Properties of Pure Substance
Introduction
A pure substance is a material that has a homogeneous chemical composition throughout its mass and consists of a single component system. A pure substance may exist in one or more phases (solid, liquid, vapour). The thermodynamic behaviour of a pure substance is described by its pressure (P), temperature (T), specific volume (v) and other properties such as enthalpy (h), internal energy (u) and entropy (s).
Saturation and Phase Change
- Saturation state: A saturation state is a state at which a change of phase can occur without a change in pressure or temperature (for example, liquid ↔ vapour at the saturation temperature for a given pressure).
- Degree of superheat: The difference between the temperature of a superheated vapour and the temperature of the saturated vapour at the same pressure.
- Sublimation: Direct transformation of a solid to vapour (solid → vapour).
- Deposition (ablimation): Direct transformation of vapour to solid (vapour → solid). Some texts use the term ablimation for this process.
Critical Point
- Critical temperature (Tc): At the critical temperature a liquid and its vapour become identical in properties and the distinct liquid-vapour boundary disappears. Above this temperature a vapour cannot be liquefied by any increase of pressure.
- Critical pressure (Pc): The minimum pressure required to liquefy a vapour at the critical temperature.
- The line that joins the critical points at varying conditions is often called the critical line on two- and three-dimensional phase diagrams.
Triple Point
- Triple point: The unique temperature and pressure at which solid, liquid and vapour phases of a substance coexist in equilibrium.
- At the triple point for a pure substance the number of degrees of freedom is zero (see Gibbs phase rule later).
- Common reference convention: at the triple point many property tables choose the triple point as a convenient reference. For such references it is sometimes stated that internal energy = 0 and entropy = 0 while enthalpy > 0 (these are reference choices, not absolute physical requirements).
Phase Diagrams and Curves
- Phase diagrams (P-T, P-v, T-v, h-s and Mollier (h-s) diagrams) show regions of solid, liquid and vapour and the lines of equilibrium between phases (fusion, vaporization, sublimation).
- The slope of the fusion (solid-liquid) curve is positive for most substances; however, for water it is negative because water expands on freezing (ice is less dense than liquid water).
- The slopes of the vaporization and sublimation curves are positive for all substances.
- The Clapeyron equation gives the slope of the coexistence (phase-change) curve on a P-T diagram:
Clapeyron equation: dp/dT = L / (T · Δv)
where L is the latent heat (per unit mass) for the phase change and Δv is the change in specific volume between the two phases (for example vg - vf for vaporization).
Examples - Properties of Water (values given in input)
- Critical pressure, Pc: 221.2 bar
- Critical temperature, Tc: 374.15 °C
- Critical specific volume, vc: 0.00317 m3/kg
- Triple point pressure: 4.58 mm Hg
- Triple point temperature: 273.16 K
Mollier Diagram (h-s) and Dryness Lines
- The Mollier diagram (h-s diagram) is frequently used for moist vapour/steam calculations and shows constant-pressure lines, constant-temperature lines, and lines of constant dryness fraction (quality).
- Dryness fraction (x): The mass fraction of vapour in a two-phase (mixture) system. If mv is mass of vapour and ml is mass of liquid then
x = mv / (mv + ml)
- For saturated liquid, x = 0.
- For saturated vapour, x = 1.
- Dryness-quality lines (constant x) in P-V, h-s and other diagrams typically originate from the critical line and span the two-phase dome.
Properties of Two-Phase Mixture (Mixture Relations)
- For a two-phase mixture at given pressure or temperature the specific properties can be expressed in terms of the saturated liquid and saturated vapour properties and the dryness fraction x.
- Notation:
v = specific volume of the mixture
vf = specific volume of saturated liquid
vg = specific volume of saturated vapour
vfg = vg - vf
h = specific enthalpy of the mixture
hf = specific enthalpy of saturated liquid
hg = specific enthalpy of saturated vapour
hfg = hg - hf
s = specific entropy of the mixture
sf = specific entropy of saturated liquid
sg = specific entropy of saturated vapour
sfg = sg - sf
- Mixture relations (linear mixing using dry mass fraction x):
v = (1 - x)·vf + x·vg = vf + x·vfg
h = (1 - x)·hf + x·hg = hf + x·hfg
s = (1 - x)·sf + x·sg = sf + x·sfg
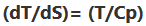
Degrees of Freedom and Gibbs Phase Rule
- The Gibbs phase rule for a system with C components and P phases is: F = C - P + 2, where F is the number of degrees of freedom (intensive properties that can be changed independently, e.g., P and T).
- For a pure substance (C = 1): F = 3 - P.
- Consequences for a one-component system:
- If P = 1 (single-phase, e.g. a gas), F = 2: two independent intensive properties (such as P and T) must be specified to fix the state.
- If P = 2 (two phases in equilibrium, e.g. saturated liquid + vapour), F = 1: only one intensive property (either P or T) is independent along the saturation line.
- If P = 3 (triple point: solid + liquid + vapour in equilibrium), F = 0: the temperature and pressure are fixed (unique) for the triple point.
Measurement of Dryness Fraction - Throttling Calorimeter
- Throttling calorimeter: An apparatus used to determine the dryness fraction (quality) of a wet steam (two-phase) sample by throttling (an irreversible adiabatic expansion through a valve or porous plug) the mixture to a lower pressure where the outlet is single-phase vapour.
- Throttling process is adiabatic and irreversible, and under typical assumptions it is approximately isenthalpic (hin = hout), i.e., enthalpy remains constant across the throttle if no heat transfer and no work interaction other than flow work occur.
- Because direct measurement of quality in a two-phase mixture is difficult, the mixture is throttled to a condition where all liquid flashes to vapour (or where the downstream condition is a known saturated vapour); the downstream temperature and pressure are measured and used, together with energy relations, to determine the upstream dryness fraction.
Useful Remarks and Practical Points
- Dryness fraction may be determined from enthalpy measurements using: x = (h - hf) / hfg.
- Similarly, x = (v - vf) / vfg or x = (s - sf) / sfg when the corresponding properties are used.
- Dryness quality lines on diagrams such as h-s or P-v help visualise two-phase mixture behaviour and are useful for steam-engine, turbine and boiler calculations.
Summary
The properties of a pure substance and its phase behaviour are essential for thermodynamic analysis. Key concepts include saturation, critical point, triple point, phase diagrams (including the Mollier h-s diagram), dryness fraction and the mixture relations that allow calculation of specific volume, enthalpy and entropy for two-phase mixtures. The Gibbs phase rule gives the degrees of freedom for a given number of phases. Practical measurement techniques such as the throttling calorimeter make it possible to determine the quality of wet steam for engineering applications.
FAQs on Properties of Pure Substance
| 1. What is a pure substance in mechanical engineering? |  |
| 2. What are the properties of a pure substance? |  |
| 3. How do specific volume and specific entropy relate to pure substances? |  |
| 4. What is the significance of specific internal energy in pure substances? |  |
| 5. How does specific enthalpy differ from specific internal energy in pure substances? |  |






