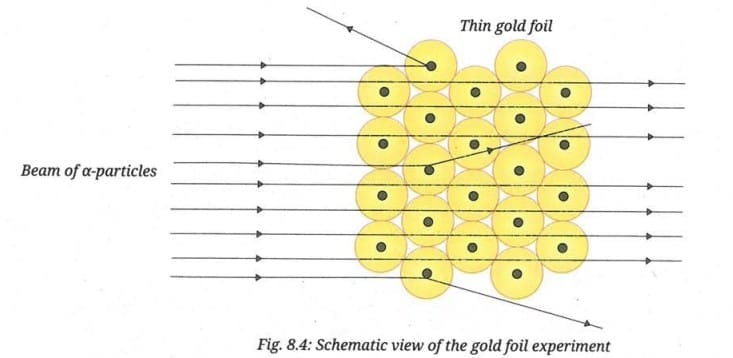
Hint: Compare thin foil vs thick foil. How does the thickness affect the chances of hitting a nucleus?
Observations:
- If the gold foil is made thicker, the beam of alpha particles encounters a greater number of gold atoms and nuclei along its path.
- More alpha particles would be deflected at large angles because the probability of a direct or near-direct collision with a nucleus increases with thickness.
- A greater number of alpha particles would also bounce back (backscattering) compared to the thin foil experiment.
- Fewer alpha particles would pass through completely undeflected, since increased thickness reduces the chance of passing through empty space without any interaction.
- The pattern of deflections would be significantly more pronounced compared to the original thin foil experiment.
Explanation:
In Rutherford's model, an atom is mostly empty space with a tiny, dense, positively charged nucleus at the centre. When the foil is thin, most alpha particles pass through the vast empty space without hitting any nucleus, and are therefore undeflected. Only a very few particles travel close enough to a nucleus to get deflected.
When the foil is made thicker, there are many more layers of gold atoms in the path of the alpha particles. This means:
- The probability of an alpha particle encountering a nucleus increases with each additional atomic layer.
- Even if one layer is mostly empty space, multiple layers together significantly raise the chance of a nucleus collision.
- Therefore, more large-angle deflections and backscattering are observed.
This reinforces Rutherford's conclusion that the nucleus is a real, concentrated region of positive charge and mass - its presence becomes statistically more detectable as foil thickness increases.
FAQs on NCERT Based Activity: Journey Inside the Atom
| 1. What is an atom and what are its main components? |  |
| 2. How do protons and neutrons differ in terms of charge and mass? |  |
| 3. What is the significance of electrons in an atom? |  |
| 4. What is meant by atomic number and mass number? |  |
| 5. How do isotopes of an element differ from each other? |  |





