Some Important Carbon Compounds – Ethanol And Ethanoic Acid
In this document for Class 10 Science, specifically covering the chapter "Carbon and Its Compounds", we explore two important carbon compounds: ethanol and ethanoic acid. Both are commercially important and commonly encountered in daily life. The presentation below explains their preparation, physical and chemical properties, uses, and safety aspects in a clear, syllabus-aligned way for learners.

What is Ethanol?
Ethanol, also called ethyl alcohol, has the molecular formula C2H5OH. It belongs to the class of compounds called alcohols and is the second member of the homologous series of alcohols. It contains a hydroxyl (-OH) functional group attached to an sp3 carbon atom.
Preparation of Ethanol
Ethanol is prepared on a commercial scale by fermentation of sugar (molasses). Fermentation is allowed to take place at 298- 303 K in the absence of air.
Ethanol is oxidised to ethanoic acid using oxidising agents such as alkaline potassium permanganate or acidified potassium dichromate.Ethanol is oxidised to ethanoic acid using oxidising agents such as alkaline potassium permanganate or acidified potassium dichromate.
Try yourself: Which of the following is the common name of the organic compound ethyl alcohol?
Physical Properties of Ethanol
- Appearance and odour: Ethanol is a colourless liquid with a characteristic pleasant smell and a burning taste.
- Boiling point: 78.1 °C (approximately).
- Freezing point (melting point): about -114.1 °C (ethanol freezes at a very low temperature).
- Density: Ethanol is lighter than water; its density is about 0.789 g mL-1 at room temperature (≈293 K).
- Solubility: Ethanol is miscible with water in all proportions because it can form hydrogen bonds with water molecules.
- Spirit grades: Ethanol containing about 5% water (i.e., ≈95% ethanol) is called rectified spirit. Ethanol that is essentially water-free is called absolute alcohol.
- Electrical conductivity and action on litmus: Ethanol is a covalent, neutral compound; it does not conduct electricity in the pure state and does not change the colour of litmus.
 Ethanol is a colurless liquid
Ethanol is a colurless liquidChemical Properties of Ethanol
- Combustion: Ethanol burns in oxygen with a blue flame to give carbon dioxide and water, releasing heat. The combustion equation is:
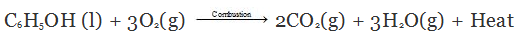
- Reaction with sodium: Ethanol reacts with sodium metal to form sodium ethoxide and hydrogen gas:
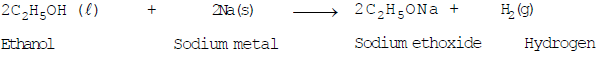
- Esterification (reaction with ethanoic acid): Ethanol reacts with ethanoic acid in the presence of concentrated sulfuric acid to form an ester, ethyl ethanoate, and water. This reversible reaction is called esterification and is used to prepare fruity-smelling esters:
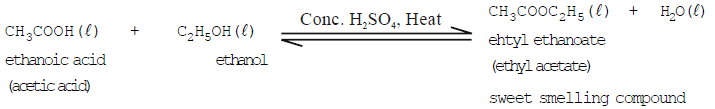
- Dehydration (formation of ethene): Ethanol when heated with concentrated sulfuric acid (a dehydrating agent) gives ethene and water:
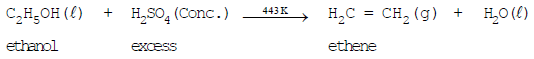
Uses and Examples of Ethanol
- Alcoholic beverages: Ethanol is the intoxicating component of beer, wine and spirits consumed for recreational purposes. It is produced by fermentation and then processed to desired strengths.
- Fuel: Ethanol is used as a biofuel or petrol additive (e.g., in blends such as E10, E85) to reduce net emissions and reliance on fossil fuels.
- Disinfectants and sanitizers: Ethanol is an effective antiseptic and is widely used in hand sanitizers and disinfectant solutions.
- Solvent: Ethanol is a common solvent in pharmaceuticals, cosmetics, paints, varnishes and perfumery.
- Extraction of plant oils: Ethanol is used to extract essential oils and other plant constituents.
- Preservative: Dilute ethanol solutions are used to preserve biological specimens and some food products.
 Ethanol in Paint
Ethanol in PaintHarmful Effects of Alcohols
- Consumption of dilute ethanol in small amounts causes intoxication or drunkenness; repeated or heavy use leads to addiction and harmful effects on health.
- Large intake of ethanol depresses the central nervous system causing impaired coordination, mental confusion, drowsiness and in extreme cases stupor or coma.
- Chronic alcohol consumption damages the liver (e.g., fatty liver, hepatitis, cirrhosis) and harms many other organs.
Denatured Alcohol
Denatured alcohol is ethanol to which small amounts of poisonous or foul-tasting substances (for example, methanol, pyridine or copper sulfate) are added to make it unfit for drinking. Denaturing prevents misuse of industrial ethanol while allowing it to be used as a solvent, fuel, or cleaning agent.
Industrial alcohol is often coloured (for example blue) so that it can be recognised easily.
Harmful Effects of Denaturants (Methanol)
- Methanol is highly poisonous; ingestion of even small quantities can be fatal.
- Methanol is metabolised in the body to formaldehyde (methanal) and formic acid, which damage tissues and may coagulate protoplasm.
- Methanol poisoning affects the optic nerve and can cause permanent blindness.
What is Ethanoic Acid?
Ethanoic acid, commonly called acetic acid, is a carboxylic acid with formula CH3COOH. A 5-8% solution of ethanoic acid in water is called vinegar and is used for preserving and flavouring foods such as pickles.
Try yourself: Which of the following statements about ethanoic acid is correct?
Physical Properties of Ethanoic Acid
- At ordinary temperatures ethanoic acid is a colourless liquid with a strong, pungent smell and a sour taste.
- Boiling point: ≈ 391 K (≈ 118 °C).
- Density: ≈ 1.08 g mL-1 at 273 K (heavier than water).
- It is miscible with water due to hydrogen bonding.
- On cooling to about 289.6 K, it solidifies into ice-like crystals; the nearly pure liquid at room temperature is called glacial acetic acid.
- It dissolves many organic substances such as iodine and some sulphur compounds.
- In non-polar solvents (e.g., benzene) ethanoic acid molecules tend to form dimers via hydrogen bonding.
Chemical Properties of Ethanoic Acid
- Esterification (reaction with alcohols): Ethanoic acid reacts with ethanol in the presence of concentrated sulfuric acid to form ethyl ethanoate (an ester) and water. Esters are often pleasant-smelling and are used in perfumes and flavourings.
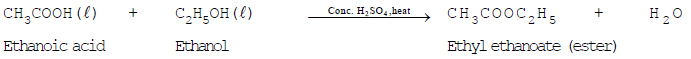
Note: Esters can be hydrolysed back to the parent alcohol and carboxylic acid. Acidic hydrolysis reverses the esterification reaction; alkaline hydrolysis is called saponification and is used industrially to produce soaps from fats and oils.
Example:
(i) Ethyl ethanoate on acid hydrolysis gives ethanoic acid and ethanol.
CH3COOC2H5(l) + H2O(l) → CH3COOH(aq.) + C2H5OH
(ii) Hydrolysis of ester in the presence of a base (alkali) is called "Saponification reaction".
Note: Alkaline hydrolysis of higher esters is used in the manufacture of soaps.
- Reaction with carbonates and bicarbonates: Ethanoic acid reacts with sodium carbonate (Na2CO3) and sodium hydrogen carbonate (NaHCO3) to give carbon dioxide gas, water and the corresponding sodium salt (sodium ethanoate). The rapid evolution of CO2 is characteristic of carboxylic acids.
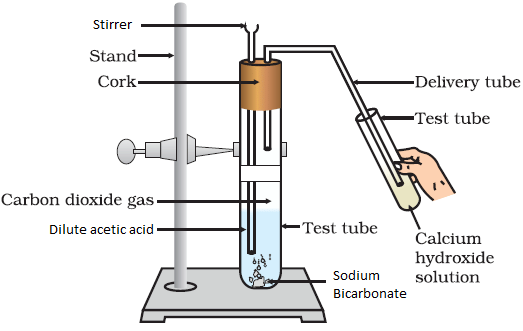 Reaction of Sodium Bicarbonate with Acetic Acid
Reaction of Sodium Bicarbonate with Acetic Acid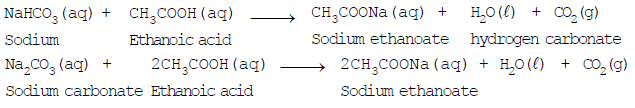
Note: The reactions of ethanoic acid with NaOH, NaHCO3, Na2CO3 and active metals demonstrate that the hydrogen of the carboxyl (-COOH) group is acidic in nature.
Uses and Examples of Ethanoic Acid
Ethanoic acid (acetic acid) has many uses across industries and in daily life:

- Food industry: Used as a preservative, acidulant and flavouring agent (vinegar) in foods such as pickles, sauces and dressings.
- Vinegar production: Ethanoic acid is the main component of vinegar, giving it its sour taste and preservative properties.
- Cleaning and disinfecting: Dilute acetic acid solutions are used as household cleaners to remove grease and mineral deposits and act as mild disinfectants.
- Pharmaceuticals: Used as a reagent and intermediate in the manufacture of medicines and chemical preparations.
- Textile industry: Used in dyeing and finishing processes to improve dye uptake and fabric properties.
- Chemical synthesis: A versatile precursor for producing esters, solvents, and polymers (e.g., cellulose acetate).
- Laboratory use: Common reagent, solvent and pH adjuster in many chemical procedures.
Safety and Handling
- Both ethanol and ethanoic acid should be handled with care. Ethanol is flammable; keep away from open flames and strong oxidisers.
- Concentrated ethanoic acid (glacial acetic acid) is corrosive and can cause burns; use appropriate protective equipment (gloves, goggles, lab coat) and work in a fume hood.
- Do not ingest industrial or denatured alcohol. Methanol contamination is toxic and can cause blindness or death.
- Store chemicals in properly labelled containers away from incompatible substances and heat sources.
Summary: Ethanol (C2H5OH) and ethanoic acid (CH3COOH) are fundamental organic compounds taught in the chapter on carbon compounds. Ethanol is mainly produced by fermentation and used as a beverage component, solvent, fuel and disinfectant. Ethanoic acid is a carboxylic acid used as vinegar, a chemical reagent and in many industrial processes. Their characteristic reactions-combustion, reaction with metals, esterification, dehydration (ethanol) and acid reactions with carbonates and alcohols (ethanoic acid)-illustrate core concepts of functional groups, reactivity and applications in everyday life.
FAQs on Some Important Carbon Compounds – Ethanol And Ethanoic Acid
| 1. What is the chemical formula of ethanol and how is it commonly produced? |  |
| 2. What are the main physical properties of ethanol? |  |
| 3. What are the primary uses of ethanol in everyday life? |  |
| 4. What is the chemical structure of ethanoic acid and how is it related to ethanol? |  |
| 5. What are the common applications of ethanoic acid in industry and household products? |  |






