First Law of Thermodynamics
First Law of Thermodynamics
The First Law of Thermodynamics states that heat is a form of energy and that in any thermodynamic process the total energy is conserved. Energy cannot be created or destroyed; it can only be transferred between a system and its surroundings or converted from one form to another.
Mathematical statement for a closed system
For a closed system undergoing a process, the First Law is commonly written as a balance between heat supplied, work done and change in internal energy:
ΔU = Q - W
where:
- ΔU is the change in internal energy of the system;
- Q is the heat supplied to the system (positive when heat flows into the system);
- W is the work done by the system on the surroundings (positive when the system does work).
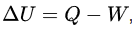
Alternate, general energy balance
For processes where several heat and work interactions occur, the net heat supplied equals the net change in the system energy plus the net work:
Qnet = ΔE + W
For example, when separate heat and work interactions are present, they may be written as
Q2 + Q3 - Q1 = ΔE + W2 + W3 - W1 - W4
Properties of energy and thermodynamic functions
- Energy is a point function (state function): its value depends only on the state of the system, not on the path taken to reach that state.
- Energy is an extensive property (it scales with the size or mass of the system). Corresponding intensive quantities (for example, internal energy per unit mass) are independent of system size.
- For an ideal gas, the internal energy depends only on temperature. Similarly, the enthalpy of an ideal gas depends only on temperature.
Heat transfer at constant volume
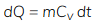
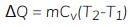
Specific heat at constant volume is denoted by Cv.
For a closed system with constant volume (no boundary work), all heat supplied appears as change in internal energy. For a mass m of substance,
ΔU = Qv = m ∫ Cv dT
- At constant volume, the heat transfer increases the internal energy of the system.
Heat transfer at constant pressure
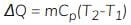
Specific heat at constant pressure is denoted by Cp.
When a system undergoes heating at constant pressure the heat supplied changes the enthalpy H of the system. For mass m:
ΔH = Qp = m ∫ Cp dT
- At constant pressure, the heat transfer increases the enthalpy of the system.
Isolated system and special cases
For an isolated system there is no exchange of heat or work with the surroundings. Therefore:
dQ = 0, dW = 0 ⇒ dE = 0
- The total energy of an isolated system remains constant.
- Perpetual Motion Machine of the First Kind (PMM-1): a device that would continuously produce work without any input of energy would violate the First Law. Such a machine is impossible.
- The First Law is essentially the statement of the principle of conservation of energy in thermodynamics.
Sign conventions and common remarks
- Be careful with sign convention: the form ΔU = Q - W assumes W is work done by the system. If work done on the system is used as positive, the equation becomes ΔU = Q + W.
- Work includes boundary (PdV) work and other forms such as shaft work, electrical work, etc. The form of W used in an energy balance must include all mechanical and non-mechanical work interactions.
- The First Law does not place any restriction on the direction of processes; it only requires energy conservation. Irreversibility and direction are addressed by the Second Law.
Simple illustrative example
Example (rigid vessel): A rigid container (constant volume) holds a gas. Heat Q is supplied. Find the change in internal energy.
Sol.
Because the vessel is rigid, boundary work is zero.
Therefore the First Law simplifies to:
ΔU = Q
If the gas has mass m and specific heat at constant volume Cv, then for a temperature change from T1 to T2:
ΔU = m ∫T₁T₂ Cv dT
If Cv is constant over the temperature range, this reduces to:
ΔU = m Cv (T₂ - T₁)
Applications and examples of use
- Energy balance for engines, compressors, turbines and pumps.
- Heating and cooling calculations in boilers and heat exchangers.
- Analysis of closed processes (isothermal, isobaric, isochoric, adiabatic) and cyclic devices such as heat engines and refrigerators.
- Estimation of work output or required heat input in thermodynamic cycles using energy conservation.
Summary
The First Law of Thermodynamics is the conservation of energy applied to thermodynamic systems. For a closed system, the change in internal energy equals heat supplied minus work done by the system. At constant volume heat changes internal energy; at constant pressure heat changes enthalpy. For an isolated system total energy remains constant, and any device that would produce work without energy input (PMM-1) is impossible.
FAQs on First Law of Thermodynamics
| 1. What is the First Law of Thermodynamics in mechanical engineering? |  |
| 2. How does the First Law of Thermodynamics apply to mechanical engineering? |  |
| 3. What are the key components of the First Law of Thermodynamics? |  |
| 4. How can the First Law of Thermodynamics be applied to an engine? |  |
| 5. What are some practical applications of the First Law of Thermodynamics in mechanical engineering? |  |







