NCERT Based Activity: Atomic Foundations of Matter
Activity 9.1: Let us investigate a physical change
- Place a clean and dry 100 mL beaker on a digital weighing balance.
- Set the balance reading to zero by pressing the tare or reset button.
- Pour about 50 mL of water into the beaker.
- Add a spatula full of common salt to the water contained in the beaker.
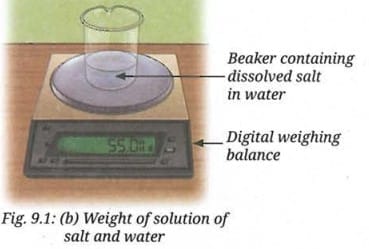
- Record the reading on the weighing balance (Fig. 9.1a).

- Swirl until the added salt dissolves and record your observations (Fig. 9.1b).
What do you observe?
Observation:
The mass of the solution is equal to the sum of the masses of water and salt taken. There is practically no change in the mass during the formation of a solution, which is a physical change. This is true for all physical changes.
Explanation:
During a physical change, no new substance is formed. The salt simply dissolves into the water - the atoms and molecules are rearranged physically but not chemically changed. Since no matter is created or destroyed, the total mass remains the same before and after the change. This is consistent with the Law of Conservation of Mass.
Activity 9.2: Let us investigate a chemical change
You have learnt about various chemical changes. Do you remember what happened when baking soda was added to vinegar? A gas, carbon dioxide, was formed during this chemical change and the reaction is represented as -
Vinegar + Baking soda (Sodium hydrogencarbonate) → Carbon dioxide + Other substances
Let us explore whether the mass remains the same before and after the change.
Experimental Set-up 1
- Place a clean, dry 100 mL conical flask and a medium-sized balloon on a weighing balance.
- Set the balance reading to zero by pressing the tare or reset button.
- Pour about 20 mL of vinegar or lemon juice into the conical flask.
- Take about 2 g of baking soda (sodium hydrogencarbonate) and put it into the balloon.
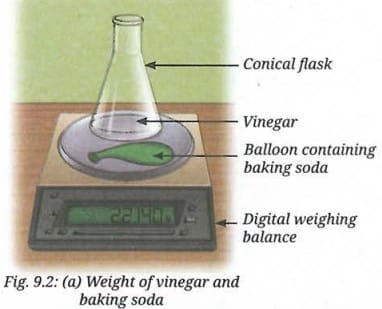
- Keep the balloon filled with baking soda on the weighing balance next to the conical flask. Record the initial reading (Fig. 9.2a).
- Carefully transfer the baking soda from the balloon into the conical flask containing vinegar (Fig. 9.2b).
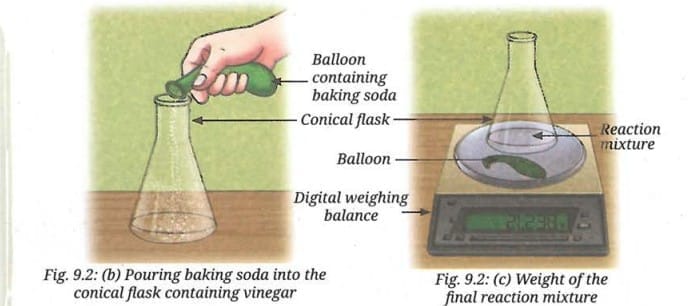
- Place the conical flask and balloon back on the weighing balance, and record the final reading (Fig. 9.2c).
- What do you observe?
- Are the initial and the final readings the same?
A brisk effervescence is observed. The final reading does not match the initial reading. What can be the reason for this?
Observation:
The reason is that this is an open system. When baking soda reacts with vinegar, carbon dioxide gas is produced. Since the balloon is not fixed to the flask, this gas escapes freely into the atmosphere and is no longer on the weighing balance. The mass of the escaped CO₂ is therefore not accounted for in the final reading, causing the final mass to appear less than the initial mass.
This does not mean the Law of Conservation of Mass is violated - the total mass is still conserved, but some of it (the CO₂ gas) has left the system and is no longer being measured.
Experimental Set-up 2
- Place a clean, dry 100 mL conical flask and a medium-sized balloon on a weighing balance.
- Set the balance reading to zero by pressing the tare or reset button.
- Pour about 20 mL of vinegar or lemon juice into the conical flask.
- Place about 2 g of baking soda (sodium hydrogencarbonate) in the balloon.
- Fix the balloon to the mouth of the conical flask using a thread, without allowing the baking soda to mix with the vinegar.
- Weigh the conical flask containing vinegar and the balloon containing baking soda, and record the reading (Fig. 9.3a).
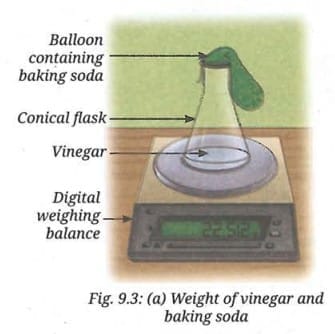
- Lift the other end of the balloon upwards, allowing the baking soda to fall into the vinegar (Fig. 9.3b).
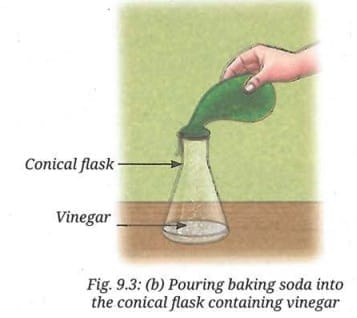
- What do you observe?
- As in experimental set-up 1, a brisk effervescence occurs, which inflates the balloon during the reaction.
- Record the final reading (Fig. 9.3c).
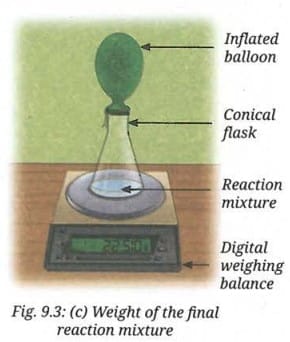
- Are the initial and the final readings the same?
Observation:
Yes, the initial and final readings are the same.
The total mass of vinegar and baking soda before the reaction equals the total mass of the reaction mixture and the carbon dioxide gas (trapped in the balloon) after the reaction. Since no gas escapes the system, the total mass is conserved.
This confirms the Law of Conservation of Mass - matter can neither be created nor destroyed in a chemical reaction. The difference seen in Set-up 1 was only because the CO₂ gas escaped into the open air, making it appear as though mass was lost. Set-up 2 proves that no mass is actually lost.
Explanation:
In Experimental Set-up 1, the gas (CO₂) produced escapes into the open atmosphere, so the measured mass decreases. This does not mean mass is destroyed - it simply escapes the system. In Experimental Set-up 2, the closed system (balloon attached to flask) traps all the gas, and the total mass remains unchanged. This demonstrates the Law of Conservation of Mass: matter can neither be created nor destroyed in a chemical reaction.
Activity 9.3: Let us verify the law - Group activity
- Place two clean and dry 100 mL conical flasks on a weighing balance, and mark them A and B.
- Set the balance reading to zero by pressing the tare or reset button.
- Pour about 10 mL of 1% m/v sodium sulfate solution into Conical Flask A.
- In Conical Flask B, pour about 10 mL of 1% m/v barium chloride solution.
- Leave both Conical Flasks A and B on the weighing balance undisturbed, and record the total mass of both the solutions (Fig. 9.4a).

- Transfer the solution from Conical Flask B to Conical Flask A and mix the two solutions carefully.
- What do you observe?
- Now, place both Conical Flasks A and B on the weighing balance again as shown in Fig. 9.4b, and note the reading.
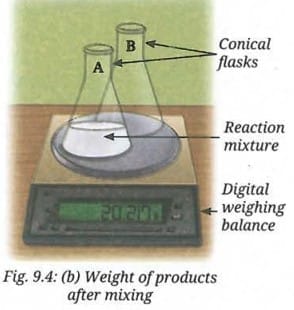
- Do you observe any change in the reading after mixing the solutions?
Observation:
When solutions of sodium sulfate and barium chloride are mixed, a white precipitate of barium sulfate is formed along with sodium chloride. The reading on the weighing balance after mixing is the same as before mixing. No change in total mass is observed.
Chemical Reaction:
Sodium sulfate + Barium chloride → Barium sulfate + Sodium chloride
Explanation:
This activity verifies the Law of Conservation of Mass for a chemical reaction carried out in a closed system (no gas is formed and lost). The total mass of the reactants (sodium sulfate and barium chloride) equals the total mass of the products (barium sulfate precipitate and sodium chloride solution). This confirms that mass is conserved during a chemical reaction.
Think as a Scientist
You are given a chemical reaction in which zinc reacts with dilute hydrochloric acid to form zinc chloride and hydrogen gas.
Zinc + Hydrochloric acid (dilute) → Zinc chloride + Hydrogen
Design and perform an experiment to test the hypothesis that mass is conserved during this chemical reaction. You may use a set-up different from the one shown in Activity 9.2.
Hypothesis:
The total mass of zinc and dilute hydrochloric acid (reactants) before the reaction will be equal to the total mass of zinc chloride and hydrogen gas (products) after the reaction, provided the system is closed so that no gas escapes.
Experimental Design:
- Take a conical flask fitted with a balloon at its mouth (similar to Experimental Set-up 2 of Activity 9.2).
- Place a small measured amount of zinc granules inside the conical flask.
- Pour a measured volume of dilute hydrochloric acid into a small test tube or container inside the flask, without allowing it to mix with the zinc yet.
- Seal the system completely using the balloon.
- Weigh the entire set-up on a digital balance and record the initial mass.
- Tilt the flask carefully to allow the hydrochloric acid to mix with the zinc granules.
- Observe the reaction - hydrogen gas is produced, which inflates the balloon.
- Once the reaction is complete, weigh the entire set-up again and record the final mass.
Observation:
- Brisk effervescence (bubbles of hydrogen gas) is observed around the zinc granules.
- The balloon inflates as hydrogen gas is produced.
- The zinc granules gradually dissolve in the acid.
- The initial mass and the final mass of the closed system are the same.
Chemical Reaction:
Zn (s) + 2HCl (aq) → ZnCl₂ (aq) + H₂ (g)
Explanation:
In a closed system where the balloon prevents the hydrogen gas from escaping, all products including the gas remain within the system. The total mass of the reactants (zinc + hydrochloric acid) equals the total mass of the products (zinc chloride + hydrogen gas). This confirms that the Law of Conservation of Mass holds true for this chemical reaction. If the system were open (no balloon), the hydrogen gas would escape and the final measured mass would appear less than the initial mass - but this would not mean mass was destroyed; it simply left the system.
Activity 9.4: Let us experiment
- Collect samples of some compounds: camphor, sodium chloride, copper sulfate, sugar, and naphthalene.
(A) Solubility in (i) water, (ii) kerosene, and (iii) petrol
- Try dissolving each sample separately in water, kerosene and petrol.
- Record your observations in Table 9.2.
(B) Electrical conductivity in water
Safety first: Do not touch the electrodes when they are connected to the battery but use a low-voltage battery to avoid the risk of shock. Petrol and kerosene are flammable liquids, so be careful while working with them.
- Take two carbon or metal electrodes and insert them into a piece of cardboard by drilling two holes.
- Connect one end of each electrode to the terminals of the 9 V battery and to the light bulb.
- Test the electrical conductivity of each solid sample and observe whether the bulb glows. Record your observations.
- Test the electrical conductivity of each sample dissolved in water by transferring the solution to the beaker one at a time and observing whether the bulb glows (Fig. 9.15).
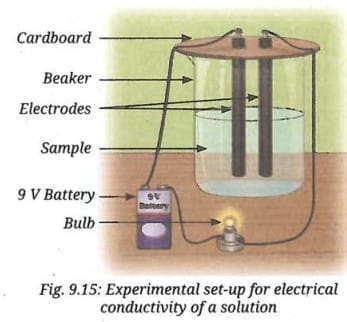
- Record your observations with other samples given in Table 9.2.
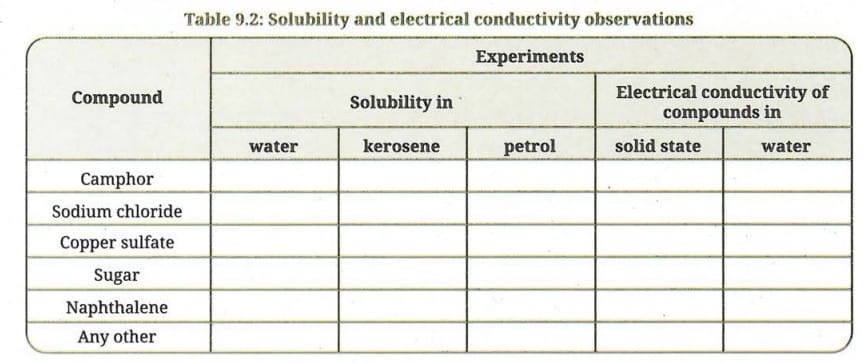
- Group the compounds showing similar properties listed in Table 9.2.
Observation:
Here is the completed Table 9.2:
| Compound | Solubility in Water | Solubility in Kerosene | Solubility in Petrol | Electrical Conductivity (Solid State) | Electrical Conductivity (Water) |
|---|---|---|---|---|---|
| Camphor | Insoluble | Soluble | Soluble | No | No |
| Sodium chloride | Soluble | Insoluble | Insoluble | No | Yes |
| Copper sulfate | Soluble | Insoluble | Insoluble | No | Yes |
| Sugar | Soluble | Insoluble | Insoluble | No | No |
| Naphthalene | Insoluble | Soluble | Soluble | No | No |
Group 1 - Ionic Compounds (Sodium chloride, Copper sulfate):
- Soluble in water, insoluble in kerosene and petrol
- Do not conduct electricity in solid state
- Conduct electricity when dissolved in water (ions are free to move)
Group 2 - Covalent Compounds (Camphor, Naphthalene):
- Insoluble in water, soluble in kerosene and petrol
- Do not conduct electricity in solid state or in water
Group 3 - Covalent Compound soluble in water (Sugar):
- Soluble in water but does not conduct electricity because it does not produce ions in solution
- Insoluble in kerosene and petrol
Explanation:
Ionic compounds are formed by the transfer of electrons, producing charged ions. In the solid state, these ions are fixed in a crystal lattice and cannot move, so they do not conduct electricity. When dissolved in water, the ions become free to move, enabling electrical conduction. Covalent compounds are formed by the sharing of electrons and do not produce free ions either in solid state or in solution (except acids), so they generally do not conduct electricity. They tend to dissolve in non-polar solvents like kerosene and petrol due to the principle "like dissolves like."
FAQs on NCERT Based Activity: Atomic Foundations of Matter
| 1. What is a physical change? |  |
| 2. What is a chemical change? |  |
| 3. How can we verify the law of conservation of mass through experiments? |  |
| 4. What are some indicators of a chemical change? |  |
| 5. Why is it important to understand the atomic foundations of matter? |  |





