Group-16 Elements: Oxygen Family
What are Group 16 Elements?
The group 16 elements of the modern periodic table comprise five commonly discussed members: oxygen (O), sulphur (S), selenium (Se), tellurium (Te) and polonium (Po). These elements are also called the chalcogens (from Greek for ore) or the ore-forming elements because many metals are found in nature as their oxides or sulphides and are extracted from these ores.
The name sulphur is historically derived from the Sanskrit word 'Sulvezi', meaning 'killer of copper'. This is part of historical nomenclature.
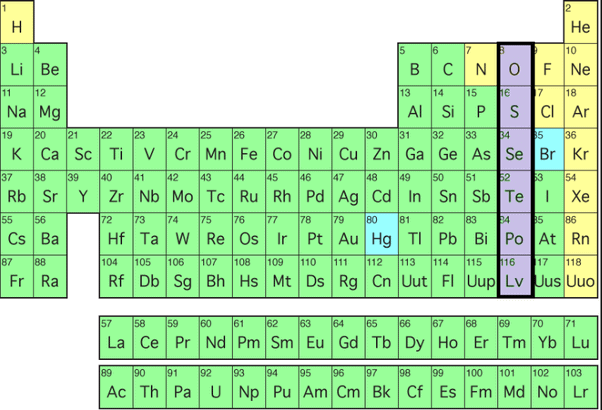
General Physical Properties of Group 16 Elements
- Electronic configuration: The general valence shell electronic configuration is ns2np4. This configuration explains their tendency to gain two electrons to attain a noble gas configuration or to form covalent bonds.
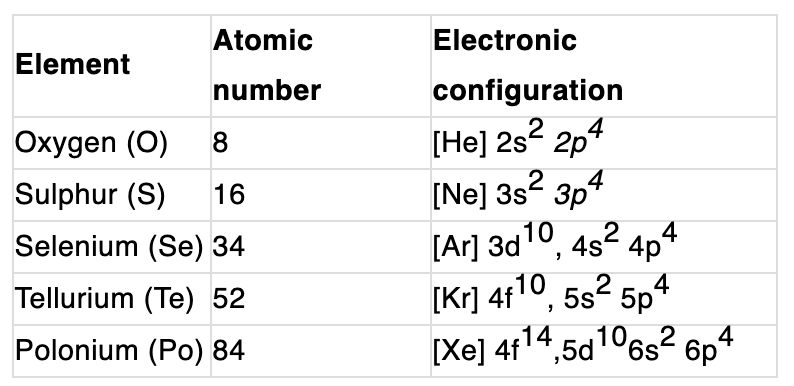
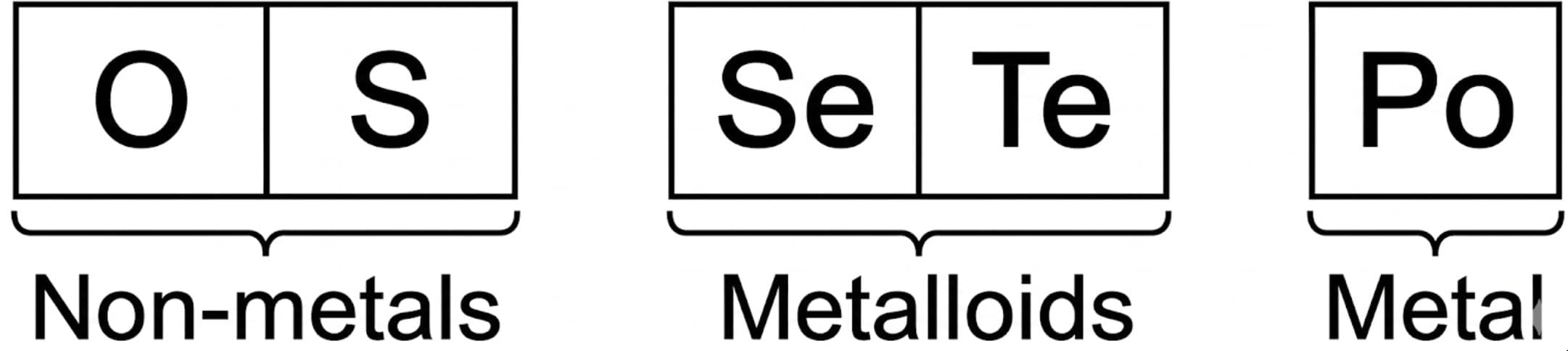
- Metallic and non-metallic character: The lightest members (oxygen, sulphur) are non-metals; selenium and tellurium show metalloid behaviour; polonium is more metallic. Metallic character increases down the group.
- Abundance in Earth's crust: The order of abundance is O > S > Se > Te > Po. Oxygen is the most abundant element by mass in the Earth's crust.
- Density: Density increases regularly down the group due to increasing atomic mass and closer packing in the heavier elements.
- Melting point and boiling point: Both generally increase down the group because of increasing molecular/atomic size and stronger Van der Waals forces in the heavier elements.
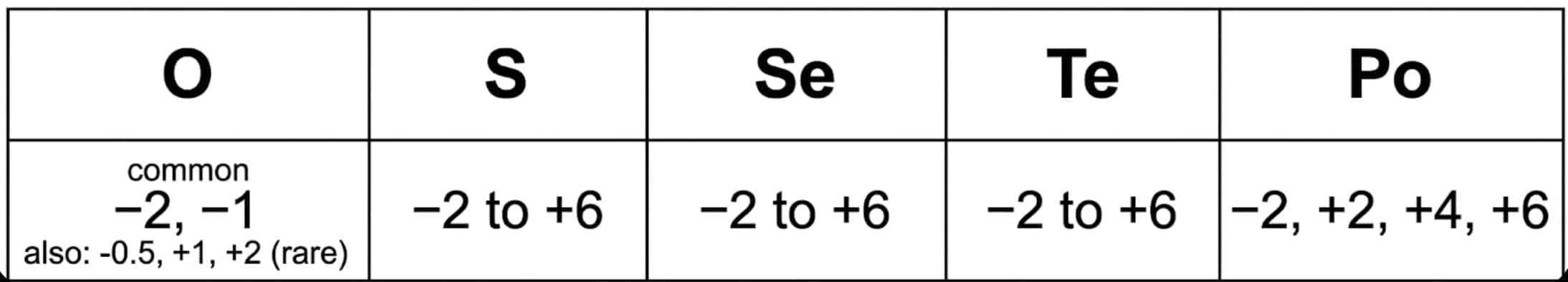
- Oxidation states: Common oxidation states are -2, +2, +4, and +6 for heavier members exhibit multiple positive oxidation states.In OF2, the oxidation state of oxygen is +2.
- Ionisation energy: These elements have relatively high ionisation energies; the first ionisation energy decreases gradually from
O → Po because atomic size increases and outer electrons are more easily removed. - Electron affinity: Electron affinity values are generally high for the light members and decrease from O → Po as the size of the atom increases, the extra added electron feels lesser attraction by the nucleus and hence, electron affinity decreases.
- Electronegativity: Electronegativity decreases down the group due to increased atomic size and shielding, so oxygen is the most electronegative of the group.
- Catenation: The ability to form chains of the same element (catenation) is most pronounced for sulphur and selenium. A typical order of single-bond catenation strength is
S-S > Se-Se > O-O > Te-Te. - Atomicity: Oxygen normally exists as a diatomic molecule, O2. Sulphur and selenium often form cyclic polyatomic molecules; elemental sulphur commonly exists as S8 rings (octatomic).
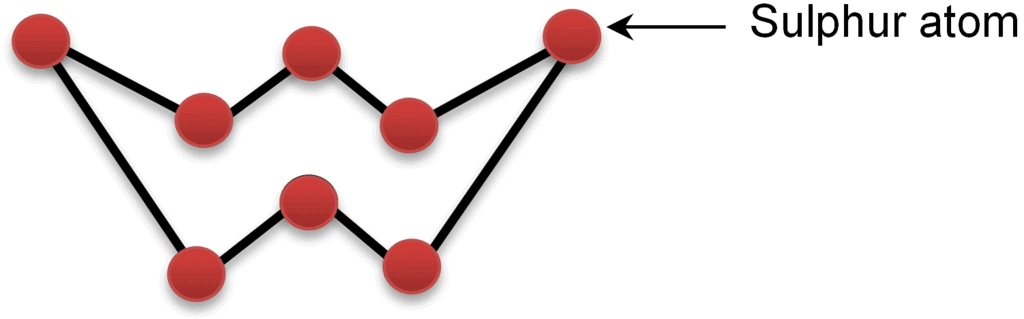
- Allotropy:Some group-16 elements exhibit allotropy - the existence of two or more different forms in the same physical state.
- Oxygen:dioxygen (O2) and ozone (O3).
- Sulphur: several allotropes, commonly rhombic sulphur (α-S8), monoclinic (β-S8), and plastic sulphur.
- Atomic and ionic radii: Atomic and ionic radii increase regularly down the group from O to Po because of the addition of electron shells.
Chemical Properties of Group 16 Elements
Hydrides
All group-16 elements form binary hydrides of the general type H2E (E = O, S, Se, Te, Po). The properties change down the group:
Examples of reactions:
2H2 + O2 ⇌ 2H2OFeS + H2SO4 → H2S + FeSO4
H2O is a liquid under standard conditions due to extensive hydrogen bonding. The heavier hydrides (H2S, H2Se, H2Te) are gases or volatile liquids with unpleasant odours and show weaker or negligible hydrogen bonding.
Acidic character of corresponding hydrides increases down the group: H2O is amphoteric, while H2S, H2Se are more acidic in aqueous solution. Except for water, these hydrides are generally reducing in character and reducing ability increases H2S → H2Se → H2Te.
Halides
Group-16 elements form a variety of halides.
- For the halide ions, the relative ease of formation or stability is often given in the series F- > Cl- > Br- > I- (this refers to reactivity/stability contexts in specific comparisons).
- Among hexahalides, the hexafluorides (EF6) are the most stable; for example, SF6 is exceptionally inert and stable because of steric factors and an electronic configuration that resists reaction.
- Physical states of tetrafluorides vary with element: SF4 (gas), SeF4 (liquid), TeF4 (solid). These tetrafluorides adopt see-saw geometries consistent with sp3d-type descriptions.They behave Lewis acid as well as Lewis base e.g.,
SF4 + BF3 → SF4 → BF3
SeF4 + 2F- → [SeF6]2- - Many mono halides (for example, S2Cl2, Se2Cl2) are dimeric and can disproportionate; e.g.:
2SeCl2 → SeCl4 + Se
Oxides
Group-16 elements form oxides with different stoichiometries; common types are AO2 and AO3 (A = element).
Ozone (O3) can be considered an oxide form of oxygen in thecontext of allotropy and reactivity.
SO2 is a bent molecule consistent with sp2 hybridisation at sulphur (V-shaped).
SO3 is planar and consistent with sp2 description; in gas phase and as molecules it is trigonal planar.
SeO2 is a volatile solid that forms polymeric non-planar chains in the solid state.
SeO3 is known to exist as a tetrameric cyclic structure in the solid state.
The acidic character (of oxides acting as acidic oxides) generally follows:
SO2 > SeO2 > TeO2 > PoO2 and for trioxides: SO3 > SeO3 > TeO3
Specific structural points:
SO2 and SO3 are the anhydrides of sulphurous acid (H2SO3, unstable) and sulphuric acid (H2SO4), respectively.
Try yourself: Which of the following can classified as an amphoteric oxide?
Note: Selenium is used as a photoconductor in some photocopying and photocopier devices because its electrical conductivity changes on exposure to light.
Oxygen and its Compounds :
Dioxygen:
Historical note: Joseph Priestley and Carl Wilhelm Scheele independently prepared oxygen by heating suitable oxygen-containing compounds in the 18th century; later Antoine Lavoisier named the element and clarified its role in combustion.
Preparation (laboratory/industrial methods): Dioxygen is prepared by methods that liberate O2 from oxygen-rich compounds. Common laboratory and industrial methods include:
(i) From oxides: (ii) From peroxides and other oxides:
(ii) From peroxides and other oxides: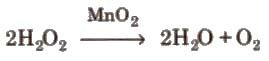
(iii) From certain compounds: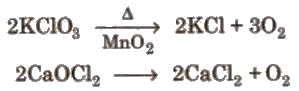
Physical properties: O2 is a colourless, odourless, tasteless gas, slightly heavier than air and sparingly soluble in water.
Chemical properties: On heating, oxygen combines directly with many elements including metals and non-metals to give oxides.
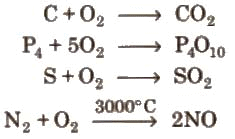
Examples:
2Mg + O2 → 2MgO
4Na + O2 → 2Na2O (or with excess O2 to peroxides such as Na2O2)
Combustion and combination reactions with O2 can be catalysed; platinum is an effective catalyst for some oxygenation reactions.
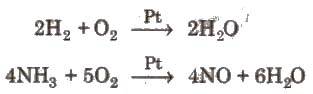
Uses: Oxygen is used in metal cutting and welding (oxy-acetylene, oxy-hydrogen torches), in steelmaking to increase furnace temperatures and oxidation of impurities, and for life-support systems in healthcare (medical oxygen), diving, aerospace and related applications.
Tests for oxygen:
1. With NO it gives reddish-brown fumes of NO2.
2. It is adsorbed by alkaline pyrogallol.
Try yourself: Which of the following in air, forms compounds readily?
Ozone (O3)
Preparation: Ozone is prepared by passing a silent electric discharge through dry oxygen; this converts some O2 into O3. The equilibrium can be written as:
3O2 ⇌ 2O3 ΔH = +284.3 kJ (per mole of ozone formed under the convention used here)
Physical properties: Ozone is a pale blue gas with a characteristic sharp smell; it is slightly soluble in water and more soluble in some organic solvents than O2.
Chemical properties:
1. Decomposition:
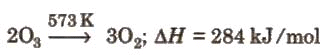
2. Oxidising action: Ozone is a powerful oxidising agent and reacts with a variety of inorganic and organic substances.
Ozone liberates iodine from aqueous iodide; this reaction is used as a qualitative test for strong oxidising agents:
2KI + H2O + O3 → 2KOH + I2 + O2
I2 + starch → blue colour (starch paper test).
Uses of ozone:
1. as a germicide and disinfectant for sterilizing water.
2. as a bleaching agent for oils, ivory wax and delicate fibres.
3. for detecting 'the position of the double bond in unsaturated compounds.
4. in destroying odours coming from the cold storage room, slaughterhouses and kitchen of hotels.
Compounds of Sulphur
Sulphur Dioxide (SO2)
Methods of preparation:
(i) By burning sulphur in air:
(ii) By roasting iron pyrites (FeS2) in excess air:
(iii) Laboratory method:
Physical properties: SO2 is a colourless gas with a pungent, suffocating odour and is highly soluble in water; it forms sulphurous acid solutions.
Chemical reactions and properties:
SO2 reacts with lime water to give calcium sulphite (milkiness). On excess SO2 the milkiness disappears due to formation of soluble bisulphite:
Ca(OH)2 + SO2 → CaSO3 + H2O (milky suspension)
CaSO3 + SO2 + H2O → Ca(HSO3)2 (soluble)
Other reactions:
2NaOH + SO2 → Na2SO3 + H2O
Na2SO3 + H2O + SO2 → 2NaHSO3
SO2 + Cl2 → SO2Cl2 (sulphuryl chloride)
2SO2 + O2 → 2SO3 (oxidation to sulphur trioxide)
Reducing behaviour: SO2 acts as a reducing agent in several reactions; for example:
2Fe3+ + SO2 + 2H2O → 2Fe2+ + SO42- + 4H+
5SO2 + 2MnO4- + 2H2O → 5SO42- + 4H+ + 2Mn2+
Wackenroder's reaction: When H2S is passed through an acidified solution containing dissolved SO2, certain intermediate sulphur species form and the solution may turn milky; this sequence of observations is historically referred to in descriptive texts as Wackenroder's reaction.
Oxoacids of Sulphur
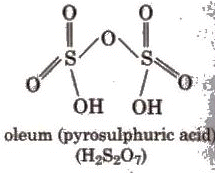
Sulphur forms several oxoacids; classical ones include sulphurous acid (H2SO3, unstable in free form), sulphuric acid (H2SO4), and related species (e.g., disulphuric/pyrosulphuric acid, H2S2O7). Structural representations and relationships among these oxoacids are commonly shown in standard textbooks.
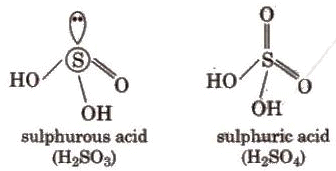
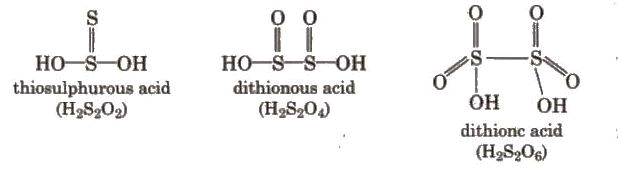
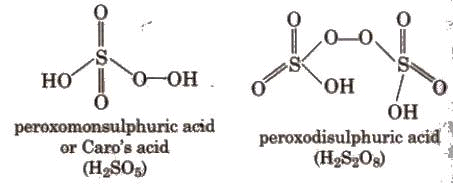
Sulphuric Acid (H2SO4)
Importance: Sulphuric acid is one of the most important industrial chemicals and is often described as the "king of chemicals" because its production is an indicator of industrial activity.
Manufacture (Contact process - main industrial route):
(i) Burning of sulphur or sulphur ores in air to generate SO2.
(ii) Conversion of SO2 to SO3 by the reaction with oxygen in the presence of a catalyst (V2O5).
(iii) Absorption of SO3 in H2SO4 to give oleum (H2S2O7) which upon hydrolysis gives H2SO4.
Properties:
1. Sulphuric acid is a colourless, dense, oily liquid.
MX + H2SO4 → 2HX + M2SO4
2. Concentrated sulphuric acid is a strong dehydrating agent. The burning sensation of concentrated H2SO4 on skin.
The burning sensation of concentrated H2SO4 on skin.
3. Hot concentrated sulphuric acid is a moderately strong oxidising agent. In this respect, it is intermediate between phosphoric acid and nitric acid.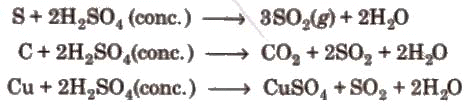
Uses: Manufacture of fertilizers (e.g., superphosphate), petroleum refining, pigments and dyes, detergents, explosives, and many other industrial chemicals.
Sodium Thiosulphate (Hypo) - Na2S2O3·5H2O
Common name: Hypo (sodium thiosulphate pentahydrate). It is an efflorescent crystalline solid that dissolves readily in water and is widely used in photographic fixing and in analytical chemistry.
Preparations:
1. By boiling a solution of sodium sulphite with excess flowers of sulphur until the alkaline reaction disappears:
Na2SO3 + S → Na2S2O3
2. By the "Spring's reaction" (historical preparative method):
Na2S + Na2SO3 + I2 → Na2S2O3 + 2NaI
Properties:
(i). It is a colourless, crystalline and efflorescent substance.
(ii). It gives white ppt with a dilute solution of AgNO3 which quickly changes into black due to the formation of Ag2S.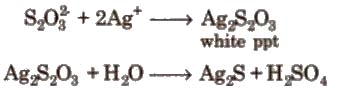
Uses:
1. Due to its property of dissolving silver halide, it is used in photography for fixing under the name hypo.
2Na2S2O3 + AgBr → Na3 [ Ag(S2O3)2] + NaBr
2. During bleaching, it is used as an antichlor.
Na2S2O3 + CI2 + H2O → Na2SO4 + S + 2HCI
3. It is used to remove iodine stain, for volumetric estimation of iodine and in medicines.
Try yourself: Which of the following is the most popular oxoacid of sulfur?
FAQs on Group-16 Elements: Oxygen Family
| 1. What are the general physical properties of Group 16 elements? |  |
| 2. What are the chemical properties of Group 16 elements? |  |
| 3. What are some compounds of oxygen and their significance? |  |
| 4. What are some compounds of sulfur and their importance? |  |
| 5. How are the Group 16 elements related to the Oxygen family? |  |






