Mnemonics: Exploring Mixtures and their Separation
🧪 Chemistry Mixtures & Separation: Mnemonics for Quick Revision
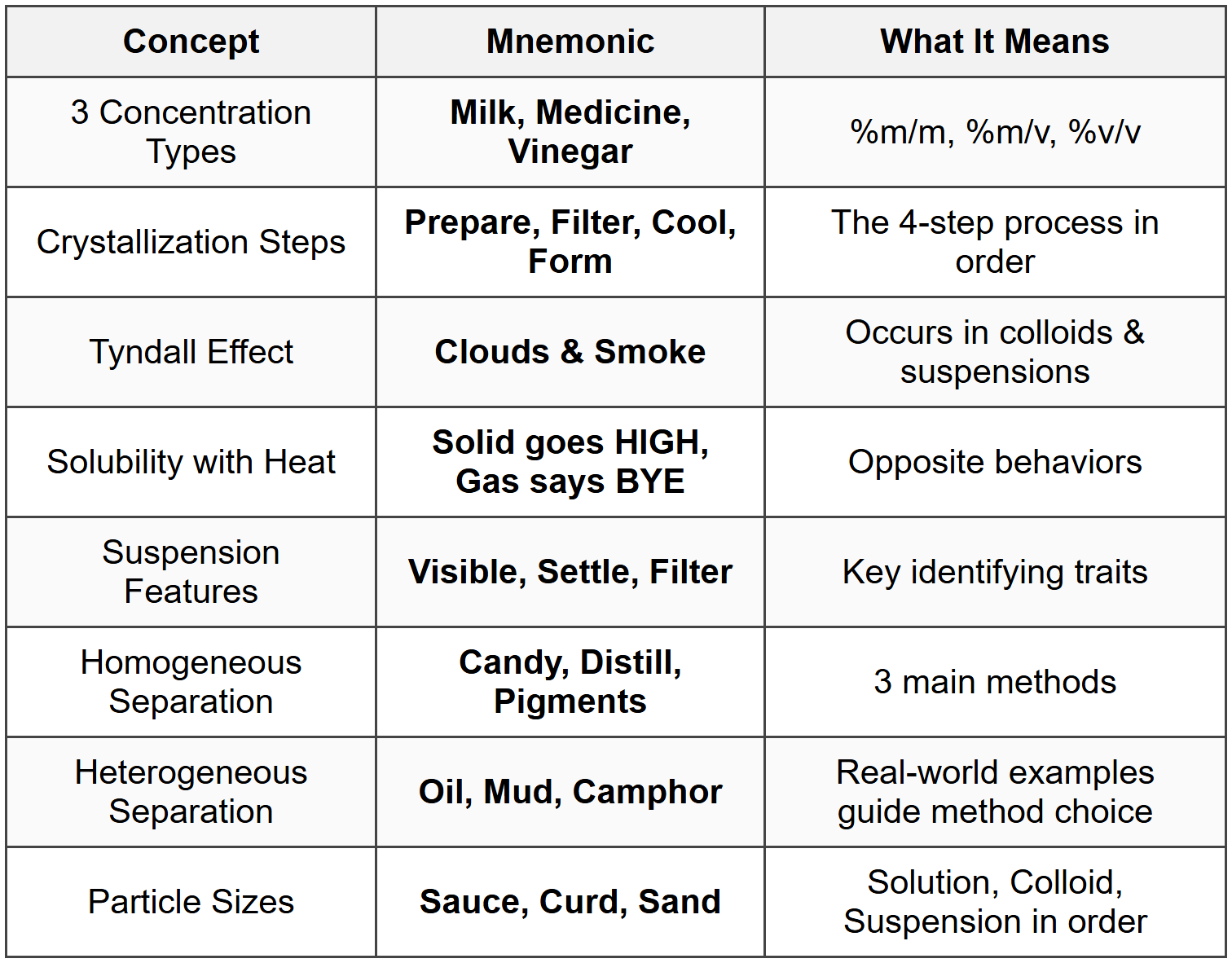
Three Ways to Express Concentration
What needs to be memorized: Mass by Mass Percentage (%m/m), Mass by Volume Percentage (%m/v), Volume by Volume Percentage (%v/v)
Mnemonic: 🥛 Milk, Medicine, Vinegar 💊 🍶
🔗 The Breakdown:
- Milk → Mass by Mass (%m/m) - Like measuring milk powder content in packaged milk packets (mass in mass)
- Medicine → Mass by Volume (%m/v) - Like glucose IV solutions (5% m/v = 5g solute in 100mL solution)
- Vinegar → Volume by Volume (%v/v) - Like vinegar concentration in cooking (measured by volume in volume)
💡 Memory Tip: Three everyday household items! When you see a milk packet, think mass in mass. When a doctor mentions IV solutions, think mass in volume. When cooking with vinegar, think volume in volume.
Steps of Crystallization Process
What needs to be memorized: Four sequential steps to form pure crystals from a saturated solution
Mnemonic: Prepare → Filter → Cool → Form
🔗 The Breakdown:
- Prepare → Prepare a saturated solution by heating and adding solute until no more dissolves
- Filter → Filter the hot solution through filter paper to remove any insoluble impurities
- Cool → Cool the filtered solution slowly without disturbance (slow cooling = bigger, better crystals)
- Form → Formation of crystals - pure crystals separate out and can be collected
💡 Memory Tip: Think of it like cooking instructions: Prepare your ingredients, Filter out impurities, Cool slowly, and the final Form appears! It's a logical flow, just like a recipe.
Tyndall Effect: Where It Occurs
What needs to be memorized: In which type of mixtures the Tyndall Effect is visible
Mnemonic: ☁️ Clouds and Smoke 💨
🔗 The Breakdown:
- Clouds → Colloids show the Tyndall Effect (light scatters through fog/clouds)
- Smoke → Suspensions show the Tyndall Effect (light beam visible in smoky room)
- NOT in True Solutions → Clear sugar water shows NO Tyndall Effect (light passes straight through)
💡 Memory Tip: When sunlight passes through clouds or a room fills with smoke, you can SEE the light beam path. That's Tyndall Effect! In a clear solution (like pure water), you can't see the light beam. Real-life observation = best memory!
How Solubility Changes with Temperature
What needs to be memorized: Opposite behaviors of solids vs gases when temperature increases
Mnemonic: 📈 Solid goes HIGH, 📉 Gas says BYE
🔗 The Breakdown:
- Solid goes HIGH → Solubility of solid solutes INCREASES with temperature (sugar dissolves better in hot chai than cold water)
- Gas says BYE → Solubility of gas solutes DECREASES with temperature (CO₂ escapes from hot fizzy drinks, less in hot water)
💡 Memory Tip: Heat sugar → it dissolves more. Heat cold drink → bubbles escape! Opposite behaviors, easy to remember with this rhyme. Solid UP, Gas AWAY!
Key Properties of Suspensions (Identifying Features)
What needs to be memorized: Three main characteristics that identify a suspension mixture
Mnemonic: Visible, Settle, Filter (VSF)
🔗 The Breakdown:
- Visible → Particles are visible to the naked eye (you can see them!)
- Settle → Particles settle to the bottom when left undisturbed (muddy water clears over time)
- Filter → Can be separated easily by filtration (filter paper traps the particles)
💡 Memory Tip: Think of muddy water: you can SEE the mud particles, they SETTLE at the bottom over time, and you can FILTER them out with filter paper. That's your test for a suspension!
Main Separation Methods for Homogeneous Mixtures
What needs to be memorized: Three primary techniques to separate solutions (solids from liquids, or liquids from liquids)
Mnemonic: 🍬 Candy, Distill, Pigments 🎨
🔗 The Breakdown:
- Candy → Crystallization (like making rock candy crystals from sugar solution by cooling)
- Distill → Distillation (heating and condensing liquids with different boiling points)
- Pigments → Paper Chromatography (separating colors/dyes from plant extracts)
💡 Memory Tip: Three visual examples: Rock candy formation (crystallization), distilled water you buy (distillation), and colored spots appearing on paper (chromatography). The examples themselves teach the method!
How to Choose Separation Method for Heterogeneous Mixtures
What needs to be memorized: Decision-making tool to identify the right separation method for different scenarios
Mnemonic: 🛢️ Oil, Mud, Camphor 🔥
🔗 The Breakdown:
- Oil (+ Water) → Separating Funnel (immiscible liquids form layers, drain the denser liquid)
- Mud (+ Water) → Coagulation or Centrifugation (fine suspended particles → add alum to clump, or spin to separate)
- Camphor (+ Sand) → Sublimation (camphor vaporizes/vanishes directly, sand remains)
💡 Memory Tip: Use real-life examples! If you're separating things like oil and water, use funnel. Like mud and water, use coagulation/centrifugation. Like camphor from sand, use sublimation. The example guides the method!
Particle Size Classification: Solution vs Colloid vs Suspension
What needs to be memorized: Size ranges of particles in each type of mixture and their characteristics
Mnemonic: 🥘 Sauce, Curd, Sand
🔗 The Breakdown:
- Sauce → Solution (< 1="" nm)="" -="" clear,="" transparent,="" like="" gravy="">
- Curd → Colloid (1-1000 nm) - Semi-solid, creamy texture, like dahi/yogurt
- Sand → Suspension (> 1000 nm) - Visible particles, grainy texture, like sand grains
💡 Memory Tip: This is PERFECT for Indian students! These are three things every Indian knows intimately. Sauce is clear (smallest particles). Curd is creamy and semi-solid (medium particles). Sand is grainy and visible (largest particles). Size increases from left to right, just like their consistency increases!
Size Order Reminder: 1 nano-meter is the boundary for Solution. The range 1-1000 nm is Colloid's "sweet spot." Above 1000 nm, particles are definitely Suspension-sized and visible to naked eye.
⚡ Quick Mnemonic Reference Card
✅ Tips for Maximum Retention
- Create visual connections: Draw pictures of Clouds & Smoke for Tyndall Effect, or Sauce-Curd-Sand in order of thickness!
- Say them aloud: "Solid goes HIGH, Gas says BYE" is easy to remember when you rhyme it out loud 3 times.
- Use during revision: When studying separation methods, first think OMC (Oil, Mud, Camphor), then recall which method goes with each.
- Create stories: Imagine yourself making rock candy (Crystallization), boiling water (Distillation), and looking at colorful dyes (Chromatography) - real scenarios stick better!
- Test yourself: Cover the breakdown, see only the mnemonic, and try to recall all the details. This builds strong memory pathways.





