Group-13 Elements: Boron Family
What are Group 13 Elements?
The Group 13 elements form the first group of the p-block of the periodic table and are commonly called the boron family. Their valence shell electronic configuration is ns2 np1, which gives them characteristic chemical and physical behaviour across the group.
 Group 13 Elements
Group 13 ElementsThe principal members of Group 13 are:
- Boron (B)
- Aluminium (Al)
- Gallium (Ga)
- Indium (In)
- Thallium (Tl)
Aluminium is the most abundant metal of the group and the third most abundant element in the Earth's crust.
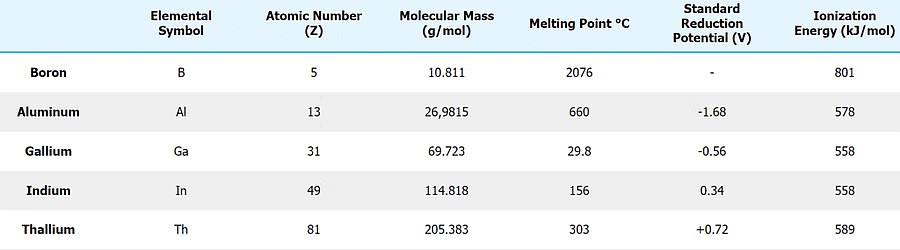
Physical Properties and Periodic Trends
Electronic Configuration
The general valence electronic configuration for Group 13 elements is ns2 np1. This single p-electron and paired s-electrons control many properties of the group.

Atomic and Ionic Radii
Compared with the corresponding alkaline earth metals, Group 13 elements have smaller sizes because of a higher effective nuclear charge (Zeff).

- Atomic radii generally increase down the group from B → Al → Ga → In → Tl.
- Gallium shows an anomaly: Ga has a smaller atomic radius than expected due to poor shielding by filled 3d electrons, causing a stronger attraction of valence electrons by the nucleus.
- Ionic radii increase regularly from B3+ to Tl3+.
Density
Density increases regularly on going down the group from boron to thallium.
Melting and Boiling Points
Melting and boiling points for Group 13 elements are generally higher than those of group 2 elements. Trends are influenced by bonding and structure.
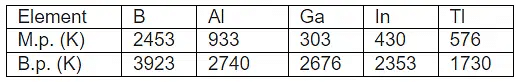
- Boron has a very high melting point due to its three-dimensional covalent network structure.
- Gallium shows a low melting point (it melts just above room temperature) because metallic Ga exists as Ga2 molecules in the solid state; it remains liquid up to high temperatures and finds specialised uses (for example, in high-temperature thermometry).
Try yourself: Which of the following element has the highest melting point?
Ionisation Enthalpy (IE)
Initial ionisation enthalpies of Group 13 elements are generally lower than those of corresponding alkaline earth metals because removal of the outer p-electron is relatively easier.
 Trends & Properties of Group 13 elements
Trends & Properties of Group 13 elements- Ionisation enthalpy decreases down the group in general, but anomalies occur: IE decreases from B → Al, then increases slightly for Ga because d-electrons provide poor shielding, making the outer electrons more tightly held; the trend then decreases for In and rises again for Tl due to relativistic and inert-pair effects.
- Thallium (Tl) often does not follow the simple monotonic trend because of the increasing importance of relativistic effects and the inert pair effect.
Oxidation States
- Boron and aluminium predominantly show the +3 oxidation state.
- Gallium, indium and thallium can show both +3 and +1 oxidation states.
- Inert pair effect is the reluctance of the s-electrons of the valence shell to take part in bonding. It occurs due to poor shielding of the ns2 - electrons by the intervening d and f - electrons. It increases down the group and thus, the lower elements of the group exhibit lower oxidation states. The ns2 electrons become less likely to participate in bonding, so the stability of the +1 oxidation state increases: Ga < In < Tl.the ns2 electrons become less likely to participate in bonding, so the stability of the +1 oxidation state increases: Ga < In < Tl.
Metallic (Electropositive) Character
- Group 13 elements are less electropositive than alkaline earth metals because of smaller size and higher ionisation enthalpies.
- Electropositive character increases from B → Al, then is influenced by d and f electrons; overall metallic character increases down the group but shows complexities for Ga and Tl due to poor shielding.
Reducing Character
- Reducing character generally decreases down the group for the M3+/M pair because electrode potentials increase in value.
- Order of reducing strength: Al > Ga > In > Tl.
Complex Formation
Because of their relatively small ionic sizes and higher charge density (especially in +3 state), Group 13 elements tend to form complexes more readily than s-block elements.
Nature of Compounds
- Boron forms only covalent compounds due to its small size and high ionisation enthalpy.
- Aluminium forms both covalent and ionic compounds.
- Going down the group the tendency to form ionic compounds increases (Tl forms more ionic compounds); anhydrous AlCl3 is covalent and exists as a dimer in the vapour/solid state under many conditions.
Chemical Properties
Action of Air
- Crystalline boron is comparatively unreactive; amorphous boron reacts more readily. At high temperatures boron burns in oxygen:
4B + 3O2 → 2B2O3
and reacts with nitrogen to give boron nitride:
2B + N2 → 2BN - Aluminium is protected from further oxidation by a thin passivating layer of Al2O3:
4Al + 3O2 → 2Al2O3 - Thallium is more reactive than Ga and In in some conditions because the +1 state stabilises its oxides:
4Tl + O2 → 2Tl2O
Reaction with Nitrogen
Boron and aluminium react with nitrogen only at high temperatures to form nitrides (for example, BN and AlN).
Action of Water
- Boron and aluminium do not react with water under normal conditions; however, amalgamated aluminium (Al-Hg) reacts with water releasing hydrogen:
2Al(Hg) + 6H2O → 2Al(OH)3 + 3H2 + 2Hg - Gallium and indium do not react with pure cold or hot water; thallium forms a surface oxide layer that can inhibit reaction.
Reaction with Alkalies
- Boron dissolves in alkalies to give borates.
- Aluminium reacts with alkali solutions, liberating hydrogen:
2Al + 2NaOH + 6H2O → 2Na[Al(OH)4] + 3H2

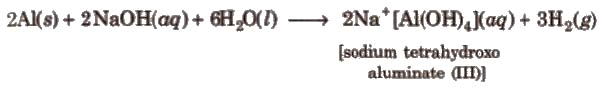
Reaction with Carbon
Aluminium forms aluminium carbide, Al4C3, which reacts with water to give methane.
Hydrides
- Group 13 elements do not combine directly with H2 easily; hydrides are prepared by indirect routes.
- Boron hydrides (boranes) such as B2H6 are well known and are typically very reactive; they burn in oxygen:
B2H6 + 3O2 → B2O3 + 3H2O - Boranes hydrolyse in water:
B2H6 + 6H2O → 2H3BO3 + 6H2 - The stability of hydrides decreases down the group because the M-H bond strength decreases from B → Tl.
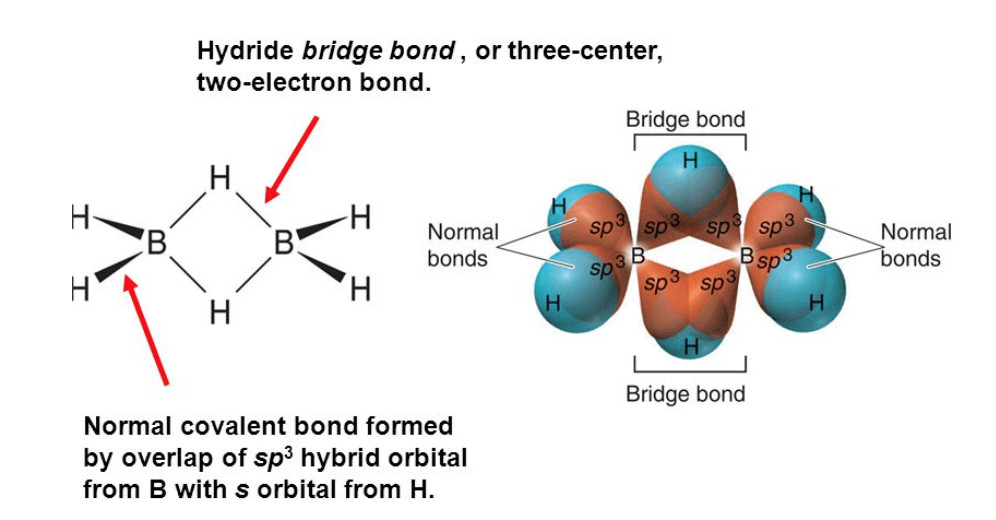
Structure of diborane
- BH3 is electron deficient and does not exist free; it dimerises to form B2H6 (diborane).
- In diborane each boron is sp3 hybridised. There are four terminal B-H bonds and two bridging B-H-B bonds. The bridges are three-centre two-electron (3c-2e) bonds (banana bonds) above and below the plane of the molecule.

Try yourself: Diborane is a ____________
Oxides
- Except for thallium, Group 13 elements form oxides of general formula M2O3 on heating in oxygen.
- Thallium forms a more stable monovalent oxide, Tl2O, than the trivalent oxide Tl2O3, due to the inert pair effect.
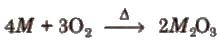
Nature of Oxides and Hydroxides
- Boric acid B(OH)3 (or H3BO3) is soluble in water; most other hydroxides of the group are insoluble.
- Moving down the group, oxides and hydroxides change character from acidic → amphoteric → basic.
Halides
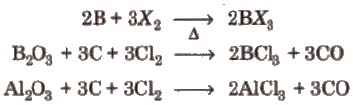
- All members except thallium form trihalides of the type MX3 (X = F, Cl, Br, I).
- Boron trihalides (BF3, BCl3, BBr3, BI3) and many aluminium trihalides are covalent. Anhydrous AlF3 is ionic.
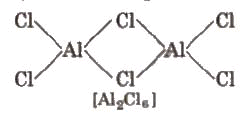
- AlCl3 commonly exists as a dimer (Al2Cl6) under many conditions; boron trihalides are often monomeric because boron is too small to expand coordination to four easily.
- Trihalides of Group 13 are Lewis acids (electron pair acceptors). Relative Lewis acidity among boron trihalides increases as:
BF3 < BCl3 < BBr3 < BI3. - BF3 is less Lewis acidic than expected because of pπ-pπ back bonding from halogen lone pairs into the vacant p-orbital on boron, reducing boron's electron deficiency.
- Acidic character of trihalides across elements: BX3 > AlX3 > GaX3 > InX3.
- TlCl3 is unstable and decomposes to TlCl and Cl2, acting as an oxidising agent:
TlCl3 → TlCl + Cl2
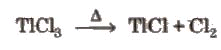
Anomalous Behaviour of Boron
Boron differs markedly from the rest of the group because of the following:
- Smallest atomic and ionic size in the group.
- High ionisation energy.
- Highest electronegativity in the group.
- Absence of low-lying vacant d-orbitals.
Consequences of these differences:
- Boron is a non-metal while the other members are metals.
- Boron shows allotropy; other members do not show comparable allotropy.
- Boron has the highest melting and boiling points in the group because of strong covalent network bonding.
- Boron forms primarily covalent compounds; other members form both ionic and covalent compounds.
- Boron trihalides exist as monomers, whereas AlCl3 often dimerises to Al2Cl6.
- Oxides and hydroxides: boron compounds are weakly acidic, aluminium compounds are amphoteric, and heavier members' compounds are basic.
- Boron is oxidised by concentrated HNO3, whereas aluminium becomes passive because of the protective oxide film on its surface.
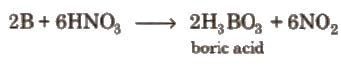
Diagonal Relationship between Boron and Silicon
Boron (in Group 13) shows a diagonal relationship with silicon (Si) in Group 14 - properties resemble because of similar charge/radius ratio and comparable electronegativit
- Both B and Si are semiconductors and show covalent bonding in many compounds.
- Both form covalent hydrides (boranes and silanes).
- Both form volatile covalent halides which hydrolyse to give acidic products that release HCl:
BCl3 + 3H2O → H3BO3 + 3HCl
SiCl4 + 4H2O → Si(OH)4 + 4HCl - Both form solid oxides which dissolve in alkali to give borates and silicates respectively.
- Both react with electropositive metals to give binary compounds that yield mixtures of boranes and silanes on hydrolysis.
Try yourself: Which of the following property is not a similarity between Boron and silicon? Try yourself: Boron shows a diagonal relationship with ____________
Metallurgy of Boron
Occurrence
Boron does not occur free in nature. Important boron minerals include:
- Borax (tincal) - Na2B4O7 · 10H2O
- Kernite - Na2B4O7 · 4H2O
- Orthoboric acid - H3BO3
Isolation
Elemental boron is obtained by reduction or by chemical routes:
- Reduction of boric oxide (B2O3) with highly electropositive metals (K, Mg, Al, Na) in the absence of air to yield boron.
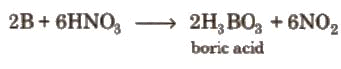
- Reduction of boron halides by hydrogen is another laboratory route to boron.

Uses of Boron
- Used as a semiconductor and as a dopant in electronics.
- Boron isotopes are used in nuclear control applications; boron rods (containing B-10) are used to control nuclear reactions because of the neutron absorption reaction:
5B10 + 0n1 → 5B11
Try yourself: Which of the following is not a use of Boron?
Compounds of Boron
1. Borax (Sodium Tetraborate Decahydrate) - Na2B4O7 · 10H2O
Preparation: Borax (tincal) occurs in dried lake beds. It can be obtained by boiling the mineral colemanite with a solution of Na2CO3. Sodium metaborate (NaBO2) produced may be converted to borax by passing CO2:
4NaBO2 + CO2 → Na2CO3 + Na2B4O7
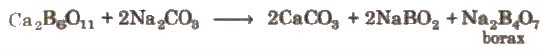
Properties:
- Its aqueous solution is basic:
Na2B4O7 + 7H2O → 2NaOH + 4H3BO3 - With ethanol and concentrated H2SO4 it gives triethyl borate, a volatile compound which burns with a green flame.
- On heating, borax undergoes characteristic changes and is used in the borax bead test for coloured radicals.
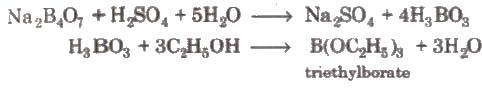
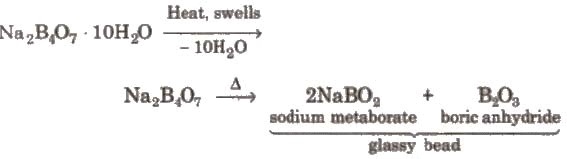
2. Boric Acid (Orthoboric Acid) - H3BO3 or B(OH)3
Preparation: By treating borax with dilute hydrochloric or sulphuric acid:
Na2B4O7 + 2HCl + 5H2O → 2NaCl + 4H3BO3
Properties:
- It is a weak monobasic acid (acts as a Lewis acid in water):
H3BO3 + 2H2O → [B(OH)4]- + H3O+ - With ethanol and conc. H2SO4, it forms triethyl borate.
- Heating causes stepwise loss of water leading to metaboric and then tetraboric acids depending on conditions.
Uses: As an antiseptic and eye lotion (boric lotion), as a preservative in some applications, and in borax/boric acid based applications in glass and ceramics.
3. Borazine (Borazole) - B3N3H6
Borazine is a six-membered ring compound with alternating B and N atoms and is often called "inorganic benzene." It is usually prepared from diborane and ammonia derivatives. Its π electrons are only partly delocalised; borazine is more reactive than benzene.

Compounds of Aluminium1. Anhydrous Aluminium Chloride - AlCl3 (or Al2Cl6)
Preparation:
- It cannot be prepared simply by heating the hydrated salt AlCl3·6H2O.
- It can be prepared by passing dry chlorine or HCl gas over hot aluminium metal.
- It can be prepared by heating a mixture of alumina (Al2O3) and carbon in a current of dry chlorine.
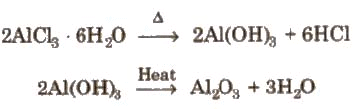
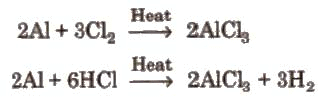
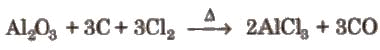
Properties:
- Anhydrous AlCl3 fumes in moist air because it hydrolyses:
AlCl3 + 3H2O → Al(OH)3 + 3HCl - It is a strong Lewis acid and is widely used as a catalyst.
Uses: As a catalyst in Friedel-Crafts reactions and as a mordant in dyeing.
2. Aluminium Oxide (Alumina) - Al2O3
Alumina is the most stable compound of aluminium and occurs naturally as corundum (Al2O3) and coloured varieties used as gemstones: ruby (red), sapphire (blue), etc. Alumina is hard, refractory and chemically inert.
Alums (Double Sulphates)
The term alum refers to hydrated double sulphates of the general formula X2SO4 · Y2(SO4)3 · 24H2O where X is a monovalent cation (K+, Na+, NH4+) and Y is a trivalent cation (Al3+, Cr3+, Fe3+, etc.). Li+ rarely forms alums because it is too small.
Try yourself: What is the chemical formula of alumina and anhydrous aluminium chloride?
Important alums:
- Potash alum: K2SO4 · Al2(SO4)3 · 24H2O
- Sodium alum: Na2SO4 · Al2(SO4)3 · 24H2O
- Ammonium alum: (NH4)2SO4 · Al2(SO4)3 · 24H2O
- Ferric alum: (NH4)2SO4 · Fe2(SO4)3 · 24H2O
- Chrome alum: K2SO4 · Cr2(SO4)3 · 24H2O
Laboratory preparation: Potash alum is obtained by mixing hot equimolar solutions of K2SO4 and Al2(SO4)3, concentrating and crystallising to yield KAl(SO4)2 · 12H2O (empirical formula).
Pseudo Alums
When the monovalent ion in ordinary alums is replaced by a divalent ion such as Mn2+, Fe2+, Mg2+, Cu2+ or Zn2+, the resulting salts are called pseudo alums.
Examples:
- FeSO4 · Al2(SO4)3 · 24H2O (ferrous aluminium pseudo alum)
- MnSO4 · Al2(SO4)3 · 24H2O (manganese aluminium pseudo alum)
Properties of Alums
- Potash alum is a white crystalline solid.
- Aqueous solutions of alums are acidic due to hydrolysis of the trivalent cation:
Al2(SO4)3 + 6H2O → 2Al(OH)3 + 3H2SO4 - On heating, alums lose water of crystallisation and swell up; the anhydrous product is called burnt alum.
- In aqueous solution, alums ionise to give their constituent ions (for potash alum: K+, Al3+, SO42-).
Uses of Alums
- Purification of water (coagulation of suspended impurities).
- Sizing of paper.
- As a styptic to stop bleeding (astringent property).
- As a mordant in dyeing and tanning.

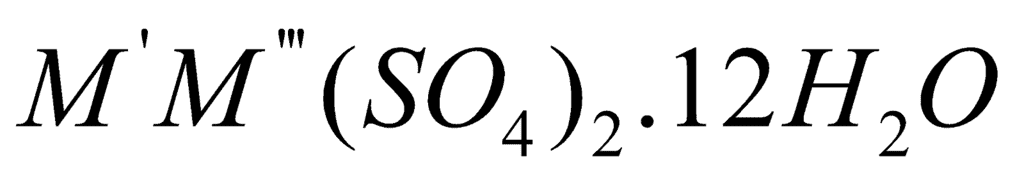
Other Aluminium Compounds and Applications
- Thermite is a mixture of aluminium powder and ferric oxide (Fe2O3) commonly taken in a weight ratio of about 1:3; on ignition it produces intense heat useful for welding and cutting applications.
- Aluminium sulphate, Al2(SO4)3, is used in water purification and for making fireproof cloth via appropriate treatments.
- Aluminium forms many useful alloys with improved mechanical and thermal properties used in transport, packaging, electrical transmission and construction.
Try yourself: Which of the following is used as a catalyst in Friedel craft reaction?
Summary
The Group 13 or boron family shows a progression from non-metallic behaviour (boron) to metallic character down the group. Key features are the ns2 np1 valence configuration, the occurrence of +3 and +1 oxidation states (particularly in heavier members due to the inert pair effect), significant covalent chemistry for boron, and an important technological and industrial role for aluminium and its compounds. Understanding the periodic trends - sizes, ionisation energies, oxidation states, acid-base behaviour of oxides and halides, and anomalies such as those of gallium and thallium - is essential for mastering Group 13 chemistry.
FAQs on Group-13 Elements: Boron Family
| 1. What are Group 13 elements and why is boron called the boron family? |  |
| 2. How do the physical properties of boron differ from aluminium in the Group 13 elements? |  |
| 3. Why does boron form covalent compounds while aluminium forms ionic compounds? |  |
| 4. What are borax and boric acid, and how are they used industrially? |  |
| 5. How does the diagonal relationship between boron and silicon affect their chemical behaviour? |  |







