Dual Nature of Matter & Heisenberg's Uncertainty Principle
De Broglie's Hypothesis and Dual Nature of Matter
- de Broglie proposed that particles such as electrons show dual nature: they behave both as material particles and as waves. This is analogous to the wave-particle duality already known for light.
- The idea that matter has a wave character led to the development of the wave-mechanical model of the atom, in which electrons (and, in general, material particles) possess associated waves called matter waves or de Broglie waves when they are in motion.
 Dual Nature of Matter
Dual Nature of MatterDerivation of de Broglie's Relationship
Consider the known relations for a photon. Its energy is given by E = hν, where h is Planck's constant and ν is frequency.
The momentum of a photon can be expressed as p = E/c and, using the relation between wavelength and frequency for electromagnetic radiation, λ = c/ν, one obtains p = h/λ.
By analogy, de Broglie extended this relation to material particles and proposed that a particle of momentum p has an associated wavelength λ given by the relation
λ = h / p
where p = mv for a particle of mass m moving with velocity v. This relation is known as the de Broglie relation.
Experimental Verification
The wave nature of electrons was experimentally confirmed by Davisson and Germer. They observed diffraction of an electron beam by a nickel crystal similar to the diffraction of X-rays by crystals.
The wavelengths of electrons determined from the diffraction experiments agreed with the wavelengths calculated using the de Broglie relation.
Derivation of Bohr's Quantisation of Angular Momentum from de Broglie Hypothesis
de Broglie argued that for an electron to form a stable standing wave around the nucleus, the associated wave must be in phase after one complete revolution. Therefore the circumference of the allowed orbit must contain an integral number of wavelengths:
That is, 2πr = nλ, where n is an integer (the principal quantum number).
Substituting λ = h / p and p = mv gives
mvr = nh / 2π = nħ
which is exactly Bohr's postulate: the angular momentum of an electron in a permitted orbit is an integral multiple of ħ = h/2π.
 Quantization of Angular Momentum of Electron
Quantization of Angular Momentum of ElectronCalculation of de Broglie Wavelength when Kinetic Energy is E
For a non-relativistic particle of mass m and kinetic energy E, kinetic energy is related to speed by
2E = mv²
The momentum p satisfies
p² = m²v² = 2mE
Therefore
p = √(2mE)
Substituting into de Broglie relation gives
λ = h / p = h / √(2mE)
If a charged particle of charge q is accelerated from rest through a potential difference V, the gain in kinetic energy is qV. For an electron (charge = e) accelerated through a potential V volts,
hence for electrons,
Example
Ex. Calculate λdB of an electron having K.E. 3 eV.
Sol.
Use the relation for an electron accelerated so that its kinetic energy E = 3 eV = 3 × 1.602 × 10⁻¹⁹ J.
For a non-relativistic electron, p = √(2mE).
Substitute m(electron) = 9.109 × 10⁻³¹ kg and E = 3 × 1.602 × 10⁻¹⁹ J to find p.
Compute the de Broglie wavelength λ = h / p with h = 6.626 × 10⁻³⁴ J·s.
Heisenberg Uncertainty Principle
- The Heisenberg Uncertainty Principle states that the position and momentum of a particle cannot be simultaneously measured with arbitrarily high precision.
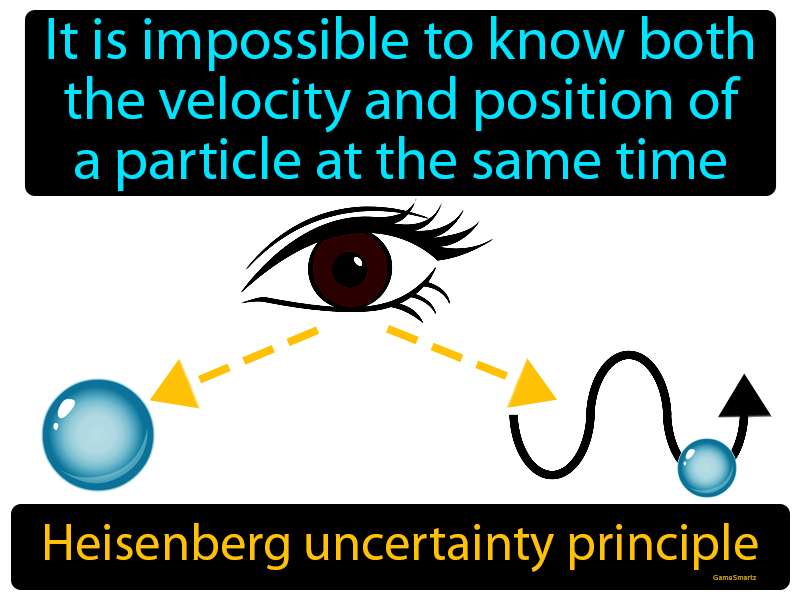 Heisenberg's Uncertainty Principle
Heisenberg's Uncertainty Principle- There is a fundamental lower bound for the product of the uncertainties of these two observables: Δx · Δp ≥ ħ/2, where ħ = h/2π.
- There is an analogous relation between energy and time: ΔE · Δt ≥ ħ/2. This relation sets a limit on how precisely energy can be defined for a state that exists only for a limited time.
Mathematical form (common forms):
- Δx · Δp ≥ ħ/2
- ΔE · Δt ≥ ħ/2
- The uncertainty principle implies that confining a particle to a very small region of space increases the minimum possible uncertainty in its momentum, and therefore increases the minimum kinetic energy associated with confinement.
- The energy required to confine particles within an atomic or nuclear region comes from the fundamental forces: the electromagnetic force confines electrons around the nucleus and the strong nuclear force confines nucleons in the nucleus.
- The value of Planck's constant, which appears in the uncertainty relations, sets the scale for how small these confinement regions can be; this, together with the strengths of the forces, determines the sizes of atoms and nuclei.
Example Illustrating the Uncertainty Principle
- To understand the principle by analogy: measuring the thickness of a sheet of paper using an unmarked metre rule is imprecise because the measuring unit (1 metre divided into unmarked units) is too large compared with the thickness. You need a tool with finer scale to measure thickness accurately.
- For electrons, to locate an electron precisely we must use probe radiation whose wavelength is comparable to or smaller than the spatial resolution required. A photon with small wavelength carries large momentum; on interacting with the electron it will impart an uncertain change in the electron's momentum.
- Thus the more precisely we determine the position of the electron (using shorter-wavelength-higher-momentum-photons), the more we disturb its momentum during measurement, and so the uncertainty in momentum increases. Conversely, if we measure momentum precisely, position becomes more uncertain. This trade-off is the physical content of the uncertainty principle.
Significance of the Heisenberg Uncertainty Principle
- The principle rules out the possibility of assigning exact classical trajectories to microscopic particles such as electrons; one cannot know both position and momentum simultaneously with arbitrary precision.
- In classical mechanics, knowing position and velocity at an instant and the forces acting allows prediction of future motion (trajectory). For microscopic particles, uncertainty prevents such precise knowledge and hence precise trajectories.
- The effect is important for microscopic systems (electrons, atoms, nuclei) and is negligible for macroscopic objects because the value of ħ is extremely small on everyday scales.
Summary: The de Broglie hypothesis connects particle momentum and wavelength via λ = h/p, giving a wave nature to matter and leading to quantisation conditions such as Bohr's angular-momentum rule. The Heisenberg Uncertainty Principle places fundamental limits on simultaneous knowledge of conjugate pairs (position-momentum, energy-time), explains why precise classical trajectories are not meaningful for microscopic particles and plays a central role in the wave-mechanical (quantum) description of matter.
FAQs on Dual Nature of Matter & Heisenberg's Uncertainty Principle
| 1. What is De Broglie's hypothesis? |  |
| 2. How is De Broglie's relationship derived? |  |
| 3. What is the Heisenberg uncertainty principle? |  |
| 4. How does the dual nature of matter relate to the Heisenberg uncertainty principle? |  |
| 5. What are some applications of the Heisenberg uncertainty principle? |  |






